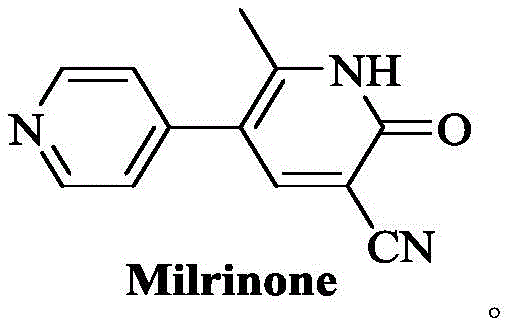
Revolutionizing Milrinone Production: A High-Yield Suzuki Coupling Strategy for Global Supply Chains
Introduction to Advanced Milrinone Synthesis
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for more efficient and safer manufacturing processes for critical drugs like Milrinone. As detailed in patent CN111718292A, a significant technological breakthrough has been achieved in the synthesis of Milrinone intermediates, addressing long-standing challenges in yield, purity, and operational safety. This innovation centers on a novel intermediate compound, designated as Formula I, which serves as a pivotal precursor in the construction of the 1,6-dihydro-2-methyl-6-oxo-[3,4'-bipyridine]-5-carbonitrile scaffold. By leveraging modern cross-coupling methodologies, this approach fundamentally shifts the paradigm from traditional, hazardous linear syntheses to a more convergent and robust strategy. The implications for global supply chains are profound, offering a pathway to reduce production costs while simultaneously enhancing the quality profile of the final Active Pharmaceutical Ingredient (API). For procurement leaders and R&D directors alike, understanding the mechanistic underpinnings and commercial viability of this route is essential for securing a competitive edge in the congestive heart failure medication market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Milrinone has been plagued by significant technical and safety hurdles that impact both cost and supply reliability. Traditional 'linear' synthetic routes, such as those described in earlier patents like US4469871A, typically initiate with 4-methylpyridine and involve multiple steps to construct the pyridone ring, often utilizing highly toxic reagents like malononitrile or potassium cyanide at critical junctures. These legacy processes frequently suffer from low overall yields, often hovering around 70%, which necessitates the processing of larger volumes of raw materials to achieve the same output, thereby inflating costs and waste generation. Furthermore, the crude products obtained from these conventional methods are notoriously difficult to purify, often exhibiting a distinct red coloration that requires extensive and costly recrystallization cycles to meet injection-grade standards. The reliance on harsh reaction conditions and toxic cyanide sources also imposes severe regulatory burdens on manufacturing facilities, requiring specialized containment systems and complex waste treatment protocols that hinder rapid scale-up and increase operational overhead.
The Novel Approach
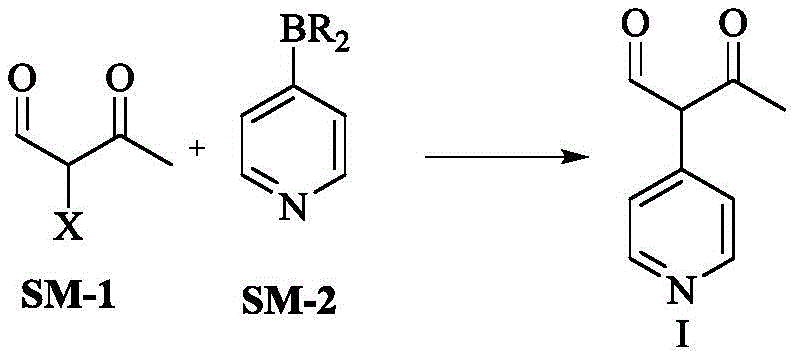
In stark contrast, the novel approach disclosed in the referenced patent introduces a streamlined convergent synthesis that dramatically simplifies the construction of the Milrinone core. This method utilizes a key intermediate, Compound I, synthesized via a palladium-catalyzed Suzuki-Miyaura coupling between an alpha-substituted acetoacetal (SM-1) and 4-pyridine boronic acid (SM-2). This strategic disconnection allows for the late-stage introduction of the cyano functionality using alpha-cyanoacetamide, effectively bypassing the need for handling free cyanide salts during the initial carbon-carbon bond formation. The result is a process that operates under milder conditions, utilizing common solvents like dimethyl sulfoxide and ethanol, which are far easier to manage on a large scale than the chlorinated solvents often required in older routes. Most critically, this new pathway consistently delivers yields exceeding 90% and produces a white crystalline product with purity levels reaching 99.9%, effectively eliminating the color issues and extensive purification steps associated with prior art. This represents a quantum leap in process efficiency, transforming a complex, hazardous operation into a manageable, high-yielding industrial process.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The cornerstone of this innovative synthesis is the efficient formation of the carbon-carbon bond between the acetoacetal fragment and the pyridine ring, achieved through a robust Suzuki-Miyaura coupling mechanism. In this catalytic cycle, a palladium(0) species, such as tetrakis(triphenylphosphine)palladium (Pd(PPh3)4), undergoes oxidative addition with the alpha-halo acetoacetal (where X is typically Br, I, or OTf). This step generates a reactive organopalladium intermediate that is primed for transmetallation. The presence of a base, preferably potassium acetate (KOAc) or potassium carbonate, facilitates the activation of the 4-pyridine boronic acid, forming a boronate species that readily transfers the pyridyl group to the palladium center. Following transmetallation, the complex undergoes reductive elimination to release the desired intermediate Compound I and regenerate the active Pd(0) catalyst, allowing the cycle to continue. The choice of solvent, particularly dimethyl sulfoxide (DMSO), plays a crucial role in stabilizing the transition states and ensuring high solubility of the inorganic bases, which is vital for maintaining reaction homogeneity and rate.
From an impurity control perspective, this mechanism offers distinct advantages over nucleophilic substitution routes used in the past. By avoiding strong nucleophiles like cyanide in the early stages, the formation of side products resulting from over-alkylation or polymerization is significantly minimized. The specificity of the palladium catalyst ensures that the coupling occurs selectively at the halogenated position of the acetoacetal, preserving the integrity of the ketone and aldehyde functionalities required for the subsequent cyclization. Furthermore, the use of boronic acids, which are generally stable and non-toxic compared to organotin or organozinc reagents used in other coupling methods, simplifies the downstream removal of metal residues. The rigorous control of reaction parameters, such as maintaining a reflux temperature and optimizing the molar ratio of base to substrate (typically around 3.4:1), ensures that the conversion is driven to completion, leaving minimal unreacted starting material that could complicate purification. This mechanistic precision is what enables the achievement of the reported >99% purity, a critical metric for regulatory compliance in API manufacturing.
How to Synthesize Milrinone Intermediate Efficiently
The practical execution of this synthesis is designed for straightforward implementation in standard chemical manufacturing facilities, requiring no exotic equipment or extreme conditions. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically argon, to protect the sensitive palladium catalyst from oxidation. Key to the success of the operation is the precise stoichiometry of the reagents, with a slight excess of the boronic acid (1.2 equivalents) and base (3.4 equivalents) relative to the alpha-bromoacetoacetal to drive the equilibrium forward. Once the coupling is complete, the workup involves a simple filtration to remove inorganic salts, followed by extraction with ethyl acetate, a solvent favored for its low toxicity and ease of recovery. The resulting organic phase is dried and concentrated to yield the intermediate Compound I as a high-purity solid, ready for the final cyclization step without the need for chromatographic purification. This operational simplicity translates directly into reduced batch times and lower labor costs, making it an attractive option for contract development and manufacturing organizations (CDMOs).
- Perform Suzuki coupling between alpha-substituted acetoacetal (SM-1) and 4-pyridine boronic acid (SM-2) using a Pd catalyst and base in DMSO.
- Isolate the intermediate compound I through extraction and concentration, achieving yields over 90%.
- React intermediate I with alpha-cyanoacetamide in ethanol under alkaline conditions to cyclize and form the final Milrinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere technical specifications. The primary value driver is the substantial reduction in manufacturing costs derived from the significant improvement in overall yield. By boosting yields from the industry standard of approximately 70% to over 90%, the consumption of raw materials per kilogram of finished product is drastically reduced, leading to direct savings on input costs. Additionally, the elimination of highly toxic reagents like potassium cyanide from the early process steps simplifies the regulatory compliance landscape, reducing the costs associated with hazardous waste disposal and specialized safety infrastructure. This simplification also mitigates supply chain risks, as the sourcing of safer, commodity-grade reagents like boronic acids and acetoacetals is more reliable and less prone to geopolitical or regulatory disruptions compared to controlled cyanide salts.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the minimization of purification steps. Traditional routes often require multiple recrystallizations to remove colored impurities, which results in significant product loss and increased solvent usage. In this new method, the product precipitates as a white crystalline solid with purity exceeding 99%, often requiring only a single wash or recrystallization to meet specification. This reduction in downstream processing not only saves on solvent recovery costs but also increases the effective capacity of existing manufacturing suites by shortening the cycle time per batch. Furthermore, the use of robust catalysts that can be sourced commercially ensures that the cost of goods sold (COGS) remains stable and predictable, shielding the supply chain from volatility associated with custom-synthesized reagents.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for pharmaceutical buyers, and this route enhances reliability by decoupling production from hazardous material constraints. The reliance on standard Suzuki coupling chemistry means that the process can be easily transferred between different manufacturing sites globally, as the requisite expertise and equipment are widely available in the fine chemical industry. The stability of the intermediate Compound I also allows for potential stockpiling, providing a buffer against demand fluctuations. By avoiding the logistical complexities of transporting and storing large quantities of cyanide, companies can streamline their vendor qualification processes and reduce the lead time for raw material procurement. This flexibility ensures that production schedules can be maintained even in the face of minor supply disruptions, guaranteeing a steady flow of API to the formulation stage.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant advancement towards green chemistry principles. The replacement of toxic cyanide sources with alpha-cyanoacetamide in the final step confines the hazard to a smaller, more manageable portion of the process, reducing the overall risk profile of the facility. The solvents employed, such as ethanol and DMSO, are well-understood and can be efficiently recycled, minimizing the environmental footprint of the manufacturing operation. The high yield and purity inherently reduce the generation of chemical waste, aligning with increasingly stringent global environmental regulations. This compliance advantage not only future-proofs the supply chain against tightening regulations but also enhances the corporate social responsibility (CSR) profile of the drug product, a factor of growing importance to healthcare providers and payers.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common questions regarding the synthesis and application of this Milrinone intermediate. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The following responses address key concerns regarding yield consistency, impurity profiles, and scalability.
Q: How does this new synthesis route improve upon traditional Milrinone manufacturing methods?
A: Traditional methods often rely on toxic reagents like potassium cyanide or malononitrile and suffer from low yields (around 70%) and poor product color. This novel route utilizes a Suzuki coupling strategy that eliminates highly toxic cyanide sources in the early stages, resulting in white crystalline products with purity exceeding 99% and yields greater than 90%.
Q: What are the safety advantages of using the alpha-substituted acetoacetal pathway?
A: The primary safety advantage is the avoidance of handling large quantities of potassium cyanide or malononitrile during the initial bond-forming steps. By deferring the introduction of the cyano group to the final cyclization step using alpha-cyanoacetamide, the process significantly reduces operator exposure risks and simplifies waste treatment protocols.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It employs mild reaction conditions, commercially available catalysts like Pd(PPh3)4, and robust solvents such as DMSO and ethanol. The high atom economy and simplified purification steps make it economically viable for commercial scale-up compared to complex multi-step linear routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milrinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthesis route requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel Suzuki coupling strategy are fully realized in a commercial setting. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Milrinone intermediate meets the highest international standards for pharmaceutical use. Our commitment to quality assurance means that we can consistently deliver the high-purity, white crystalline material described in the patent, enabling our clients to streamline their own API manufacturing processes with confidence.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this advanced synthetic route, we can help you optimize your supply chain for both cost and reliability. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive value for your organization. Let us collaborate to bring safer, more affordable cardiovascular therapies to the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
