Commercializing High-Efficiency Carbazole-BODIPY Dyes for Advanced Laser Applications
Commercializing High-Efficiency Carbazole-BODIPY Dyes for Advanced Laser Applications
The development of advanced fluorescent materials has become a cornerstone of modern optoelectronics and biomedical imaging, driving a continuous search for compounds that offer superior photostability and tunable emission wavelengths. Patent CN101565554B introduces a groundbreaking class of strong-fluorescence boron dipyrromethene (BODIPY) dyes that incorporate a carbazole structure, effectively addressing the long-standing challenge of achieving efficient red-emitting laser dyes. This technology leverages a robust synthetic pathway where fluoroboron dipyrrole derivatives react with substituted carbazole aldehydes to form extended conjugated systems. The resulting molecules exhibit a remarkable red shift in their absorption spectrum by approximately 80 nanometers and an emission spectrum red shift of about 90 nanometers, all while maintaining a high fluorescence quantum yield of 0.67. For R&D directors and procurement specialists in the fine chemical sector, this represents a significant opportunity to access high-performance electronic chemical intermediates that were previously difficult to synthesize with consistent quality.
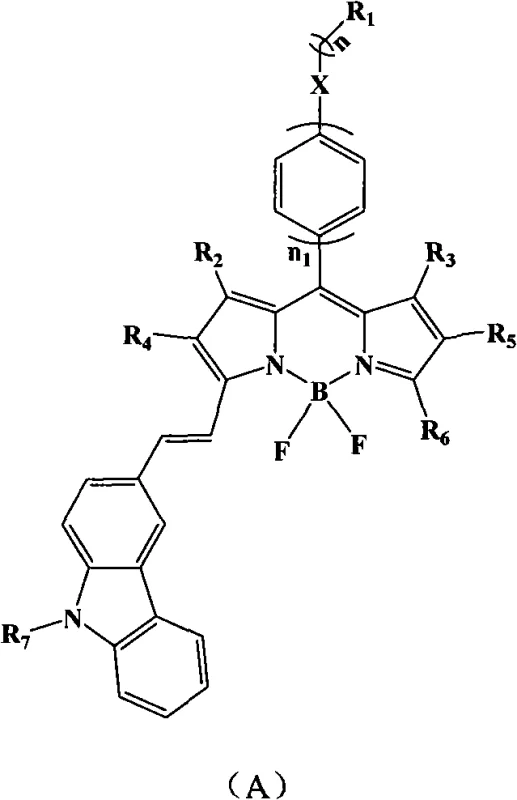
The strategic incorporation of the carbazole moiety is not merely a structural modification but a fundamental enhancement of the electronic properties of the BODIPY core. By extending the pi-conjugation through the vinyl linkage between the pyrrole ring and the carbazole unit, the energy gap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) is reduced. This reduction is critical for shifting the emission into the red region of the spectrum, which is highly desirable for deep-tissue imaging and specific laser applications where green or yellow emission is insufficient. Furthermore, the patent details a versatile general structure, designated as Formula A, which allows for extensive derivatization at multiple positions (R1 through R7), enabling fine-tuning of solubility, crystallinity, and further spectral adjustments to meet specific high-purity OLED material or laser dye requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing red-emitting BODIPY dyes have often been plagued by complex multi-step sequences that rely heavily on transition metal catalysis, such as palladium-coupled reactions, which introduce significant cost and purity challenges. In many conventional routes, achieving a substantial red shift often comes at the expense of fluorescence quantum yield, leading to materials that absorb well but emit poorly, thereby reducing lasing efficiency. Additionally, the purification of these traditional dyes frequently requires rigorous removal of heavy metal residues, adding layers of complexity to the manufacturing process and increasing the environmental footprint. The instability of some red-shifted precursors under standard reaction conditions also leads to inconsistent batch-to-batch reproducibility, a major pain point for reliable agrochemical intermediate supplier networks and pharma partners who demand strict quality control. Consequently, the commercial availability of high-efficiency red laser dyes has remained limited, hindering the broader adoption of these materials in cost-sensitive applications.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by utilizing a direct condensation reaction between a pre-formed BODIPY core and a carbazole aldehyde, driven by a simple organic base catalyst. This method eliminates the need for expensive transition metals entirely, relying instead on piperidine and molecular sieves to facilitate the Knoevenagel-type condensation. The use of 4A molecular sieves acts as an in-situ dehydrating agent, effectively shifting the reaction equilibrium towards the product side without requiring harsh azeotropic distillation setups. This streamlined process not only simplifies the reaction setup but also significantly reduces the formation of metal-based impurities, resulting in a cleaner crude product that is easier to purify. The ability to operate at temperatures between 100°C and 150°C in common organic solvents like toluene or orthodichlorobenzene makes this process highly adaptable to existing reactor infrastructure, facilitating easier commercial scale-up of complex polymer additives and dye intermediates.
Mechanistic Insights into Piperidine-Catalyzed Condensation
The core of this synthesis lies in the base-catalyzed condensation mechanism, where piperidine acts as a nucleophilic catalyst to activate the active methylene group on the BODIPY core. The mechanism initiates with the deprotonation of the methyl group adjacent to the pyrrole nitrogen by the piperidine, generating a reactive carbanion intermediate. This nucleophile then attacks the carbonyl carbon of the carbazole aldehyde, forming a beta-hydroxy intermediate. Under the elevated reaction temperatures (100-150°C) and the dehydrating influence of the molecular sieves, this intermediate undergoes elimination of water to form the stable carbon-carbon double bond characteristic of the final vinyl-linked product. The presence of the electron-rich carbazole ring stabilizes the transition state through resonance, facilitating the formation of the extended conjugated system. This mechanistic pathway is highly efficient because it avoids the high-energy barriers associated with oxidative coupling methods, allowing the reaction to proceed smoothly under inert atmospheres of argon or nitrogen.
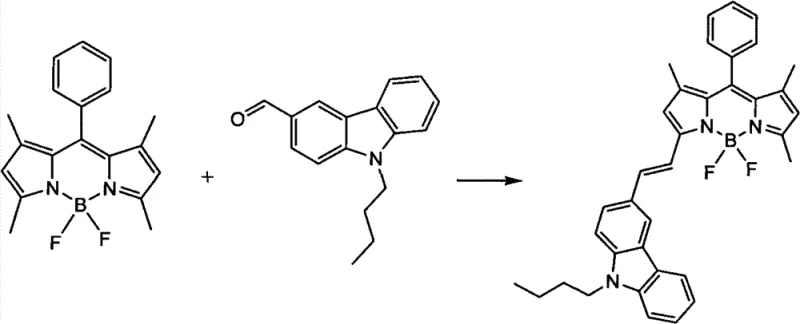
Impurity control in this system is primarily managed through the stoichiometric balance of reactants and the rigorous exclusion of moisture. The patent specifies a molar ratio of BODIPY to carbazole aldehyde ranging from 1:2 to 1:5, ensuring that the aldehyde is in excess to drive the consumption of the more valuable BODIPY core. The use of 4A molecular sieves is critical not just for yield, but for preventing the hydrolysis of the sensitive BODIPY core or the reversal of the condensation reaction. Side reactions are minimized by the selectivity of the piperidine catalyst, which preferentially activates the specific active methylene protons without attacking other sensitive functional groups on the carbazole or BODIPY rings. This high level of chemoselectivity ensures that the impurity profile is dominated by unreacted starting materials rather than complex byproducts, simplifying the downstream purification via silica gel column chromatography and ensuring the final product meets the stringent purity specifications required for high-purity electronic chemical applications.
How to Synthesize Carbazole-BODIPY Dyes Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced dyes, emphasizing the importance of precise stoichiometry and moisture control. The process begins with the selection of appropriate solvents, such as toluene, N-Methyl pyrrolidone, or chlorobenzene, which must be dry and capable of sustaining the required reaction temperature. The reactants are combined in a reactor equipped with a stirrer and an inert gas inlet, followed by the addition of the catalyst and dehydrating agent. The reaction is then heated and maintained for a period ranging from 8 to 50 hours, depending on the specific reactivity of the substituents involved. Detailed standardized synthesis steps for optimizing yield and purity are provided in the guide below, ensuring reproducibility across different production scales.
- Prepare the reaction mixture by combining the BODIPY core material and substituted carbazole aldehyde in a molar ratio of 1: 2 to 1:5 within an organic solvent such as toluene or orthodichlorobenzene.
- Add piperidine as the catalyst (0.01 to 0.20 molar equivalents) and 4A molecular sieves as a dehydrating agent to drive the equilibrium forward.
- Heat the mixture to 100-150°C under an inert argon or nitrogen atmosphere with stirring for 8 to 50 hours, followed by purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers distinct advantages by simplifying the raw material portfolio and reducing dependency on critical metals. The shift from transition metal catalysis to organic base catalysis fundamentally alters the cost structure of the manufacturing process, removing the volatility associated with palladium or platinum pricing. Furthermore, the use of commodity chemicals like piperidine and molecular sieves ensures a stable and predictable supply chain, mitigating risks related to raw material shortages. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the wide temperature and time windows provide operators with significant leeway to optimize throughput without compromising product quality. This flexibility is crucial for maintaining reducing lead time for high-purity fluorescent dyes in a fast-paced market environment.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts results in substantial cost savings, as there is no longer a need for specialized ligands or extensive metal scavenging processes during purification. The simplified workup procedure, which primarily involves filtration of the molecular sieves followed by standard chromatography, reduces solvent consumption and labor hours associated with complex extraction protocols. Additionally, the high atom economy of the condensation reaction minimizes waste generation, lowering disposal costs and aligning with green chemistry principles. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), making these high-performance dyes more accessible for mass-market applications.
- Enhanced Supply Chain Reliability: The reliance on readily available organic building blocks, such as substituted carbazoles and standard BODIPY cores, ensures a resilient supply chain that is less susceptible to geopolitical disruptions affecting rare earth or precious metal markets. The stability of the reactants allows for long-term storage and bulk purchasing strategies, further buffering against price fluctuations. Moreover, the reaction's tolerance to various solvents provides procurement teams with the flexibility to source materials locally, reducing logistics costs and carbon footprint. This reliability is essential for securing long-term contracts with downstream users in the display and laser industries who require uninterrupted material flow.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous nature and the absence of hazardous gaseous reagents, allowing for straightforward translation from laboratory glassware to industrial reactors. The use of molecular sieves as a solid dehydrating agent simplifies waste management compared to liquid desiccants or azeotropic distillation, reducing the volume of hazardous waste generated. The high selectivity of the reaction minimizes the formation of toxic byproducts, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility enhances the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole-substituted BODIPY dyes. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing product lines or research pipelines.
Q: What is the primary advantage of introducing a carbazole structure to BODIPY dyes?
A: Introducing a carbazole structure extends the pi-conjugated system, resulting in a significant red shift of both absorption (approx. 80 nm) and emission spectra (approx. 90 nm), while maintaining a high fluorescence quantum yield of around 0.67.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes piperidine as an organic base catalyst and 4A molecular sieves for dehydration, eliminating the need for costly palladium or other transition metal catalysts and simplifying downstream purification.
Q: What are the typical reaction conditions for this condensation?
A: The reaction is typically conducted in high-boiling organic solvents like toluene or NMP at temperatures between 100°C and 150°C under an inert atmosphere for 8 to 50 hours to ensure complete conversion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole-BODIPY Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this carbazole-BODIPY technology in advancing the fields of optoelectronics and bio-imaging. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of dye meets the exacting standards required for high-performance laser and display applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our advanced synthesis capabilities. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can support your next generation of fluorescent products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
