Advanced Ionic Liquid Nitration Strategy for Commercial Scale Trinitrophloroglucinol Production
Advanced Ionic Liquid Nitration Strategy for Commercial Scale Trinitrophloroglucinol Production
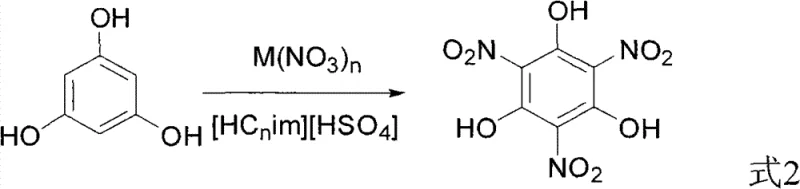
The chemical industry is constantly seeking safer and more efficient pathways for synthesizing high-value energetic material precursors, and patent CN101898968B presents a groundbreaking methodology for the production of trinitrophloroglucinol (TNPG). This specific intellectual property details a novel synthetic route that utilizes acidic ionic liquids in conjunction with metal nitrates to achieve direct nitration of phloroglucinol under remarkably mild conditions. Unlike traditional processes that rely on hazardous mixed acids, this innovation leverages the unique solvation and catalytic properties of imidazolium-based ionic liquids to facilitate the transformation at temperatures ranging from 30 to 100 degrees Celsius. For R&D directors and process engineers, this represents a significant shift towards greener chemistry, offering a robust alternative that mitigates the severe corrosion issues historically associated with nitration reactions. The technical depth of this patent suggests a viable pathway for scaling up the production of TNPG, a critical intermediate for insensitive high-energy explosives like TATB, without compromising on safety or yield efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of trinitrophloroglucinol has been plagued by significant engineering challenges and safety hazards inherent to traditional nitration chemistries. The most common legacy method involves the use of concentrated sulfuric acid and concentrated nitric acid as a mixed acid system, which acts as both the dehydrating agent and the nitrating source. While chemically effective, this approach generates substantial amounts of waste acid and water, leading to extreme corrosion of reactor vessels and necessitating the use of expensive corrosion-resistant materials like glass-lined steel or hastelloy. Furthermore, alternative protection-deprotection strategies, such as those utilizing acetic anhydride to protect hydroxyl groups prior to nitration, introduce unnecessary complexity by adding multiple synthetic steps and requiring large volumes of organic solvents for recovery. These conventional routes not only inflate the operational expenditure due to high energy consumption for acid concentration and recycling but also pose severe environmental disposal challenges regarding spent acid streams.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology disclosed in CN101898968B introduces a streamlined one-pot synthesis that fundamentally alters the reaction medium. By dissolving phloroglucinol directly into an acidic ionic liquid, specifically alkylimidazolium hydrogen sulfates, the process creates a highly organized reaction environment that stabilizes the transition states required for nitration. The introduction of solid metal nitrates, such as zirconyl nitrate or bismuth nitrate, serves as a controlled source of nitronium ions, avoiding the violent exotherms often seen with liquid nitric acid. This novel approach allows the reaction to initiate at moderate temperatures of 30 to 100 degrees Celsius and proceed to completion at room temperature, drastically reducing the thermal load on the system. The elimination of free strong mineral acids from the bulk reaction mixture means that standard stainless steel reactors can potentially be utilized, representing a massive capital expenditure saving for manufacturing facilities looking to adopt this technology for commercial scale-up of complex energetic intermediates.
Mechanistic Insights into Ionic Liquid-Mediated Nitration
The core of this technological breakthrough lies in the synergistic interaction between the acidic ionic liquid solvent and the metal nitrate salt, which together create a super-electrophilic environment without the bulk acidity of sulfuric acid. The hydrogen sulfate anion in the ionic liquid provides the necessary proton source to activate the nitrate anion from the metal salt, effectively generating the nitronium ion (NO2+) in situ within the ionic matrix. This mechanism ensures a steady and controlled supply of the electrophile, preventing the localized overheating and runaway reactions that characterize traditional batch nitrations. Moreover, the ionic liquid acts as a template that may orient the phloroglucinol substrate favorably for triple substitution, ensuring high regioselectivity for the 2,4,6-trinitro product. For research teams, understanding this mechanistic nuance is crucial for optimizing reaction parameters, as the viscosity and acidity of the ionic liquid can be tuned by altering the alkyl chain length on the imidazolium cation to further enhance mass transfer rates.
From an impurity control perspective, this mechanism offers distinct advantages over mixed acid nitration by minimizing oxidative degradation of the aromatic ring. In traditional strong acid media, the electron-rich phloroglucinol ring is susceptible to over-oxidation, leading to the formation of tarry byproducts and quinone-like impurities that are difficult to separate. The milder oxidative potential of metal nitrates in the ionic liquid medium suppresses these side reactions, resulting in a cleaner reaction profile with fewer colored impurities. This high level of chemoselectivity simplifies the downstream purification process, often allowing for direct crystallization or simple column chromatography rather than complex multi-stage recrystallizations. For quality assurance teams, this translates to a more consistent impurity profile in the final API intermediate, reducing the risk of batch failures and ensuring that the material meets the stringent specifications required for downstream conversion into insensitive munitions or pharmaceutical derivatives.
How to Synthesize Trinitrophloroglucinol Efficiently
The practical implementation of this synthesis route requires careful attention to the stoichiometry of the metal nitrate and the loading of the ionic liquid to ensure complete conversion while maintaining economic viability. The patent outlines a general procedure where phloroglucinol is first dissolved in the acidic ionic liquid, followed by the addition of the nitrating agent, typically in a molar excess ranging from 3 to 25 equivalents relative to the substrate. Detailed standardized synthetic steps see the guide below, which breaks down the precise operational parameters for temperature ramping and workup procedures to maximize yield and safety. Operators should note that while the reaction can initiate at elevated temperatures, the bulk of the conversion occurs upon cooling to room temperature, highlighting the importance of thermal management during the induction period. Adhering to these optimized conditions ensures that the unique benefits of the ionic liquid system are fully realized in a production setting.
- Dissolve phloroglucinol in an acidic ionic liquid (e.g., [Hmim][HSO4]) with a ratio of 1-50g ionic liquid per 0.50mmol reactant.
- Add metal nitrate nitrating agent (3-25 equivalents) such as zirconyl nitrate or bismuth nitrate to the mixture.
- Initiate reaction at 30-100°C, then maintain at room temperature for 0.1-24 hours before extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-based nitration technology offers profound strategic advantages that extend far beyond simple yield improvements. The most immediate impact is the drastic reduction in infrastructure costs, as the elimination of corrosive mixed acids removes the requirement for specialized glass-lined or tantalum-clad reactors, allowing facilities to utilize standard stainless steel equipment for production. This shift significantly lowers the barrier to entry for manufacturing this high-value intermediate and reduces the long-term maintenance burden associated with acid corrosion, thereby enhancing overall asset reliability and uptime. Furthermore, the use of solid metal nitrates simplifies logistics and storage compared to handling large volumes of fuming nitric acid, improving site safety profiles and reducing insurance premiums related to hazardous material storage.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the removal of expensive acid recovery units and the reduction in waste treatment costs associated with spent mixed acids. By utilizing reusable ionic liquids and generating less hazardous aqueous waste during the workup phase, the operational expenditure per kilogram of product is significantly lowered compared to traditional methods. Additionally, the simplified purification train reduces solvent consumption and energy usage for distillation, contributing to a leaner and more cost-effective manufacturing process that improves gross margins for the final energetic material intermediate.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable metal nitrate salts ensures a robust supply chain that is less vulnerable to the fluctuations and regulatory restrictions often placed on concentrated nitric and sulfuric acids. This stability allows for more predictable production scheduling and inventory management, reducing the risk of supply disruptions caused by raw material shortages or transportation hazards. The ability to store the ionic liquid catalyst for extended periods without degradation further adds to the resilience of the supply chain, enabling manufacturers to maintain buffer stocks and respond quickly to surges in demand from the defense or pharmaceutical sectors.
- Scalability and Environmental Compliance: From an environmental compliance standpoint, this green chemistry approach aligns perfectly with increasingly stringent global regulations regarding volatile organic compounds and acidic emissions. The non-volatile nature of the ionic liquid minimizes atmospheric emissions, while the reduced generation of toxic waste streams simplifies the permitting process for facility expansion or new plant construction. This environmental friendliness not only future-proofs the manufacturing asset against regulatory tightening but also enhances the corporate sustainability profile, which is becoming a critical factor in supplier selection for major multinational corporations committed to responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid nitration technology for trinitrophloroglucinol production. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application requirements. Understanding these nuances is essential for making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of using acidic ionic liquids for TNPG synthesis?
A: The use of acidic ionic liquids eliminates the need for corrosive mixed acids (sulfuric/nitric), significantly reducing equipment maintenance costs and allowing for milder reaction conditions between 30-100°C.
Q: Which metal nitrates are suitable for this nitration process?
A: The patent specifies hydrated zirconyl nitrate, vanadyl nitrate, or bismuth nitrate as effective nitrating agents, with zirconyl nitrate being preferred for its low cost and promoting effect.
Q: How does this method impact the purity of the final energetic material intermediate?
A: By avoiding harsh oxidative conditions associated with traditional mixed acid nitration, this method minimizes side reactions and tar formation, leading to a cleaner crude product that is easier to purify via column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trinitrophloroglucinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-performance energetic material intermediates like trinitrophloroglucinol. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of both pilot-scale development and full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our expertise in green chemistry and ionic liquid technologies positions us as a forward-thinking partner capable of navigating the complexities of modern chemical synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements and operational constraints. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-quality trinitrophloroglucinol that meets your exact technical needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
