Revolutionizing TATB Production: A Safe, One-Pot Nitration Strategy for Commercial Scale-Up
The global demand for high-performance insensitive explosives continues to drive innovation in energetic material synthesis, with 1,3,5-triamino-2,4,6-trinitrobenzene (TATB) standing out as a critical compound due to its exceptional thermal stability and insensitivity to shock. However, traditional manufacturing routes have long been plagued by safety hazards and environmental concerns, creating a pressing need for greener alternatives. Patent CN113004151B introduces a transformative safe synthesis method that fundamentally reimagines the production pathway for this vital chemical. By leveraging the unique isomeric properties of 1,3,5-cyclohexanetrione imine and employing a sophisticated multi-stage one-pot trinitration strategy, this technology offers a robust solution for cost reduction in energetic materials manufacturing. For R&D directors and supply chain leaders, this patent represents a significant leap forward, replacing hazardous chlorinated routes with a streamlined, environmentally benign process that utilizes common industrial reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of TATB has relied heavily on pathways starting from 1,3,5-trichlorobenzene (TCB) or phloroglucinol, both of which present substantial operational and economic drawbacks. The TCB route, while chemically straightforward, suffers from the inherent difficulty of completely substituting chlorine atoms during the amination step, often leaving residual chlorine-containing byproducts that compromise the purity and safety performance of the final explosive. Furthermore, the amination reactions in these traditional methods typically require extreme conditions, such as high temperature and high pressure, which escalate energy consumption and introduce significant safety risks in a production environment. The use of dimethyl sulfoxide (DMSO) as a solvent in some variations further complicates matters by increasing wastewater treatment costs due to the difficulty of degrading this solvent. These factors collectively create a bottleneck for commercial scale-up of complex energetic intermediates, limiting the ability of manufacturers to meet growing demand efficiently.
The Novel Approach
In stark contrast, the methodology disclosed in CN113004151B utilizes 1,3,5-cyclohexanetrione as a readily available and cost-effective starting material, bypassing the need for chlorinated precursors entirely. This innovative route proceeds through a sequence of trioximation, N-O bond cleavage-isomerization, and N-acylation to generate a stable intermediate, N,N',N''-triacetyl-1,3,5-triaminobenzene. The true breakthrough lies in the subsequent multi-stage one-pot trinitration reaction, which cleverly integrates dinitration, hydrolysis, and final nitration into a single streamlined operation. This approach not only eliminates the dangerous step of directly aminating explosive polynitro compounds but also simplifies the workflow by avoiding the isolation and purification of unstable intermediates. By adopting this strategy, manufacturers can achieve a reliable TATB supplier status with improved safety profiles and reduced environmental footprint.
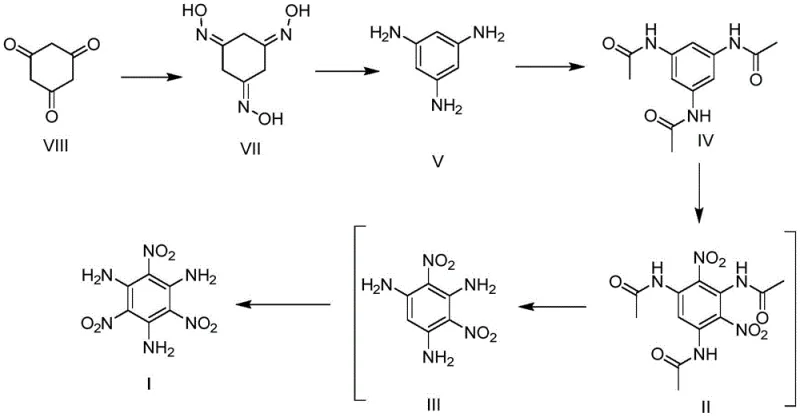
Mechanistic Insights into Multi-Stage One-Pot Trinitration
The core of this technological advancement is the meticulously designed multi-stage one-pot trinitration mechanism, which orchestrates three distinct chemical transformations within a single reactor system to maximize efficiency and safety. The process begins with a controlled first nitration of the N-acetylated intermediate, where the acetyl groups serve as protective masks, moderating the reactivity of the amino groups and allowing for the selective introduction of two nitro groups under relatively mild conditions. Following this, the system undergoes a hydrolysis stage where water is introduced directly into the acidic reaction mixture. This step is critical as it cleaves the acetyl protecting groups, regenerating the free amino groups which act as powerful electron-donating substituents. This regeneration dramatically increases the electron density on the benzene ring, thereby activating the final position for nitration without requiring the harsh conditions typical of direct nitration of deactivated rings.
Furthermore, the mechanistic design incorporates an ingenious resource utilization strategy during the second nitration phase. The addition of an acid anhydride, such as acetic anhydride, serves a dual purpose: it removes the residual water generated during hydrolysis, shifting the equilibrium towards product formation, and it reacts to generate additional acid in situ, which acts as a catalyst for the final nitration step. This self-sustaining cycle minimizes the need for external reagent additions and reduces waste generation. From an impurity control perspective, the stepwise nature of the reaction ensures that highly unstable polynitro intermediates are never isolated, significantly lowering the risk of thermal runaway or detonation. This level of process control is essential for producing high-purity TATB that meets the stringent specifications required for nuclear and defense applications, ensuring consistent performance and reliability.
How to Synthesize 1,3,5-Triamino-2,4,6-trinitrobenzene Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure optimal yield and safety, particularly during the exothermic nitration stages. The process initiates with the conversion of 1,3,5-cyclohexanetrione to its trioxime derivative, followed by catalytic hydrogenation to form 1,3,5-triaminobenzene, which is then immediately acetylated to stabilize the molecule for nitration. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and quenching procedures for each stage of the one-pot trinitration, are outlined in the guide below to assist technical teams in replicating this efficient protocol.
- Convert 1,3,5-cyclohexanetrione to 1,3,5-triaminobenzene via trioximation and catalytic hydrogenation.
- Protect the amine groups via N-acylation to form N,N',N''-triacetyl-1,3,5-triaminobenzene.
- Execute a multi-stage one-pot trinitration involving dinitration, hydrolysis, and final nitration to yield TATB.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits that extend beyond mere chemical efficiency. By shifting away from chlorinated starting materials like TCB, the process eliminates the complex and costly downstream processing required to remove chlorine residues and treat halogenated waste streams. This simplification of the waste profile not only reduces disposal costs but also mitigates regulatory compliance risks associated with hazardous effluent discharge. Moreover, the reliance on commodity chemicals such as hydroxylamine, acetic anhydride, and nitric acid ensures a stable and resilient supply chain, shielding production schedules from the volatility often seen with specialized or geographically constrained raw materials.
- Cost Reduction in Manufacturing: The elimination of expensive chlorinated precursors and the reduction in unit operations through the one-pot strategy lead to substantial cost savings. By avoiding the isolation of intermediates, manufacturers save on solvent usage, filtration equipment time, and drying energy, resulting in a leaner production cost structure. Additionally, the ability to perform hydrolysis and nitration in the same vessel without extensive workup steps drastically cuts down on labor and utility expenses, driving down the overall cost of goods sold for high-value energetic materials.
- Enhanced Supply Chain Reliability: The use of universally available industrial reagents ensures that production is not bottlenecked by the scarcity of niche starting materials. The robustness of the one-pot method also means that production cycles are shorter and more predictable, allowing for tighter inventory management and faster response times to market demand. This reliability is crucial for maintaining continuous supply lines to downstream customers in the defense and aerospace sectors, where delays are not an option.
- Scalability and Environmental Compliance: The inherent safety of avoiding explosive intermediate isolation makes this process highly scalable, allowing facilities to increase batch sizes with confidence. The green chemistry principles embedded in the method, such as the avoidance of refractory organic solvents and the minimization of waste, align perfectly with modern environmental standards. This facilitates easier permitting for capacity expansion and enhances the corporate sustainability profile, which is increasingly important for securing contracts with government and international partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this safe TATB synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of adopting this novel route for your manufacturing portfolio.
Q: Why is the one-pot trinitration method safer than conventional TATB synthesis?
A: Conventional methods often involve the direct amination of highly explosive polynitro intermediates under harsh conditions. This patent avoids that by nitrating a protected, stable intermediate first, then hydrolyzing and performing the final nitration under milder conditions, significantly reducing explosion risks.
Q: What are the cost advantages of using 1,3,5-cyclohexanetrione as a starting material?
A: Unlike 1,3,5-trichlorobenzene (TCB), which is expensive and generates hazardous chlorine waste, 1,3,5-cyclohexanetrione is a common industrial chemical. This switch eliminates the need for costly chlorine removal steps and reduces raw material procurement costs.
Q: How does this process improve supply chain reliability for energetic materials?
A: The process utilizes widely available reagents like hydroxylamine, acetic anhydride, and nitric acid, avoiding specialized or regulated precursors. The simplified one-pot nature also reduces processing time and equipment complexity, ensuring more consistent production schedules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,5-Triamino-2,4,6-trinitrobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of safety and efficiency in the production of high-performance energetic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN113004151B can be successfully translated from the laboratory to the plant floor. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of TATB meets the exacting standards required for insensitive munitions and advanced applications.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthesis technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can enhance your operational capabilities and secure your position as a leader in the energetic materials market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
