Advanced Synthesis of Triphenylamine-Vinyl Phenanthroimidazole for High-Efficiency OLEDs
Advanced Synthesis of Triphenylamine-Vinyl Phenanthroimidazole for High-Efficiency OLEDs
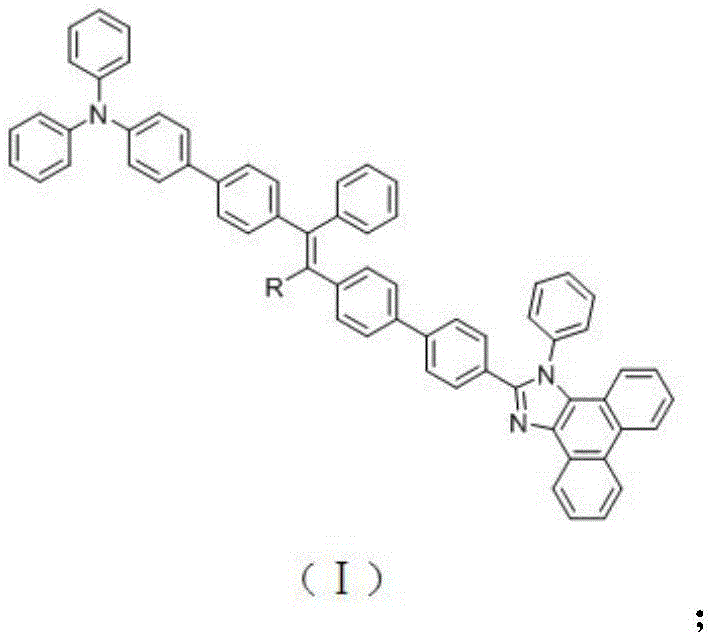
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit high luminescence efficiency but also possess balanced charge transport properties to ensure device longevity and stability. Patent CN109705041B introduces a groundbreaking class of triphenylamine substituted-vinyl modified phenanthroimidazole compounds that address the critical bottleneck of aggregation-caused quenching (ACQ) in traditional planar emitters. By strategically integrating a triphenylamine donor unit with a phenanthroimidazole acceptor through a conjugated vinyl bridge, this invention achieves a superior donor-π-acceptor architecture. This structural innovation facilitates effective π-electron delocalization, which is paramount for balancing hole and electron injection rates within the emissive layer. Consequently, these materials demonstrate exceptional fluorescence quantum yields in the solid-state thin film aggregated state, making them ideal candidates for next-generation full-color displays and solid-state lighting applications where efficiency and color purity are non-negotiable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, phenanthroimidazole-based luminescent materials have been widely explored for small-molecule organic electroluminescent devices due to their good thermal stability and electron-transporting capabilities. However, a significant drawback inherent to many of these conventional structures is their planar molecular geometry. In the aggregated state, such as within a solid-state thin film, these planar molecules tend to stack tightly via strong π-π interactions. This stacking phenomenon leads to severe fluorescence quenching, drastically reducing the external quantum efficiency of the resulting OLED device. Furthermore, while some vinyl-modified materials exhibit aggregation-induced emission (AIE) properties to counteract quenching, they often suffer from intrinsically weak electron and hole transport capabilities. This imbalance in carrier transport results in poor device performance, characterized by high driving voltages and reduced operational lifetimes, creating a persistent dilemma for material scientists attempting to兼顾 high carrier mobility with high solid-state quantum yield.
The Novel Approach
The methodology disclosed in the patent overcomes these limitations through a sophisticated molecular design that merges the best attributes of distinct functional moieties. The novel approach utilizes a triphenylamine unit as a robust electron donor, which is known for its excellent hole-transporting properties and ability to prevent efficiency roll-off caused by excessive charge transfer. This donor is linked to the phenanthroimidazole acceptor via a vinyl-containing conjugated bridge, specifically triphenylethylene or tetraphenylethylene derivatives. This specific connectivity not only disrupts the planar stacking that causes quenching but also extends the conjugation system to enhance π-electron delocalization. The result is a material that maintains high fluorescence efficiency even in the aggregated state while simultaneously ensuring balanced carrier injection and transport. This dual functionality allows for the fabrication of stable, high-efficiency organic electroluminescent devices that emit blue-green light with high color purity, representing a significant leap forward in OLED material technology.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling and Condensation
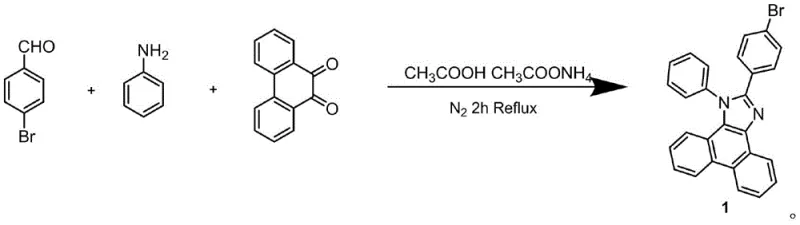
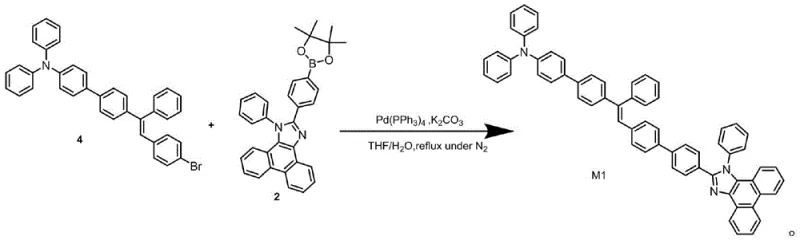
The synthesis of these advanced materials relies heavily on precise palladium-catalyzed cross-coupling reactions, specifically the Suzuki-Miyaura coupling, which serves as the cornerstone for constructing the complex carbon-carbon bonds required for the donor-acceptor architecture. The mechanism begins with the formation of the phenanthroimidazole core through a one-pot condensation reaction involving 4-bromobenzaldehyde, aniline, and 9,10-phenanthrenequinone in the presence of ammonium acetate and acetic acid. This step is critical as it establishes the electron-accepting scaffold. Subsequently, the bromine handle on this scaffold is converted into a boronic ester via a palladium-catalyzed borylation using bis(pinacolato)diboron. This transformation is essential for enabling the final coupling step, where the boronyl-phenanthroimidazole reacts with a brominated triphenylamine-vinyl derivative. The catalytic cycle involves oxidative addition of the aryl halide to the Pd(0) species, transmetallation with the organoboron compound, and reductive elimination to form the final C-C bond, regenerating the active catalyst.
Controlling impurities in this multi-step synthesis is vital for achieving the high purity required for electronic grade materials. The use of specific ligands, such as tetrakis(triphenylphosphine)palladium or Pd(dppf)Cl2, ensures high selectivity and minimizes the formation of homocoupling byproducts or dehalogenated impurities. Furthermore, the reaction conditions, including temperature control between 80°C to 120°C and the use of strong bases like potassium carbonate, are optimized to drive the reactions to completion while suppressing side reactions. The purification process typically involves column chromatography using silica gel with petroleum ether and dichloromethane gradients, which effectively separates the target high-purity product from unreacted starting materials and catalyst residues. This rigorous control over the reaction mechanism and purification protocol ensures that the final triphenylamine substituted-vinyl modified phenanthroimidazole compounds possess the structural integrity necessary for optimal device performance.
How to Synthesize Triphenylamine Phenanthroimidazole Efficiently
The preparation of these high-performance OLED materials follows a modular synthetic strategy that allows for the systematic assembly of the donor, bridge, and acceptor components. The process begins with the construction of the phenanthroimidazole core, followed by the independent synthesis of the triphenylamine-vinyl arm, and concludes with their convergence via a final cross-coupling reaction. This convergent approach offers significant advantages in terms of yield optimization and impurity management compared to linear synthesis routes. The detailed standardized synthesis steps, including specific molar ratios, solvent systems, and workup procedures required to achieve pharmaceutical or electronic grade purity, are outlined in the guide below.
- Synthesize 4-bromophenanthroimidazole via one-pot condensation of 4-bromobenzaldehyde, aniline, and 9,10-phenanthrenequinone.
- Perform Suzuki borylation on the bromo-intermediate to generate the key 4-boronyl phenanthroimidazole building block.
- Execute the final Suzuki coupling between the boronyl phenanthroimidazole and the triphenylamine-vinyl bromide derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding cost structure and supply security. The reliance on widely available commodity chemicals such as 4-bromobenzaldehyde, aniline, and benzophenone derivatives mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the robustness of the Suzuki coupling reactions used in the final steps ensures high reproducibility across different batch sizes, which is crucial for maintaining consistent quality during scale-up from laboratory to commercial production. The elimination of exotic or highly unstable intermediates simplifies the logistics of storage and handling, thereby reducing overall operational overheads and enhancing the reliability of the supply chain for downstream OLED manufacturers.
- Cost Reduction in Manufacturing: The synthetic pathway described avoids the use of expensive transition metal catalysts beyond standard palladium systems, which can be recovered and recycled, significantly lowering the cost of goods sold. Additionally, the high yields reported in the patent examples reduce the amount of waste generated per kilogram of product, leading to substantial cost savings in raw material consumption and waste disposal. The streamlined purification process further contributes to cost efficiency by minimizing solvent usage and processing time, making the commercial production of these high-purity OLED materials economically viable.
- Enhanced Supply Chain Reliability: By utilizing a convergent synthesis strategy where key intermediates like the boronyl phenanthroimidazole and the triphenylamine-vinyl bromide can be prepared independently, the manufacturing process becomes more resilient to disruptions. If one branch of the synthesis faces delays, the other can continue, allowing for better inventory management and faster response times to market demand. This modularity ensures a steady flow of materials, reducing lead times for high-purity electronic chemicals and providing a competitive edge in the fast-paced display technology sector.
- Scalability and Environmental Compliance: The reaction conditions employed, such as refluxing in THF or dioxane under nitrogen, are well-suited for large-scale reactor operations without requiring extreme pressures or temperatures. This inherent scalability facilitates the transition from gram-scale laboratory synthesis to ton-scale commercial production with minimal process re-engineering. Moreover, the use of standard organic solvents and the potential for catalyst recycling align with increasingly stringent environmental regulations, ensuring that the manufacturing process remains compliant and sustainable as production volumes increase to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triphenylamine substituted-vinyl modified phenanthroimidazole compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for integration into their supply chains or R&D pipelines.
Q: How does the vinyl modification improve OLED efficiency?
A: The vinyl group connects the donor and acceptor units, effectively improving π-electron delocalization. This balances carrier injection and transport, significantly enhancing fluorescence quantum efficiency in the solid state.
Q: What is the primary advantage of this synthesis route for mass production?
A: The process utilizes robust palladium-catalyzed Suzuki couplings and standard condensation reactions with commercially available starting materials, ensuring high scalability and consistent batch-to-batch reproducibility.
Q: Does this material exhibit Aggregation-Caused Quenching (ACQ)?
A: No, the incorporation of triphenylamine and vinyl groups induces Aggregation-Induced Emission (AIE) characteristics, preventing fluorescence quenching in the aggregated solid-state film typical of planar phenanthroimidazoles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylamine Phenanthroimidazole Supplier
As the demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage in bringing these advanced compounds to market. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of triphenylamine phenanthroimidazole meets the exacting standards required for high-performance optoelectronic applications.
We invite you to collaborate with our technical team to explore how this patented technology can enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and accelerate your time to market with next-generation display materials.
