Advanced Manufacturing of Pentafluoroaniline via Optimized Hofmann Degradation for Global Supply Chains
Introduction to Patent CN103012162A and Technological Breakthroughs
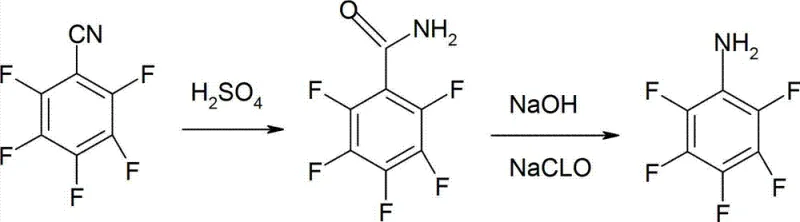
The chemical industry constantly seeks more efficient pathways for producing critical intermediates, and the preparation method detailed in patent CN103012162A represents a significant leap forward in the synthesis of pentafluoroaniline (CAS 771-60-8). This compound serves as a vital building block for pharmaceuticals, agrochemicals, and liquid crystal materials, yet its historical production has been plagued by complex multi-step sequences and hazardous reagents. The disclosed innovation introduces a streamlined two-step protocol starting from pentafluorobenzonitrile, utilizing concentrated sulfuric acid for hydrolysis followed by a classic Hofmann degradation. By shifting the synthetic entry point to a nitrile precursor, the methodology circumvents the severe safety risks and supply chain bottlenecks associated with perchlorobenzene fluorination or the costly activation of pentafluorobenzoic acid. For R&D directors and procurement specialists alike, this patent offers a compelling blueprint for achieving high-purity outputs exceeding 99% while maintaining yields consistently above 80%, thereby establishing a new benchmark for economic and operational efficiency in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pentafluoroaniline has relied on pathways that are increasingly untenable for modern, cost-sensitive supply chains. One prevalent traditional route involves the fluorination of perchlorobenzene, a process severely hampered by the scarcity of the raw material and the extreme reaction conditions required to introduce fluorine atoms onto the aromatic ring. Alternatively, another common method utilizes pentafluorobenzoic acid as a starting material, necessitating conversion to an acid chloride followed by reaction with hydroxylamine hydrochloride to form a hydroxamic acid intermediate before final hydrolysis. This latter approach is particularly disadvantageous due to its excessive length, involving multiple isolation steps that accumulate yield losses and generate substantial volumes of saline waste. Furthermore, the reliance on specialized reagents like hydroxylamine hydrochloride drives up raw material costs significantly, while the harsh chlorination steps pose serious corrosion challenges for standard reactor equipment, leading to increased maintenance downtime and capital expenditure for specialized alloy vessels.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach outlined in the patent leverages pentafluorobenzonitrile as a robust and commercially accessible feedstock to drastically simplify the molecular construction. The process initiates with a direct acid-catalyzed hydrolysis using sulfuric acid concentrations between 70% and 98%, converting the nitrile group directly into an amide functionality under relatively mild thermal conditions of 70-120°C. This eliminates the need for activating agents like thionyl chloride or phosphorus pentachloride, thereby reducing both reagent costs and the generation of acidic gaseous byproducts. The subsequent transformation employs a Hofmann degradation strategy using sodium hydroxide and sodium hypochlorite, a reagent combination that is inexpensive, widely available, and easy to handle compared to the exotic catalysts often required in cross-coupling alternatives. This strategic pivot not only shortens the overall synthetic timeline but also enhances the safety profile of the operation, making it an ideal candidate for reliable pentafluoroaniline supplier networks aiming to secure long-term production stability.
Mechanistic Insights into Acid-Catalyzed Hydrolysis and Hofmann Degradation
The core chemical elegance of this process lies in the precise orchestration of two fundamental organic transformations that maximize atom economy while minimizing side reactions. In the first stage, the electron-deficient nature of the pentafluorobenzonitrile ring facilitates nucleophilic attack by water molecules activated by the strong protonic acid environment of sulfuric acid. The reaction proceeds through an imidate intermediate which rapidly tautomerizes to the stable pentafluorobenzamide, a solid that precipitates upon dilution with water, allowing for easy physical separation from the acidic mother liquor without the need for energy-intensive solvent extraction. This precipitation mechanism is crucial for impurity control, as it inherently excludes soluble inorganic salts and unreacted starting materials, setting the stage for a high-purity downstream process. The control of temperature between 70°C and 120°C is critical; too low and the hydrolysis stalls, too high and potential defluorination or polymerization could occur, though the patent data suggests a wide operating window that provides robust process tolerance.
The second mechanistic phase involves the Hofmann rearrangement, where the amide nitrogen is converted into an isocyanate intermediate via N-halogenation and subsequent base-induced elimination. By maintaining the initial reaction temperature between -10°C and 20°C during the addition of the hypohalite, the formation of the N-chloroamide is controlled to prevent over-oxidation or decomposition of the sensitive perfluorinated ring system. Upon heating to 70-110°C, the rearrangement occurs with migration of the aryl group from carbon to nitrogen, releasing carbon dioxide and generating the free amine. The use of steam distillation as the final isolation technique is particularly insightful for this specific molecule; since pentafluoroaniline possesses sufficient volatility with steam, this method allows for the separation of the product from non-volatile polymeric tars and inorganic salts without requiring organic solvents, thus aligning perfectly with green chemistry principles and reducing the environmental footprint of cost reduction in pharmaceutical intermediates manufacturing.
Furthermore, the choice of sodium hypochlorite (bleach) as the oxidant offers a distinct advantage over elemental bromine or chlorine gas, which are difficult to meter and pose significant inhalation hazards. The in situ generation of the active halogenating species ensures a homogeneous reaction environment, promoting consistent conversion rates across large batches. The stoichiometry is carefully balanced, with a molar ratio of alkali to amide preferably between 1:1.5 and 1:4.5, ensuring that the basic conditions necessary for the rearrangement are maintained throughout the exothermic process. This mechanistic understanding allows process chemists to fine-tune the addition rates and cooling capacities to manage the thermal profile effectively, ensuring that the commercial scale-up of complex fluorinated intermediates remains safe and predictable. The result is a product with a gas chromatography purity of greater than 99%, demonstrating that high selectivity can be achieved without resorting to expensive chromatographic purification techniques.
How to Synthesize Pentafluoroaniline Efficiently
Implementing this synthesis route requires careful attention to the specific parameters outlined in the patent embodiments to ensure reproducibility and optimal yield. The process begins with the preparation of the hydrolysis mixture, where the weight ratio of pentafluorobenzonitrile to sulfuric acid is optimized between 1:3 and 1:6 to ensure complete dissolution and reaction kinetics. Operators must monitor the reaction progress via HPLC or GC until the nitrile content drops below 1%, indicating full conversion to the amide. Following filtration and washing of the amide cake, the second step requires precise thermal management during the hypohalite addition to prevent runaway exotherms. The detailed standardized synthesis steps, including specific stirring speeds, addition times, and workup procedures validated at the 1000L scale, are provided below to guide your technical team in replicating these results.
- Hydrolyze pentafluorobenzonitrile in 70-98% sulfuric acid at 70-120°C for 3-5 hours to obtain pentafluorobenzamide.
- Mix the resulting amide with alkali solution and hypohalite (e.g., NaClO), maintaining -10 to 20°C for 4-6 hours.
- Heat the mixture to 70-110°C for 0.5-3 hours to complete Hofmann degradation, followed by steam distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this nitrile-based hydrolysis route offers profound strategic benefits that extend beyond simple unit price considerations. The primary driver of value is the substitution of scarce and expensive raw materials with commodity chemicals; pentafluorobenzonitrile is increasingly available from global fluorine chemical producers, whereas perchlorobenzene faces tightening regulatory restrictions and supply volatility. By adopting this method, manufacturers can decouple their production schedules from the bottlenecks of specialized halogenated precursors, thereby enhancing supply chain reliability and reducing the risk of stockouts that can halt downstream API production lines. Additionally, the simplification of the workflow from a multi-step sequence to a two-pot process significantly reduces labor hours and utility consumption, translating into substantial cost savings that can be passed down to customers or reinvested into capacity expansion.
- Cost Reduction in Manufacturing: The elimination of expensive activating agents such as thionyl chloride and hydroxylamine hydrochloride removes a major cost center from the bill of materials. Furthermore, the use of sulfuric acid and sodium hypochlorite, which are bulk commodities with stable pricing, insulates the production cost from the fluctuations typical of fine chemical reagents. The high yield reported in the patent, reaching upwards of 80% on a multi-hundred-kilogram scale, ensures that raw material utilization is maximized, minimizing the cost per kilogram of the final active ingredient. This efficiency is compounded by the ability to recover and recycle the aqueous acid streams, further driving down the variable costs associated with waste treatment and neutralization chemicals.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the wide temperature tolerance for the hydrolysis step (70-120°C), means that the process is less susceptible to minor deviations in heating or cooling utility performance. This operational flexibility is critical for maintaining consistent output in large-scale facilities where equipment variability can sometimes impact batch quality. Moreover, the solid intermediate, pentafluorobenzamide, can be isolated, dried, and stored, providing a buffer inventory that allows for flexible scheduling of the second degradation step. This decoupling of the two stages allows supply chain planners to optimize reactor usage and manage logistics more effectively, ensuring reducing lead time for high-purity pentafluoroaniline deliveries to key pharmaceutical clients.
- Scalability and Environmental Compliance: The patent explicitly validates the process at the 1000L reactor scale, demonstrating that heat transfer and mixing issues do not impede performance as volume increases. From an environmental perspective, the avoidance of heavy metal catalysts and the minimization of organic solvent use through water-based crystallization and steam distillation significantly lowers the facility's environmental burden. The "three wastes" (waste water, gas, and solids) are described as minimal, which simplifies the permitting process for new production lines and reduces the ongoing costs of environmental compliance and waste disposal. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor increasingly weighted in vendor selection criteria by multinational corporations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries regarding the process specifics and quality attributes. These responses are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is essential for assessing the compatibility of this route with current GMP standards and facility capabilities.
Q: What are the primary advantages of this synthesis route over traditional methods?
A: Unlike traditional methods using perchlorobenzene or pentafluorobenzoic acid which suffer from harsh conditions and expensive reagents, this route uses readily available pentafluorobenzonitrile and achieves yields over 80% with purity exceeding 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability from laboratory glassware to 1000L reactors, highlighting simple operations, convenient post-treatment via steam distillation, and minimal three-waste discharge.
Q: How is product purity controlled in this method?
A: High purity (>99%) is ensured through precise temperature control during the Hofmann degradation and the final purification step involving steam distillation, which effectively separates the volatile aniline from non-volatile impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pentafluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced fluorinated intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand the critical nature of stringent purity specifications in the pharmaceutical and agrochemical sectors, which is why our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every batch against the highest international standards. By leveraging the optimized hydrolysis and Hofmann degradation route, we are positioned to offer a supply solution that balances cost-effectiveness with uncompromising quality assurance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume requirements and quality targets. Whether you require a Customized Cost-Saving Analysis comparing this route to your current supply chain or need to review specific COA data and route feasibility assessments for your next project, we are ready to provide the support you need. Partnering with us ensures access to a stable, scalable, and economically viable source of high-purity pentafluoroaniline, empowering your R&D and production teams to focus on what they do best: developing life-changing therapies and sustainable agricultural solutions.
