Scaling High-Performance Metalloporphyrin-Triphenylamine Semiconductors for Commercial Optoelectronics
The landscape of organic optoelectronics is rapidly evolving, driven by the demand for materials that combine high charge carrier mobility with robust environmental stability. Patent CN102453233A introduces a groundbreaking class of organic semiconductor materials featuring a unique hybrid architecture: a metalloporphyrin-triphenylamine backbone integrated with silafluorene units. This structural innovation addresses critical bottlenecks in current photovoltaic and electroluminescent technologies by synergizing the high quantum yield of porphyrins with the superior hole-transport properties of triphenylamines. The resulting polymer, defined by structural formula (I), exhibits a tunable band gap and extended absorption into the near-infrared region, making it an ideal candidate for next-generation organic solar cells and OLED displays.
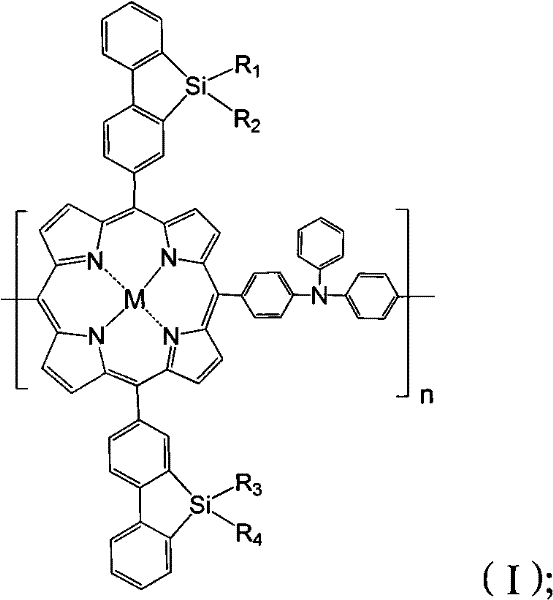
For R&D directors and procurement specialists, the significance of this patent lies not just in performance metrics but in the versatility of the molecular design. The variable substituents R1 through R4 allow for precise modulation of solubility and film-forming properties, ranging from short methyl groups to long C32 alkyl chains. This adaptability ensures that the material can be processed using standard solution-based techniques, a crucial factor for reducing manufacturing costs in large-area electronics. By leveraging the specific coordination chemistry of metal ions such as Zn²⁺, Cu²⁺, or Fe²⁺ within the porphyrin core, manufacturers can tailor the electronic properties to specific device requirements without altering the fundamental synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organic semiconductor materials often suffer from a trade-off between stability and processability. Conventional porphyrin-based polymers, while possessing excellent photoelectric properties, frequently exhibit poor solubility in common organic solvents, necessitating harsh processing conditions or complex side-chain engineering that drives up production costs. Furthermore, standard triphenylamine derivatives, though effective hole transporters, often lack the broad spectral absorption required for high-efficiency light harvesting in solar applications. The spectral response of many legacy materials does not adequately match the solar radiation spectrum, leading to significant energy losses. Additionally, the thermal stability of purely organic conjugated systems can be insufficient for the rigorous thermal budgets encountered during device encapsulation and operation, limiting their commercial lifespan and reliability in outdoor environments.
The Novel Approach
The methodology outlined in CN102453233A overcomes these deficiencies through a strategic integration of silafluorene moieties into the porphyrin-triphenylamine framework. This novel approach utilizes the silicon atom's ability to modify the electronic structure of the conjugated system, resulting in higher electron affinity and improved electron injection capabilities. The synthesis begins with the condensation of dipyrromethane and silafluorene-aldehyde derivatives, a reaction that constructs the robust macrocyclic core essential for charge delocalization. 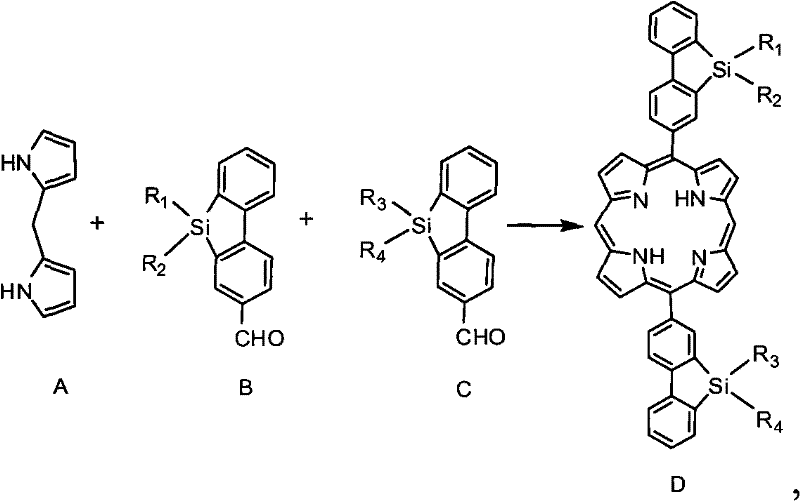 By incorporating long alkyl or alkoxy chains at the silicon center, the material achieves excellent solubility without compromising the planarity of the conjugated backbone. This structural design effectively prevents close intermolecular pi-stacking that leads to undesirable crystallization, promoting the formation of stable amorphous films which are critical for uniform device performance.
By incorporating long alkyl or alkoxy chains at the silicon center, the material achieves excellent solubility without compromising the planarity of the conjugated backbone. This structural design effectively prevents close intermolecular pi-stacking that leads to undesirable crystallization, promoting the formation of stable amorphous films which are critical for uniform device performance.
Mechanistic Insights into Suzuki Coupling Polymerization and Metal Coordination
The core of the commercial synthesis relies on a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, a robust and widely understood transformation in industrial organic chemistry. In the final polymerization step, a dibromo-silafluorene metalloporphyrin derivative reacts with a bis-boronate triphenylamine monomer. This step is critical as it links the electron-rich triphenylamine units with the electron-deficient metalloporphyrin cores, creating a donor-acceptor type conjugated polymer. The reaction typically proceeds in solvents like toluene or THF at temperatures between 50°C and 120°C, using catalysts such as Pd(PPh₃)₂Cl₂ or Pd(PPh₃)₄. The mechanism involves the oxidative addition of the palladium catalyst to the carbon-bromine bond of the porphyrin, followed by transmetallation with the boronate species and reductive elimination to form the new carbon-carbon bond, extending the polymer chain.
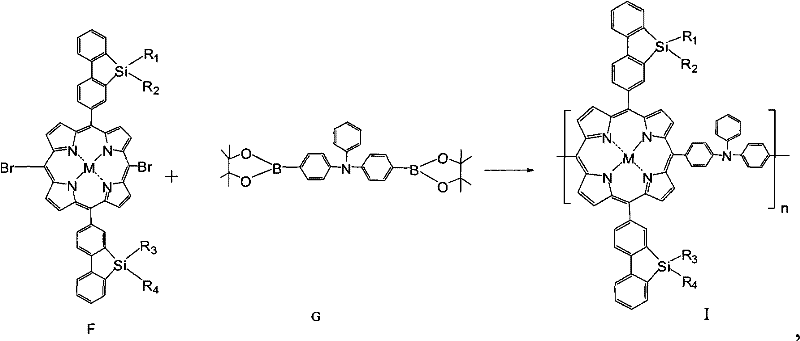
Beyond the polymerization mechanics, the metalation step plays a pivotal role in defining the material's optoelectronic profile. The patent details the coordination of various metal ions (M) into the porphyrin cavity, a process that occurs under mild conditions (0-30°C) using metal salts like zinc acetate or copper sulfate. This coordination saturates the nitrogen lone pairs within the porphyrin ring, altering the HOMO-LUMO energy levels and enhancing the rigidity of the macrocycle. This rigidity reduces non-radiative decay pathways, thereby improving the quantum yield of charge transfer. For supply chain managers, this modularity is advantageous; the same polymer backbone can be metallated with different ions post-synthesis or during the monomer stage to produce a library of materials with distinct emission colors or absorption profiles from a single set of intermediates.
How to Synthesize Metalloporphyrin-Triphenylamine Polymers Efficiently
The synthesis protocol described in the patent offers a clear, stepwise route suitable for scale-up, beginning with the preparation of the boronate monomer and concluding with the final coupling. The process is designed to maximize yield while minimizing impurities through careful control of reaction stoichiometry and temperature. Detailed operational parameters, including specific solvent choices and workup procedures, are critical for achieving the high molecular weights necessary for optimal film formation.
- Synthesize the bis-boronate triphenylamine monomer via lithiation of dibromo-triphenylamine at -78°C followed by quenching with pinacol boronic ester.
- Condense dipyrromethane with silafluorene-aldehyde derivatives using acid catalysis and DDQ oxidation to form the porphyrin macrocycle.
- Perform bromination of the porphyrin intermediate, followed by metalation with transition metal salts, and finally execute Suzuki coupling polymerization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this silafluorene-porphyrin technology offers substantial strategic advantages over legacy semiconductor materials. The synthetic route utilizes commodity chemicals such as dibromotriphenylamine, pyrrole, and standard silafluorene derivatives, ensuring a stable and diverse supply base. The reliance on established catalytic cycles like Suzuki coupling means that existing manufacturing infrastructure can often be adapted with minimal capital expenditure. Furthermore, the enhanced solubility imparted by the silafluorene alkyl groups simplifies downstream processing, eliminating the need for expensive high-boiling solvents or complex purification trains that are often required for insoluble conjugated polymers.
- Cost Reduction in Manufacturing: The streamlined synthesis eliminates the need for specialized, high-cost monomers often associated with high-performance polymers. By using readily available silafluorene precursors and efficient palladium catalysis, the overall cost of goods sold is significantly optimized. The high yields reported in the embodiments, such as the 92% yield in the boronate synthesis and up to 97% in metalation steps, indicate a process with minimal waste generation. This efficiency translates directly to lower raw material consumption and reduced waste disposal costs, providing a clear economic advantage in high-volume electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of monomer production from polymerization. Intermediates like the dibromo-porphyrin can be stockpiled, buffering against fluctuations in the supply of final catalysts or coupling partners. Additionally, the use of robust chemical bonds (C-C and Si-C) ensures the material has a long shelf life, reducing the risk of inventory degradation. The ability to source alternative metal salts (e.g., switching between Zinc and Copper based on availability) further de-risks the supply chain against specific commodity shortages.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and pressures, avoiding the extreme conditions that often complicate scale-up and increase energy consumption. The solvents employed, primarily chlorinated hydrocarbons and aromatics, are standard in the fine chemical industry and have well-established recycling protocols. The high atom economy of the condensation and coupling reactions minimizes byproduct formation. Consequently, scaling this technology from laboratory grams to metric tons presents fewer engineering hurdles compared to more exotic deposition techniques, facilitating a smoother transition to commercial production while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organic semiconductor technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for feasibility assessments.
Q: What are the primary advantages of incorporating silafluorene units into porphyrin polymers?
A: According to patent CN102453233A, introducing silafluorene units significantly enhances thermal stability and electron affinity while lowering the LUMO energy level, which improves electron injection and transport capabilities compared to traditional carbon-based analogues.
Q: How does this material improve solar cell efficiency?
A: The material features a widened visible spectrum absorption region that extends into the near-infrared due to the conjugated porphyrin system. This better matches the solar radiation spectrum, thereby increasing the sunlight utilization ratio and carrier mobility in photovoltaic devices.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation method utilizes standard organic synthesis techniques such as Suzuki coupling and acid-catalyzed condensation. The process is described as simple to operate and control, utilizing commercially available catalysts like palladium complexes and common solvents like THF and toluene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metalloporphyrin-Triphenylamine Polymer Supplier
As the global demand for high-efficiency organic photovoltaics and flexible displays accelerates, securing a supply of advanced semiconductor materials is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in complex organic synthesis to deliver high-purity metalloporphyrin-triphenylamine polymers. Our facilities are equipped to handle the nuanced requirements of organometallic chemistry, ensuring that every batch meets stringent purity specifications essential for electronic applications. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical capability to translate the promising laboratory results of CN102453233A into reliable industrial reality.
We invite procurement leaders and R&D teams to collaborate with us on optimizing this supply chain. Whether you require custom substitution patterns to tune band gaps or need to secure a long-term supply of the zinc or copper variants, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis, specific COA data, and comprehensive route feasibility assessments tailored to your device architecture. Let us help you engineer the next generation of optoelectronic devices with materials that combine performance, stability, and cost-effectiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
