Scalable Manufacturing of High-Purity 3,3',4,4'-Tetraaminobiphenyl for Advanced Dye Applications
The global demand for high-performance organic pigments and advanced polyimide precursors has necessitated a rigorous re-evaluation of supply chains for critical biphenyl derivatives. Specifically, the synthesis of 3,3',4,4'-tetraaminobiphenyl (TAB) represents a pivotal challenge in the fine chemical sector due to the complexity of introducing amine groups onto the biphenyl scaffold with high regioselectivity. A groundbreaking approach detailed in patent CN108218711B offers a transformative solution to these longstanding manufacturing bottlenecks. This proprietary methodology departs from conventional electrolytic or zinc-based reduction pathways, introducing a sophisticated multi-step sequence that leverages 1,4-naphthoquinone catalysis and copper-mediated high-pressure amination. For R&D directors and procurement strategists alike, this innovation signals a shift towards more sustainable, high-yield production capabilities that align with modern green chemistry mandates while ensuring the structural integrity required for next-generation dye intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 3,3',4,4'-tetraaminobiphenyl has relied heavily on electrochemical reduction or chemical reduction using zinc powder in alkaline media. While these methods are chemically feasible, they suffer from severe operational drawbacks that hinder efficient commercial scale-up of complex dye intermediates. The zinc powder reduction process, in particular, generates voluminous amounts of zinc oxide sludge and heavy metal-contaminated wastewater, creating a significant environmental liability and escalating waste treatment costs. Furthermore, the post-treatment purification required to remove residual metals often compromises the overall yield and introduces variability in the impurity profile, which is unacceptable for high-end electronic or optical applications. The difficulty in treating these residues often renders the process economically unviable for strict regulatory environments, forcing manufacturers to seek cleaner alternatives that do not sacrifice throughput or product quality.
The Novel Approach
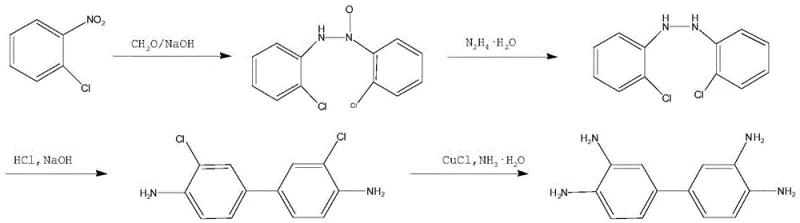
In stark contrast, the novel pathway outlined in the patent data introduces a streamlined, four-stage synthetic route that effectively circumvents the heavy metal waste issues associated with zinc reduction. By utilizing a condensation reaction followed by a hydrazine hydrate reduction catalyzed by an aluminum-nickel alloy and 1,4-naphthoquinone, the process achieves a much cleaner reaction profile. The subsequent rearrangement and copper-catalyzed amination steps are optimized for high conversion rates under controlled thermal and pressure conditions. This approach not only simplifies the workup procedures—eliminating the need for complex metal filtration steps—but also enhances the stability of the production process. The result is a robust manufacturing protocol that delivers consistent quality while significantly reducing the environmental footprint, making it an ideal candidate for reliable agrochemical intermediate and dye intermediate supplier networks seeking long-term sustainability.
Mechanistic Insights into Copper-Catalyzed Amination and Hydrazine Reduction
The core of this technological advancement lies in the precise orchestration of catalytic cycles that drive the transformation from o-chloronitrobenzene to the final tetraamine product. The initial condensation step utilizes 1,4-naphthoquinone not merely as an additive but as a critical redox mediator that facilitates the coupling of nitro compounds with aldehydes under mild alkaline conditions. This is followed by a reduction phase where hydrazine hydrate serves as the hydrogen donor, activated by the synergistic action of the aluminum-nickel alloy and the quinone catalyst. This specific catalytic combination ensures the selective reduction of the azo-linkages without over-reducing sensitive functional groups, a common pitfall in less refined protocols. The mechanistic elegance continues into the final stage, where a copper salt catalyst enables the nucleophilic substitution of chlorine atoms with amino groups under high temperature and pressure, a reaction that typically requires harsh conditions but is rendered efficient and selective in this specific solvent system.
Furthermore, the control of impurities is meticulously managed through the specific sequencing of pH adjustments and temperature gradients. During the rearrangement phase, the acidity is carefully modulated between 25% and 28% to ensure the complete migration of substituents to the 3,3' positions without inducing polymerization or degradation side reactions. The final amination step operates at elevated pressures of 4-5 MPa, which thermodynamically favors the formation of the C-N bond while suppressing the formation of dehalogenated byproducts. This rigorous control over reaction parameters ensures that the final crude product requires minimal refinement to meet stringent purity specifications, directly addressing the concerns of R&D teams regarding batch-to-batch consistency and the presence of trace metallic contaminants that could poison downstream polymerization catalysts.
How to Synthesize 3,3',4,4'-tetraaminobiphenyl Efficiently
Implementing this synthesis route requires precise adherence to the defined stoichiometric ratios and thermal profiles to maximize yield and safety. The process begins with the condensation of o-chloronitrobenzene, followed by a critical reduction step using hydrazine hydrate which must be handled with care due to its energetic nature. The subsequent acid rearrangement and high-pressure amination require specialized reactor equipment capable of withstanding corrosive acidic environments and elevated pressures. For process engineers looking to adopt this technology, understanding the interplay between the emulsifier (sodium dodecyl benzene sulfonate) and the catalyst system is vital for maintaining homogeneity during the reduction phase. The detailed standardized synthetic steps for laboratory and pilot-scale validation are provided in the guide below.
- Perform condensation of o-chloronitrobenzene with alkali and aldehyde using 1,4-naphthoquinone as a catalyst.
- Execute reduction using hydrazine hydrate with aluminum-nickel alloy and 1,4-naphthoquinone catalysts.
- Conduct acid-catalyzed rearrangement of the hydrogenation product to form the dichlorobenzidine derivative.
- Complete the synthesis via high-temperature, high-pressure amination using a copper salt catalyst and ammonia.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling strategic advantages beyond mere technical feasibility. The elimination of zinc powder as a reducing agent fundamentally alters the cost structure of production by removing the expensive and logistically complex disposal of heavy metal sludge. This shift translates directly into cost reduction in dye intermediate manufacturing, as the overhead associated with environmental compliance and waste treatment is drastically simplified. Moreover, the use of readily available raw materials such as formaldehyde, hydrazine hydrate, and ammonia ensures a stable supply chain that is less susceptible to the volatility often seen with specialized reducing agents. The robustness of the process parameters, which operate effectively within broad temperature ranges (e.g., 55-60°C for key steps), further enhances supply continuity by reducing the risk of batch failures due to minor thermal fluctuations.
- Cost Reduction in Manufacturing: The transition away from zinc-based reduction eliminates the need for costly heavy metal removal and wastewater treatment infrastructure. By utilizing a hydrazine-based system with recoverable catalysts, the operational expenditure related to effluent management is significantly lowered. Additionally, the high selectivity of the copper-catalyzed amination step reduces the loss of valuable intermediates to side reactions, thereby improving the overall mass balance and reducing the cost per kilogram of the final active pharmaceutical ingredient or dye precursor.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like ammonia and formaldehyde, rather than niche reducing metals, insulates the production schedule from raw material shortages. The process is designed to be scalable, with reaction conditions that are compatible with standard industrial stainless steel reactors, ensuring that production capacity can be ramped up quickly to meet market demand without requiring exotic equipment. This reliability is crucial for maintaining just-in-time delivery schedules for downstream customers in the textile and electronics sectors.
- Scalability and Environmental Compliance: The process generates significantly less solid waste compared to traditional methods, aligning with increasingly strict global environmental regulations. The simplified post-treatment workflow, which involves straightforward crystallization and filtration, allows for faster batch turnover times. This efficiency not only supports commercial scale-up of complex polymer additives and dyes but also future-proofs the manufacturing site against tightening environmental legislation, securing long-term operational licenses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production lines or for qualifying new suppliers who utilize this advanced methodology.
Q: What are the primary environmental advantages of this new synthesis route compared to traditional zinc powder reduction?
A: Traditional methods utilizing zinc powder reduction generate significant amounts of solid residue and zinc-containing wastewater, which are difficult and costly to treat. The patented method replaces this with a hydrazine hydrate reduction system catalyzed by aluminum-nickel alloys and 1,4-naphthoquinone, drastically reducing solid waste generation and simplifying post-treatment purification, thereby lowering environmental compliance costs.
Q: What purity levels can be achieved using this copper-catalyzed amination process?
A: The process is designed to yield high-quality 3,3',4,4'-tetraaminobiphenyl suitable for sensitive dye applications. Experimental data from the patent indicates that the final product consistently achieves HPLC purity levels exceeding 99%, with specific examples demonstrating purity up to 99.7%, ensuring minimal impurity profiles for downstream polymerization or dye synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the methodology is explicitly optimized for mass production. It utilizes robust reaction conditions, such as controlled temperatures between 55-60°C for initial steps and manageable high-pressure conditions (4-5 MPa) for the final amination. The use of common solvents like water and ethanol, along with stable catalysts, ensures the process is scalable and operationally stable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3',4,4'-tetraaminobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to cleaner, more efficient synthetic routes is critical for maintaining competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art HPLC and NMR analysis to verify the absence of critical impurities. Our capability to handle high-pressure amination reactions safely and efficiently positions us as a preferred partner for clients demanding high-quality biphenyl derivatives for advanced applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
