Advanced Synthesis of 3,3'-Azobis Oxadiazole Derivatives for Commercial Energetic Material Applications
The landscape of high energy density materials (HEDM) is undergoing a significant transformation driven by the demand for safer, more efficient, and environmentally friendly energetic compounds. A pivotal development in this sector is documented in patent CN108689959B, which details the synthesis of 3,3'-azobis[1,2,4-oxadiazole-5-ketone-3-yl]-1,2,5-oxadiazole and its corresponding energetic ionic salts. This technology addresses critical limitations in traditional explosive formulations by introducing nitrogen-rich ionic structures that offer superior thermal stability and density. For R&D directors and procurement specialists in the advanced materials sector, understanding the nuances of this synthetic pathway is essential for integrating next-generation propellants and explosives into strategic weapon systems and industrial applications. The patent highlights a method that not only enhances performance metrics such as detonation velocity and pressure but also prioritizes safety through reduced impact sensitivity, marking a substantial leap forward in energetic material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for high energy density materials often rely on molecular analogues that suffer from inherent drawbacks such as high vapor pressure, which leads to the generation of toxic steam during processing and storage. Furthermore, many conventional explosives lack the necessary thermal stability required for modern tactical and strategic weaponry systems, posing significant safety risks during manufacturing and deployment. The absence of acidic hydrogens in standard furazan rings limits their ability to form stable ionic salts, restricting the tunability of their physical and chemical properties. Consequently, manufacturers face challenges in achieving the desired balance between high energy output and low sensitivity, often resulting in compounds that are too dangerous for widespread commercial scale-up of complex energetic intermediates. These limitations necessitate a shift towards more robust chemical architectures that can withstand rigorous environmental conditions without compromising performance.
The Novel Approach
The innovative methodology presented in the patent overcomes these hurdles by designing nitrogen-rich ionic salts that leverage the high formation enthalpy of furazan rings combined with the stability of ionic bonding. By introducing electron-withdrawing oxygen atoms into the furazan ring, the synthesis creates a conjugated structure that significantly improves thermal stability and reduces sensitivity to impact and friction. This approach allows for the formation of anions capable of reacting with various Lewis bases, thereby enabling the creation of a diverse library of energetic salts with tailored properties. The result is a class of materials that decompose primarily into nitrogen gas, offering an environmentally friendly profile while delivering detonation velocities ranging from 8013 to 8914 m/s. This novel strategy effectively bridges the gap between high performance and operational safety, providing a viable pathway for the development of advanced propellants and explosives.
Mechanistic Insights into Oxidative Coupling and Cyclization
The core of this synthesis lies in a sophisticated multi-step oxidative process that constructs the azo-bridge and oxidizes the amino groups to ketones with high precision. Initially, the precursor 4-[5-amino-1,2,4-oxadiazol-3-yl]-3-amino-1,2,5-oxadiazole undergoes oxidative coupling using potassium permanganate in an acidic acetonitrile medium at room temperature. This step is critical for forming the central azo linkage, which serves as the backbone for the high-energy structure. Following this, the intermediate is subjected to a rigorous oxidation reaction using a mixed solution of acetic anhydride and 100% nitric acid at controlled low temperatures between -20°C and 10°C. This specific condition is vital for converting the amino functionalities into the desired oxadiazolone rings without degrading the sensitive azo bond. The careful control of reaction parameters ensures high purity and yield, minimizing the formation of by-products that could compromise the energetic performance of the final material.
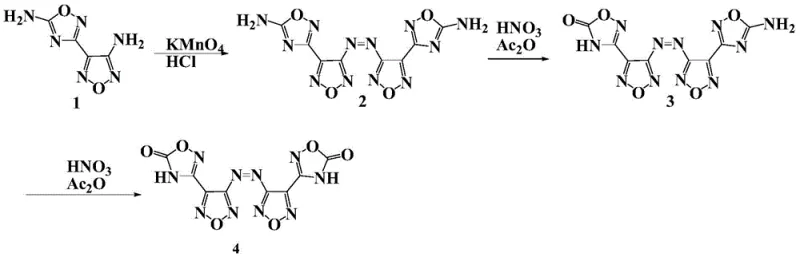
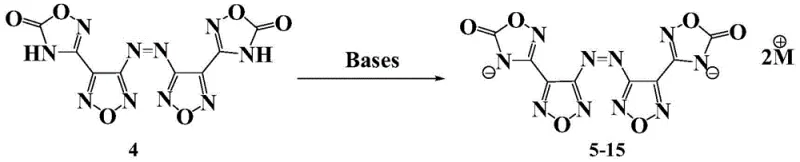
Following the formation of the core ketone structure, the process transitions into the generation of energetic ionic salts through a neutralization reaction. The ketone compound, possessing acidic protons due to the electron-withdrawing nature of the oxadiazolone rings, reacts readily with various alkaline compounds. This step is remarkably versatile, allowing for the incorporation of cations such as ammonium, guanidine, aminoguanidine, and various triazole derivatives. The reaction typically proceeds in solvents like water, methanol, or ethanol at room temperature, leading to the precipitation of the target ionic salts. This mild condition is a significant advantage for cost reduction in high energy density material manufacturing, as it eliminates the need for extreme heating or pressure. The resulting salts exhibit densities between 1.70 and 1.89 g/cm³, surpassing many traditional molecular explosives, and demonstrate decomposition temperatures mostly above 200°C, ensuring stability during storage and transport.
How to Synthesize 3,3'-Azobis Oxadiazole Derivatives Efficiently
The standardized synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value compounds with consistent quality and safety. The process begins with the preparation of the azo-bridged intermediate, followed by the critical oxidation step to form the ketone, and concludes with the salt formation. Each stage requires precise control over stoichiometry and temperature to maximize yield and purity. For instance, the oxidation step utilizes a mass ratio of 2:1 for acetic anhydride to nitric acid, a specific detail that is crucial for achieving the desired conversion without side reactions. Detailed standardized synthesis steps see the guide below for exact operational parameters and safety precautions required for laboratory and pilot-scale production.
- Oxidative Coupling: React 4-[5-amino-1,2,4-oxadiazol-3-yl]-3-amino-1,2,5-oxadiazole with potassium permanganate in acidic acetonitrile solution at room temperature to form the azo-bridge intermediate.
- Oxidation to Ketone: Treat the amino-intermediate with a mixture of acetic anhydride and 100% nitric acid at low temperatures (-20 to 10°C) to convert amino groups into oxadiazolone rings.
- Salt Formation: Neutralize the resulting ketone compound with various alkaline compounds such as ammonia or guanidine in alcoholic solvents to precipitate the final energetic ionic salts.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthesis method described in patent CN108689959B offers substantial benefits that directly address the pain points of sourcing high-performance energetic materials. The use of common and readily available reagents such as potassium permanganate, acetic anhydride, and nitric acid simplifies the procurement process and reduces dependency on exotic or controlled substances. Furthermore, the ability to use water and simple alcohols as solvents significantly lowers the environmental burden and waste disposal costs associated with production. This aligns perfectly with the industry's shift towards greener chemistry, making the material more attractive for regulatory approval and long-term sustainability goals. The robustness of the synthesis route ensures reliable supply chain continuity, as the process is less susceptible to disruptions caused by the scarcity of specialized catalysts or reagents.
- Cost Reduction in Manufacturing: The elimination of complex transition metal catalysts and the use of mild reaction conditions contribute to a significant reduction in overall production costs. By avoiding expensive purification steps typically required to remove heavy metal residues, manufacturers can streamline their operations and achieve better economic efficiency. Additionally, the high yields reported in the patent examples indicate a material-efficient process that minimizes raw material waste, further enhancing the cost-effectiveness of the synthesis. This economic advantage makes the technology highly competitive for large-scale production of energetic intermediates where margin optimization is critical.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup and the use of stable, non-hazardous solvents enhance the reliability of the supply chain. Facilities can operate with standard chemical engineering equipment without needing specialized high-pressure reactors or cryogenic systems, which reduces capital expenditure and maintenance requirements. The insensitivity of the final products (>40J impact sensitivity) also lowers the costs and complexities associated with transportation and storage, as they do not require the same level of hazardous material handling as traditional explosives. This ease of logistics ensures that delivery schedules can be met consistently, reducing lead time for high-purity energetic salts.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on batch reactions that can be easily adapted from laboratory to industrial scales. The generation of minimal three wastes, as highlighted in the patent, simplifies compliance with environmental regulations and reduces the liability associated with hazardous waste management. The decomposition of these nitrogen-rich compounds primarily into nitrogen gas further underscores their eco-friendly nature, appealing to stakeholders focused on corporate social responsibility. This combination of scalability and environmental stewardship positions the technology as a sustainable solution for the future of energetic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these energetic ionic salts. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and safety profiles. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating these materials into their existing product lines or research programs. The information below serves as a preliminary guide for technical discussions and feasibility assessments.
Q: What are the thermal stability characteristics of these energetic ionic salts?
A: According to patent CN108689959B, the synthesized energetic ionic salts exhibit excellent thermal stability with decomposition temperatures mostly exceeding 200°C, and some specific salts demonstrating decomposition temperatures above 300°C, which is critical for safe handling and storage in industrial applications.
Q: How does the sensitivity of these compounds compare to traditional explosives?
A: The impact sensitivity of the energetic ionic salts described in the patent is measured to be greater than 40J, classifying them as insensitive explosives. This low sensitivity significantly reduces the risk of accidental detonation during transport and processing compared to many conventional high-energy materials.
Q: Is the synthesis process scalable for commercial production?
A: Yes, the synthesis method utilizes common solvents like water, methanol, and acetonitrile, and avoids extremely hazardous conditions. The process is described as having a short reaction time and high yield, making it highly suitable for commercial scale-up of complex energetic intermediates without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Azobis Oxadiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating advanced patent technologies into commercially viable products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising synthesis route for 3,3'-azobis[1,2,4-oxadiazole-5-ketone-3-yl]-1,2,5-oxadiazole can be realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the high standards required for energetic applications. Our commitment to quality and safety makes us an ideal partner for organizations seeking to deploy next-generation high energy density materials in their strategic systems.
We invite you to collaborate with us to explore the full potential of this technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volumes. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning. Let us help you secure a reliable supply of high-performance energetic intermediates that drive innovation and efficiency in your operations.
