Advanced Synthesis of Dibutyryl Cyclophosphate Adenosine for Scalable Pharmaceutical Manufacturing
The pharmaceutical landscape continuously demands more efficient synthetic routes for critical bioactive intermediates, particularly those involved in cellular metabolism regulation. Patent CN112321660B introduces a significant advancement in the preparation of dibutyryl cyclophosphate adenosine compounds and their metal salts, which are vital derivatives of the second messenger cyclic adenosine monophosphate (cAMP). These compounds play a pivotal role as G protein-coupled receptor drugs, offering therapeutic potential for conditions ranging from angina pectoris to myocardial infarction by protecting dying cells and improving renal hemodynamics. The disclosed technology addresses the limitations of existing single-method preparations by providing a robust, scalable acylation strategy that operates effectively in aprotic solvents. By leveraging organic bases and optimizing solvent-to-substrate ratios, this innovation promises to enhance the commercial viability of producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis methods for calcium dibutyryl cyclophosphate have historically been burdened by multi-step inefficiencies that hinder large-scale production capabilities. The conventional pathway typically necessitates an initial step of forming a triethylamine salt, followed by a separate acylation reaction with acylating reagents, and finally, the formation of the corresponding calcium salt. This fragmented approach not only increases the operational complexity but also extends the overall production timeline significantly. The requirement to isolate and handle intermediate quaternary ammonium salts introduces additional unit operations such as filtration, drying, and transfer between vessels, each representing a potential point for yield loss and contamination. Furthermore, the cumulative solvent usage across these distinct stages often results in higher waste generation and increased environmental compliance costs, making the traditional process less attractive for modern green chemistry standards in API intermediate manufacturing.
The Novel Approach
In stark contrast, the novel methodology described in the patent streamlines the synthesis by enabling direct acylation of adenosine cyclophosphate in the presence of an organic base within an aprotic solvent system. This approach effectively omits the cumbersome preparation of a separate quaternary ammonium salt, thereby collapsing multiple process steps into a more cohesive workflow. The reaction conditions are meticulously optimized, utilizing a volume-to-mass ratio of aprotic solvent to adenosine cyclophosphate ranging from 2 to 40 ml/g, which ensures adequate solubility while minimizing excess solvent waste. By integrating the acylation and salt formation logic more efficiently, the process achieves a liquid phase content higher than 99%, demonstrating superior impurity control. This reduction in procedural complexity directly translates to a shortened production period, offering a compelling value proposition for manufacturers seeking cost reduction in pharmaceutical intermediate manufacturing without compromising on product quality or regulatory compliance.
Mechanistic Insights into Organic Base-Catalyzed Acylation
The core of this synthetic breakthrough lies in the precise interaction between the hydroxyl groups of the adenosine cyclophosphate backbone and the n-butyric anhydride, facilitated by a carefully selected organic base. In this mechanism, bases such as diisopropylethylamine, triethylamine, or 4-dimethylaminopyridine act as proton scavengers, activating the nucleophilic oxygen atoms on the ribose ring to attack the carbonyl carbon of the anhydride. The reaction proceeds optimally at temperatures between 50°C and 80°C, providing sufficient thermal energy to overcome the activation barrier while maintaining the stability of the sensitive cyclic phosphate structure. The molar ratio of n-butyric anhydride to the substrate is maintained between 7:1 and 10:1, ensuring a substantial excess of the acylating agent to drive the equilibrium towards the fully substituted dibutyryl product. This stoichiometric excess is critical for suppressing the formation of mono-acylated byproducts, which are common impurities in partial acylation scenarios.
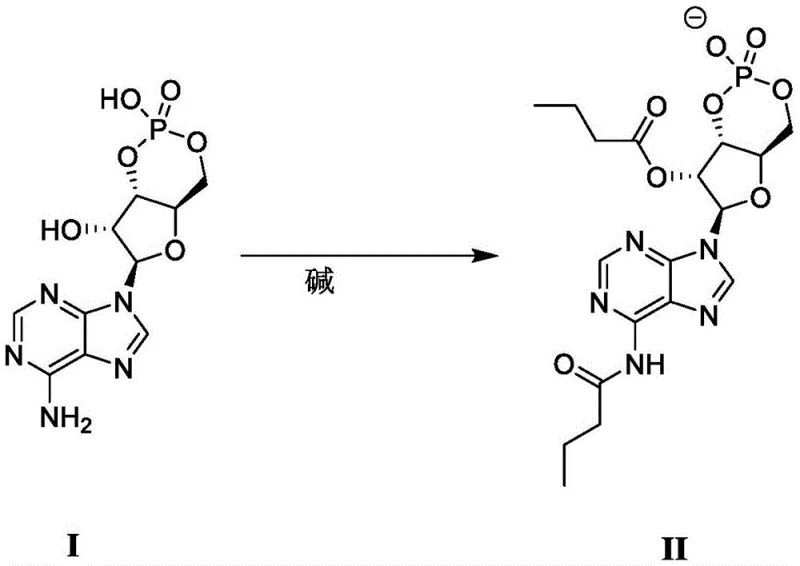
Furthermore, the choice of solvent plays a non-innocent role in stabilizing the transition states and the resulting ionic species. Aprotic solvents like N,N-dimethylformamide (DMF) or pyridine not only dissolve the polar starting material effectively but also stabilize the developing charges during the acylation event. The resulting product exists as an anion (Formula II) paired with a cation derived from the organic base or a proton, which can subsequently be exchanged for a metal ion like calcium in a downstream step. This mechanistic clarity allows for rigorous impurity control; by monitoring the reaction progress via HPLC or TLC over a period of 2 to 42 hours, operators can ensure complete conversion before initiating the workup. The subsequent extraction protocol, involving washes with mixed organic solvents like ethyl acetate and petroleum ether followed by dichloromethane and methanol mixtures, is designed to selectively remove unreacted anhydride and base residues, ensuring the final isolate meets stringent purity specifications required for clinical applications.
How to Synthesize Dibutyryl Cyclophosphate Adenosine Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to maximize yield and minimize side reactions. The process begins with the dissolution of the starting material in the chosen aprotic solvent under an inert atmosphere, typically nitrogen, to prevent moisture-induced hydrolysis of the anhydride. Following the addition of reagents and the thermal reaction phase, the workup involves a careful quenching with water and solvent removal, followed by a multi-stage extraction process to isolate the crude dibutyryl compound. The detailed standardized synthesis steps below outline the precise operational sequence validated by the patent examples, serving as a foundational guide for process chemists aiming to replicate this high-efficiency pathway in a pilot or production setting.
- Dissolve adenosine cyclophosphate (Formula I) in an aprotic solvent such as DMF or pyridine with a volume-to-mass ratio of 2-40 ml/g.
- Add n-butyric anhydride and an organic base (e.g., DIPEA or triethylamine) under nitrogen protection and heat to 50-80°C for acylation.
- Perform workup by adding water, removing solvent, extracting with organic solvents, and reacting the resulting compound with a metal salt to form the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this streamlined synthesis route offers tangible benefits that extend beyond mere technical feasibility. By eliminating the discrete step of quaternary ammonium salt preparation, the process inherently reduces the consumption of auxiliary reagents and the associated handling costs. This simplification of the manufacturing workflow leads to a significant reduction in the overall production cycle time, allowing facilities to increase throughput without expanding physical infrastructure. For supply chain managers, the ability to produce high-purity intermediates with fewer unit operations translates to enhanced reliability and reduced risk of batch failures. The robustness of the reaction conditions, which tolerate a range of aprotic solvents and organic bases, provides flexibility in sourcing raw materials, thereby mitigating supply chain disruptions caused by vendor-specific shortages.
- Cost Reduction in Manufacturing: The elimination of the separate quaternary ammonium salt preparation step represents a direct saving in both labor and material costs. Traditional methods require additional vessels, filtration equipment, and drying cycles for the intermediate salt, all of which consume energy and time. By consolidating the process, the novel method drastically simplifies the operational footprint, leading to substantial cost savings in utility consumption and equipment depreciation. Furthermore, the optimized solvent usage ratio minimizes the volume of waste solvent that requires treatment or disposal, contributing to lower environmental compliance costs and a leaner cost of goods sold (COGS) structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The versatility in solvent selection, allowing for the use of common industrial solvents like DMF, pyridine, or THF, ensures that production is not bottlenecked by the availability of exotic reagents. This flexibility empowers procurement teams to negotiate better terms with multiple suppliers, fostering a resilient supply network. Additionally, the high purity of the crude product (>99% liquid phase content) reduces the burden on downstream purification processes, which are often the most time-consuming and yield-limiting stages in pharmaceutical manufacturing. This efficiency ensures a more predictable delivery schedule for downstream API manufacturers, securing the continuity of supply for critical cardiovascular medications.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial production, with reaction conditions that are readily transferable from laboratory to commercial scale. The use of nitrogen protection and controlled heating profiles aligns with standard safety protocols for large-scale reactors, minimizing the risk of thermal runaways. Moreover, the reduced solvent load and the avoidance of heavy metal catalysts or complex separation technologies simplify the waste management profile. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the technology an attractive option for environmentally conscious pharmaceutical companies seeking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: What is the primary advantage of the method in Patent CN112321660B?
A: The primary advantage is the omission of the separate preparation of a quaternary ammonium salt, which significantly shortens the production cycle and simplifies the process compared to traditional methods.
Q: What purity levels can be achieved with this synthesis route?
A: The liquid phase content of the final product is reported to be higher than 99%, indicating a high-purity profile suitable for pharmaceutical applications without extensive additional purification.
Q: Which solvents are compatible with this acylation reaction?
A: The process utilizes conventional aprotic solvents including tetrahydrofuran, acetonitrile, 1,4-dioxane, pyridine, and N,N-dimethylformamide, with DMF and pyridine being particularly preferred.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibutyryl Cyclophosphate Adenosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the development of life-saving cardiovascular therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced acylation technologies similar to those described in recent patents, we can offer customized manufacturing solutions that optimize yield and minimize impurity profiles for complex nucleotide derivatives.
We invite you to collaborate with us to explore how this streamlined synthesis can enhance your supply chain efficiency. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering expertise can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
