Advanced Cadmium Complex Dye Sensitizers for High-Efficiency Solar Cell Manufacturing
Advanced Cadmium Complex Dye Sensitizers for High-Efficiency Solar Cell Manufacturing
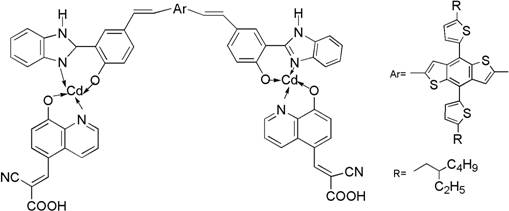
The rapid evolution of renewable energy technologies has placed dye-sensitized solar cells (DSSCs) at the forefront of photovoltaic research, driven by their potential for low-cost production and high conversion efficiency. A pivotal advancement in this field is documented in patent CN112209951B, which introduces a novel cadmium complex dye sensitizer known as BDTT-bi-Cd. This sophisticated molecular architecture leverages a unique D (-A-pi-A) 2 configuration, integrating benzimidazole derivatives and 8-hydroxyquinoline derivatives coordinated with cadmium. The resulting material demonstrates a remarkable Photoelectric Conversion Efficiency (PCE) of 8.40%, coupled with exceptional thermal stability exceeding 320°C. For industrial stakeholders, this represents a significant leap forward in developing robust, high-performance optoelectronic materials capable of meeting the rigorous demands of commercial solar applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional dye sensitizers often rely on simpler D-pi-A (Donor-pi-Acceptor) architectures, which, while effective, frequently encounter limitations in optimizing the balance between electron injection and dye regeneration. In many classic organic dyes, the electron-pushing ability of the donor and the electron-pulling capability of the acceptor are difficult to fine-tune independently, leading to suboptimal intramolecular charge transfer efficiency. Furthermore, standard organic sensitizers often suffer from insufficient thermal stability, which can compromise the longevity of the solar cell under operational heat stress. The lack of auxiliary acceptors in these conventional designs restricts the ability to effectively lower the energy gap between the LUMO and HOMO levels, thereby limiting the breadth of the light absorption spectrum. Consequently, manufacturers face challenges in scaling up production of dyes that can consistently deliver both high efficiency and long-term durability in harsh environmental conditions.
The Novel Approach
The innovative strategy outlined in the patent addresses these deficiencies by introducing a cadmium complex as an auxiliary electron acceptor within a D (-A-pi-A) 2 framework. By incorporating the metal complex, the design significantly enhances the electron-withdrawing capacity of the acceptor moiety, which facilitates more efficient intramolecular charge transfer and broadens the absorption range into the visible spectrum. This structural modification not only improves the photovoltaic performance but also imparts superior thermal stability to the molecule, ensuring it remains intact at temperatures above 320°C. The use of benzodithiophene bithiophene (BDTT) as the electron donor further optimizes the electronic properties, creating a synergistic effect that maximizes light harvesting. This approach provides a robust pathway for developing next-generation sensitizers that overcome the intrinsic limitations of purely organic D-pi-A systems.
Mechanistic Insights into Heck Coupling and Coordination Chemistry
The synthesis of the BDTT-bi-Cd complex relies on a precise sequence of coordination chemistry and cross-coupling reactions, culminating in a palladium-catalyzed Heck coupling. The process begins with the formation of key ligands, such as 2-cyano-3-(8-hydroxyquinolin-5-yl)acrylic acid, which acts as the anchoring group and primary acceptor. These ligands are then coordinated with cadmium acetate to form the intermediate bi-Cd complex, establishing the core metal-organic framework. The final and most critical step involves the coupling of this cadmium-containing intermediate with the BDTT donor derivative. This reaction utilizes a palladium catalyst, specifically palladium acetate, along with tri(2-methylphenyl)phosphine as a ligand to facilitate the carbon-carbon bond formation. The reaction is conducted in DMF solvent with triethylamine as a base at elevated temperatures, ensuring high conversion rates and the formation of the extended pi-conjugated system essential for efficient electron transport.
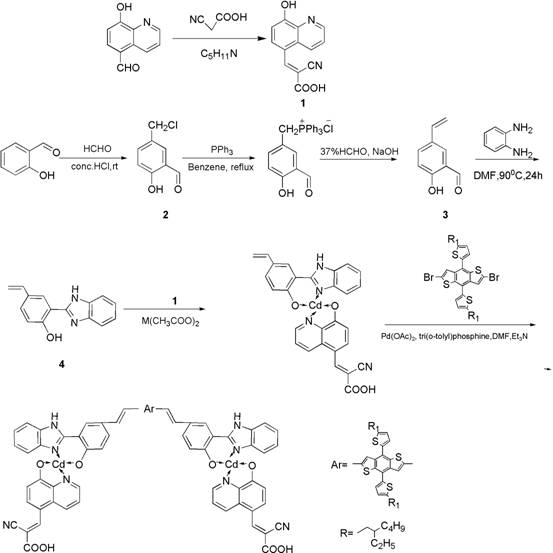
Controlling impurities during this multi-step synthesis is paramount to achieving the high purity required for electronic applications. The protocol specifies rigorous purification methods, including multiple recrystallizations and column chromatography steps, to remove unreacted starting materials and catalytic residues. For instance, the intermediate ligands are purified via recrystallization from ethanol or petroleum ether, while the final complex undergoes washing with deionized water and anhydrous methanol to eliminate inorganic salts and polar byproducts. The use of inert atmospheres during the Heck coupling prevents oxidation of the sensitive phosphine ligands and the palladium catalyst, thereby minimizing the formation of side products. This meticulous attention to purification ensures that the final BDTT-bi-Cd product meets the stringent quality standards necessary for consistent device performance, reducing the risk of charge recombination caused by impurity traps.
How to Synthesize BDTT-bi-Cd Efficiently
The preparation of this high-performance dye sensitizer involves a streamlined yet precise multi-step protocol that balances yield with purity. The process integrates classical organic synthesis techniques with modern organometallic coordination strategies to construct the complex D (-A-pi-A) 2 architecture. Detailed operational parameters, including specific solvent ratios, temperature controls, and reaction times, are critical for reproducing the reported 8.40% efficiency. For a comprehensive understanding of the standardized synthesis steps required for industrial implementation, please refer to the structured guide below.
- Synthesize the 2-cyano-3-(8-hydroxyquinolin-5-yl)acrylic acid auxiliary ligand via condensation of 5-formyl-8-hydroxyquinoline and cyanoacetic acid.
- Prepare the benzimidazole-vinyl phenol ligand and react with cadmium acetate to form the intermediate bi-Cd complex.
- Perform the final Heck coupling reaction between the bi-Cd complex and the BDTT donor derivative using a palladium catalyst to yield the target BDTT-bi-Cd dye.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this cadmium complex dye sensitizer offers distinct advantages over traditional organic dyes, particularly regarding material stability and process scalability. The enhanced thermal stability of the BDTT-bi-Cd complex reduces the risk of material degradation during storage and device fabrication, leading to more predictable inventory management and reduced waste. Furthermore, the synthetic route utilizes commercially available precursors such as salicylaldehyde and benzimidazole derivatives, which mitigates the risk of supply chain disruptions associated with exotic or proprietary starting materials. This accessibility ensures a more reliable supply chain for large-scale manufacturing operations.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for extremely expensive noble metal catalysts in the initial ligand synthesis stages, relying instead on more cost-effective reagents like cadmium acetate and palladium acetate in catalytic amounts. By optimizing the reaction conditions to achieve respectable yields without requiring excessive purification cycles, the overall production cost is significantly lowered. Additionally, the high thermal stability of the final product reduces the need for specialized cooling or handling equipment during downstream processing, contributing to substantial operational cost savings in display & optoelectronic materials manufacturing.
- Enhanced Supply Chain Reliability: The reliance on fundamental chemical building blocks such as quinoline and benzimidazole derivatives ensures that the raw material supply is robust and less susceptible to geopolitical or market volatility. The synthesis does not depend on rare earth elements or highly restricted substances, facilitating smoother customs clearance and logistics for international distribution. This stability allows procurement managers to negotiate long-term contracts with greater confidence, ensuring a continuous flow of high-purity dye sensitizer to meet production schedules without unexpected delays.
- Scalability and Environmental Compliance: The described synthesis is amenable to scale-up, as the reaction conditions (e.g., reflux in DMF or THF) are standard in industrial chemical processing and do not require extreme pressures or cryogenic temperatures. The use of standard solvents allows for established recycling and recovery protocols, aligning with modern environmental compliance standards and reducing the ecological footprint of the manufacturing process. The ability to produce the complex in multi-kilogram batches while maintaining structural integrity supports the commercial scale-up of complex cadmium complexes for widespread photovoltaic deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the BDTT-bi-Cd dye sensitizer, derived directly from the experimental data and specifications provided in the patent documentation. These insights are intended to clarify the performance metrics and synthesis feasibility for potential partners and technical evaluators. Understanding these details is crucial for assessing the viability of integrating this material into existing solar cell production lines.
Q: What is the photoelectric conversion efficiency of the BDTT-bi-Cd dye sensitizer?
A: According to patent CN112209951B, the dye-sensitized solar cell utilizing the BDTT-bi-Cd complex achieves a Photoelectric Conversion Efficiency (PCE) of 8.40%, demonstrating superior performance compared to many conventional D-pi-A structures.
Q: How does the thermal stability of this cadmium complex compare to standard organic dyes?
A: The BDTT-bi-Cd complex exhibits exceptional thermal stability with a decomposition temperature exceeding 320°C, which is critical for maintaining device integrity during long-term operation and processing in photovoltaic applications.
Q: What represents the key structural innovation in this dye sensitizer design?
A: The innovation lies in the D (-A-pi-A) 2 configuration, where a cadmium complex serves as an auxiliary electron acceptor. This design effectively lowers the energy gap between LUMO and HOMO levels, widening the light absorption range and enhancing electron transmission.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BDTT-bi-Cd Supplier
As the demand for high-efficiency photovoltaic materials continues to surge, partnering with an experienced chemical manufacturer is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of BDTT-bi-Cd meets the exacting standards required for high-performance dye-sensitized solar cells.
We invite you to collaborate with us to optimize your supply chain and reduce time-to-market for your next-generation energy solutions. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your strategic goals in the renewable energy sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
