Advanced Synthesis of Metal-Free Triplet Photosensitizers for Commercial Optoelectronic Applications
The landscape of optoelectronic materials is undergoing a significant transformation driven by the demand for non-toxic, cost-effective, and high-performance photon management systems. Patent CN113461698B introduces a groundbreaking trimeric indenyl biscoumarin-corrole triplet photosensitizer, designated as Compound (I), which represents a major leap forward in metal-free organic photochemistry. This innovative molecule leverages a sophisticated star-shaped architecture where two coumarin donor molecules and one corrole acceptor molecule are strategically connected via a rigid trimeric indene bridging unit. This structural design is not merely aesthetic; it is engineered to facilitate efficient intramolecular energy transfer, a critical requirement for high-performance triplet-triplet annihilation (TTA) upconversion systems. By eliminating the reliance on scarce and toxic heavy metals such as iridium or platinum, this technology opens new avenues for sustainable manufacturing in the display and solar energy sectors.
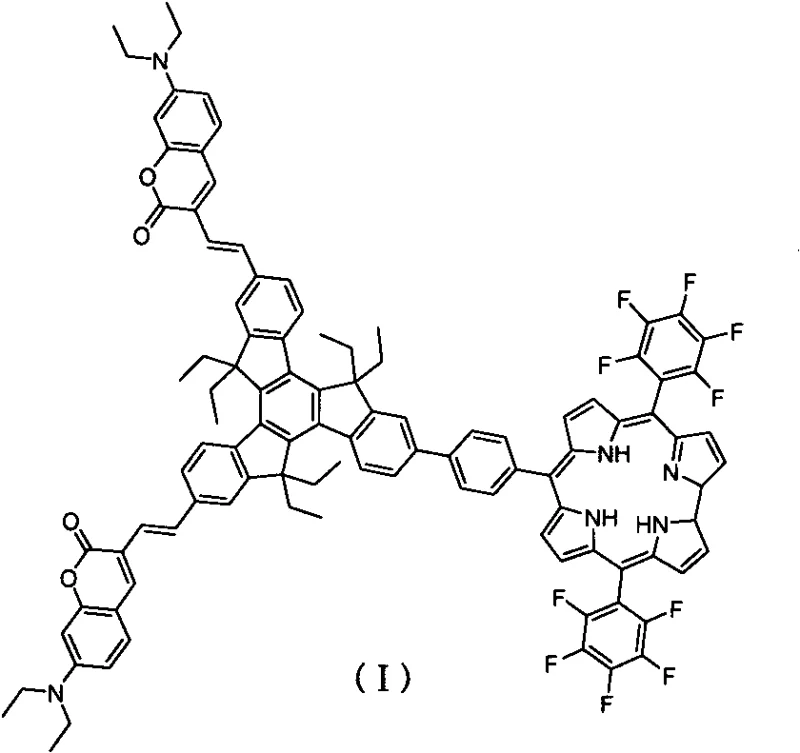
For R&D directors evaluating new material platforms, the structural integrity and synthetic accessibility of Compound (I) are paramount. The molecule features a large rigid conjugated system that ensures strong absorption in the visible region and a prolonged triplet excited state lifetime. The integration of coumarin derivatives, known for their high fluorescence quantum yields and large Stokes shifts, with corrole derivatives, which possess strong Soret and Q band absorptions, creates a synergistic effect. This binary system allows for the harvesting of low-energy photons and their conversion into higher-energy emissions, a process vital for next-generation photovoltaic devices and bio-imaging probes where deep tissue penetration is required without the phototoxicity associated with UV excitation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of efficient triplet photosensitizers has been dominated by organometallic complexes containing heavy atoms. While these materials often exhibit high intersystem crossing efficiencies due to the heavy atom effect, they suffer from severe drawbacks that limit their commercial viability. The primary concern is the inherent toxicity of heavy metals, which poses significant regulatory hurdles for biomedical applications such as photodynamic therapy and in vivo imaging. Furthermore, the reliance on precious metals like platinum, palladium, or iridium drives up the raw material costs exponentially, making large-scale deployment in consumer electronics or solar farms economically unfeasible. Additionally, the synthesis of these metal complexes often requires harsh conditions and specialized ligands, complicating the supply chain and increasing the environmental footprint through difficult waste disposal processes.
The Novel Approach
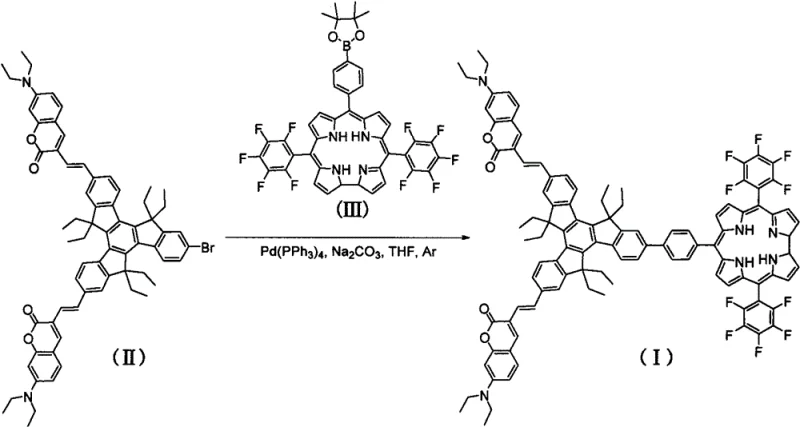
The novel approach detailed in the patent circumvents these issues by utilizing a purely organic framework that achieves efficient triplet state population without heavy atoms. The synthesis relies on a robust Suzuki coupling reaction between a brominated trimeric indene precursor and a boronate-functionalized corrole derivative. This methodology allows for the precise assembly of the complex star-shaped topology under relatively mild conditions. By using a trimeric indene core, the molecule gains exceptional rigidity, which minimizes non-radiative decay pathways and enhances the triplet lifetime. This strategy not only reduces the cost of goods sold by avoiding precious metals but also simplifies the purification process, as the removal of residual metal catalysts—a common bottleneck in organometallic synthesis—is less critical compared to removing toxic heavy metal residues from the final API or material.
Mechanistic Insights into Suzuki Coupling and Energy Transfer
The core of this synthesis lies in the palladium-catalyzed cross-coupling mechanism, specifically the Suzuki-Miyaura reaction, which forms the carbon-carbon bonds linking the functional arms to the central core. In this catalytic cycle, the tetrakis(triphenylphosphine)palladium(0) catalyst undergoes oxidative addition with the aryl bromide moiety of the trimeric indene derivative. This is followed by transmetallation with the organoboron species derived from the corrole precursor, facilitated by the base (sodium carbonate) in the biphasic solvent system of THF, methanol, and water. The final reductive elimination step releases the coupled product and regenerates the active palladium(0) species. The choice of reaction conditions, specifically maintaining a temperature between 65-75°C for 18-22 hours, is critical to ensure complete conversion while preventing the decomposition of the sensitive corrole macrocycle.
Beyond the synthesis, the photophysical mechanism of Compound (I) is equally fascinating for material scientists. Upon excitation, the coumarin units act as antenna chromophores, absorbing light efficiently and transferring the energy to the corrole center via Förster resonance energy transfer (FRET). The corrole moiety then undergoes intersystem crossing to populate the triplet state. Because the molecule is metal-free, this intersystem crossing is driven by the specific electronic configuration of the corrole ring and the spin-orbit coupling enhanced by the extended conjugation, rather than external heavy atoms. The resulting long-lived triplet state (118μs) is then available to transfer energy to an annihilator molecule, such as perylene diimide (PBI), initiating the upconversion process that emits high-energy yellow light from low-energy red excitation.
How to Synthesize Trimeric Indenyl Biscoumarin-Corrole Efficiently
The preparation of this advanced photosensitizer requires careful control of stoichiometry and atmospheric conditions to maximize yield and purity. The process begins with the dissolution of the brominated precursor and the boronate corrole in anhydrous tetrahydrofuran under an inert argon atmosphere to prevent catalyst deactivation. A mixed aqueous base solution is then introduced to facilitate the transmetallation step essential for the coupling. The reaction is heated to a precise thermal window to drive the kinetics without degrading the sensitive fluorophores. Following the reaction, a rigorous workup involving extraction and silica gel chromatography is employed to isolate the pure product from side products and catalyst residues. For a detailed breakdown of the specific reagent quantities and purification parameters, please refer to the standardized protocol below.
- Dissolve 2-bromo-7,12-dicoumarinyl trimeric indene derivatives and meso-phenylboronate corrole derivatives in tetrahydrofuran under argon protection.
- Add anhydrous sodium carbonate and a mixed solution of water and methanol, followed by the addition of tetrakis(triphenylphosphine)palladium catalyst.
- Heat the reaction mixture to 65-75°C for 18-22 hours, then perform extraction, drying, and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free photosensitizer offers substantial strategic benefits beyond mere technical performance. The elimination of precious metal catalysts from the molecular structure itself removes a major volatility factor from the cost model, as the price of the final product is no longer tethered to the fluctuating spot prices of iridium or platinum markets. Furthermore, the synthetic route utilizes commodity chemicals and standard catalytic systems that are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. This diversification of the supply base ensures greater continuity of supply, which is critical for long-term manufacturing contracts in the electronics and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the avoidance of heavy metal precursors, which are notoriously expensive and subject to stringent import/export regulations. By utilizing an all-organic scaffold constructed via Suzuki coupling, the raw material costs are drastically simplified. Additionally, the downstream processing is streamlined because there is no need for specialized metal scavenging resins or complex purification steps to reduce metal content to ppm levels, which are often required for electronic grade materials. This reduction in processing complexity translates directly into lower operational expenditures and a more favorable margin structure for high-volume production.
- Enhanced Supply Chain Reliability: The precursors for this synthesis, including coumarin derivatives and indene scaffolds, are established intermediates in the fine chemical industry with mature supply chains. Unlike custom-synthesized organometallic ligands that may have long lead times and limited vendors, these building blocks can be sourced reliably from a broad network of chemical manufacturers. This abundance of raw materials allows for rapid scaling of production capacity to meet surging demand in the optoelectronics market without the typical delays associated with sourcing exotic reagents. It also mitigates the geopolitical risks often associated with the mining and refining of rare earth elements and precious metals.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is significantly greener than traditional heavy metal-based syntheses. The absence of toxic heavy metals simplifies waste stream management and reduces the liability associated with hazardous waste disposal. The reaction conditions are moderate, avoiding extreme temperatures or pressures that would require specialized high-pressure reactors, thus allowing the process to be scaled up in standard glass-lined or stainless steel reactors commonly found in multipurpose chemical plants. This compatibility with existing infrastructure accelerates the timeline from pilot plant to commercial scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photosensitizer technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the material's capabilities and limitations for potential adopters.
Q: What are the advantages of this metal-free photosensitizer over traditional heavy metal complexes?
A: Unlike traditional sensitizers relying on expensive and toxic heavy atoms like iridium or platinum to promote intersystem crossing, this metal-free organic design utilizes a rigid conjugated structure to achieve efficient triplet state generation. This eliminates heavy metal toxicity concerns for biological applications and significantly reduces raw material costs associated with precious metal catalysts.
Q: What is the expected yield and purity profile for this synthesis route?
A: According to the patent data, the Suzuki coupling reaction yields the target compound in the range of 20% to 24% after purification. The process utilizes standard silica gel column chromatography with a dichloromethane-petroleum ether system, ensuring high purity suitable for sensitive optoelectronic and bio-imaging applications.
Q: What are the primary photophysical properties of the synthesized compound?
A: The compound exhibits a high molar extinction coefficient of 1.96×10^5 cm^-1 mol^-1 L and a long triplet lifetime of 118μs. These properties enable efficient triplet-triplet annihilation upconversion, specifically converting red light excitation at 589nm into yellow emission, which is critical for solar energy harvesting and deep-tissue imaging.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimeric Indenyl Biscoumarin-Corrole Supplier
As the global demand for advanced optoelectronic materials continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market deployment is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of triplet lifetimes and molar extinction coefficients, guaranteeing that every batch meets the high performance standards required for display and solar applications.
We invite you to engage with our technical procurement team to discuss how this metal-free photosensitizer can optimize your product portfolio. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this organic alternative. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your unique volume requirements, ensuring a supply chain that is both resilient and cost-effective.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
