Advanced Synthesis of Dipyridamole Intermediate 2,4,6,8-Tetrahydroxypyrimido[5,4-d]pyrimidine
Advanced Synthesis of Dipyridamole Intermediate 2,4,6,8-Tetrahydroxypyrimido[5,4-d]pyrimidine
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the production of critical cardiovascular drug intermediates. A pivotal advancement in this domain is detailed in patent CN101899046B, which discloses a novel method for synthesizing 2,4,6,8-tetrahydroxypyrimido[5,4-d]pyrimidine, a key precursor to the antiplatelet agent Dipyridamole (Persantine). This technology represents a significant departure from classical high-temperature condensation methods, introducing a milder, solution-phase approach that leverages sodium cyanate chemistry and catalytic hydrogenation. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, this patent offers a compelling blueprint for enhancing process efficiency. By shifting away from harsh thermal conditions and eliminating tedious isolation steps, the methodology not only improves chemical yield but also fundamentally alters the economic landscape of manufacturing this complex heterocyclic scaffold.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this pyrimido-pyrimidine core has relied heavily on variations of the Wöhler synthesis or high-temperature urea fusion techniques, which present formidable engineering challenges. As illustrated in the classical pathway, the condensation of amino orotic acid with urea typically requires heating the reaction mixture to extreme temperatures, often reaching 190°C to 210°C, to facilitate the melting of urea and drive the cyclization forward. 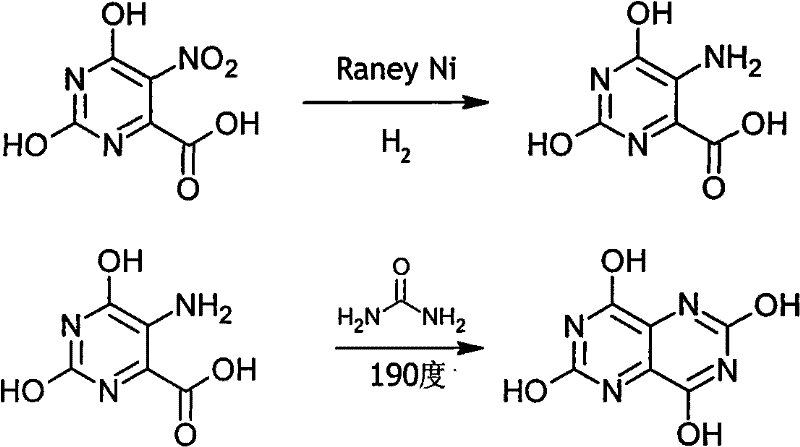 These severe thermal conditions impose rigorous demands on reactor materials, necessitating specialized alloys capable of withstanding prolonged exposure to molten urea and high heat, thereby accelerating equipment depreciation and increasing capital expenditure. Furthermore, the viscosity of the reaction mass at such temperatures creates significant mass transfer limitations, making effective stirring difficult and leading to potential hot spots that can degrade product quality. The legacy processes also mandate the isolation of the amino orotic acid intermediate via filtration and drying, a unit operation that is not only labor-intensive but also introduces opportunities for product loss and contamination, ultimately extending the total production cycle to approximately 32 hours.
These severe thermal conditions impose rigorous demands on reactor materials, necessitating specialized alloys capable of withstanding prolonged exposure to molten urea and high heat, thereby accelerating equipment depreciation and increasing capital expenditure. Furthermore, the viscosity of the reaction mass at such temperatures creates significant mass transfer limitations, making effective stirring difficult and leading to potential hot spots that can degrade product quality. The legacy processes also mandate the isolation of the amino orotic acid intermediate via filtration and drying, a unit operation that is not only labor-intensive but also introduces opportunities for product loss and contamination, ultimately extending the total production cycle to approximately 32 hours.
The Novel Approach
In stark contrast, the innovative route disclosed in the patent utilizes a telescoped, solution-phase strategy that dramatically lowers the thermal burden and simplifies the operational workflow.  Instead of relying on solid-state urea fusion, this method generates the necessary urea functionality in situ through the reaction of amino orotic acid with sodium cyanate under weakly acidic conditions. This clever chemical maneuver allows the subsequent cyclization to proceed in an aqueous alkaline environment at a maximum temperature of merely 95°C. This reduction in thermal severity is transformative for cost reduction in API manufacturing, as it permits the use of standard glass-lined or stainless steel reactors without the need for exotic high-temperature ratings. Moreover, the process is designed to be telescoped; the amino orotic acid produced in the initial hydrogenation step is not isolated but is directly utilized in the subsequent carbamylation reaction. This elimination of the intermediate work-up step saves a massive amount of time and manual labor, compressing the total reaction time to roughly 16 hours and significantly boosting overall plant throughput.
Instead of relying on solid-state urea fusion, this method generates the necessary urea functionality in situ through the reaction of amino orotic acid with sodium cyanate under weakly acidic conditions. This clever chemical maneuver allows the subsequent cyclization to proceed in an aqueous alkaline environment at a maximum temperature of merely 95°C. This reduction in thermal severity is transformative for cost reduction in API manufacturing, as it permits the use of standard glass-lined or stainless steel reactors without the need for exotic high-temperature ratings. Moreover, the process is designed to be telescoped; the amino orotic acid produced in the initial hydrogenation step is not isolated but is directly utilized in the subsequent carbamylation reaction. This elimination of the intermediate work-up step saves a massive amount of time and manual labor, compressing the total reaction time to roughly 16 hours and significantly boosting overall plant throughput.
Mechanistic Insights into Raney Ni-Catalyzed Hydrogenation and Cyclization
The success of this synthetic strategy hinges on the precise control of two distinct chemical transformations: the catalytic reduction of the nitro group and the subsequent base-mediated ring closure. The process initiates with the hydrogenation of nitroorotic acid in an alkaline medium, utilizing Raney Nickel as a highly active heterogeneous catalyst. Operating at moderate pressures (0.8 MPa) and temperatures (80°C), the Raney Nickel facilitates the selective reduction of the nitro group to an amine without affecting the sensitive carboxylic acid or pyrimidine ring functionalities. The resulting amino orotic acid exists as a soluble salt in the alkaline filtrate, which is crucial for the next stage. Upon acidification to pH 3, the addition of sodium cyanate generates isocyanic acid in low concentrations within the solution. This reactive species immediately attacks the nucleophilic amine of the amino orotic acid to form a urea derivative intermediate. This in situ generation of the carbamoylating agent avoids the handling of hazardous isocyanates and ensures a homogeneous reaction environment, which is far superior to the heterogeneous solid-liquid mixing required in traditional urea melts.
Following the formation of the urea derivative, the reaction mixture is subjected to strong alkaline conditions by adjusting the pH to 13 with sodium hydroxide. Under these basic conditions, the urea moiety undergoes an intramolecular nucleophilic attack on the adjacent carbonyl carbon, driving the cyclization to form the fused pyrimido[5,4-d]pyrimidine ring system. The mild heating to 95°C provides sufficient activation energy for this ring closure while minimizing the risk of thermal decomposition or polymerization side reactions that plague higher-temperature methods. Finally, acidification to pH 3 with sulfuric acid precipitates the product as the free acid, which is easily filtered and washed. This mechanism inherently controls impurities; by avoiding the harsh thermal stress of 190°C, the formation of tarry by-products and degradation impurities is significantly suppressed, leading to a crude product purity exceeding 90% and simplifying downstream purification requirements for high-purity pharmaceutical intermediates.
How to Synthesize 2,4,6,8-Tetrahydroxypyrimido[5,4-d]pyrimidine Efficiently
Implementing this patented methodology requires careful attention to pH control and temperature profiling to maximize the benefits of the telescoped design. The process begins with the dissolution of nitroorotic acid in sodium hydroxide solution, followed by hydrogenation over Raney Nickel. Crucially, the catalyst is removed by filtration, but the filtrate containing the amino species is retained for immediate use. The subsequent addition of sodium cyanate must be performed under controlled acidic conditions to regulate the release of isocyanic acid, preventing rapid decomposition. The final cyclization step relies on a sharp pH swing to alkaline conditions followed by heating. For a comprehensive understanding of the specific stoichiometric ratios, agitation speeds, and safety protocols required for industrial execution, please refer to the standardized synthesis guide below.
- Hydrogenate nitroorotic acid in alkaline solution using Raney Nickel catalyst at 80°C and 0.8MPa to form amino orotic acid solution without isolation.
- Adjust pH to 3 with hydrochloric acid, add sodium cyanate at 70°C to generate the urea derivative in situ.
- Raise pH to 13 with sodium hydroxide and heat to 95°C for cyclization, then acidify to pH 3 to precipitate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the classical Wöhler-type synthesis to this novel sodium cyanate route offers profound strategic advantages beyond mere chemical elegance. The primary value driver is the drastic simplification of the unit operations involved. By eliminating the isolation and drying of the amino orotic acid intermediate, the process removes a bottleneck that traditionally consumes significant labor hours and facility capacity. This streamlining directly translates to a more agile manufacturing schedule, allowing for faster turnover of reactor vessels and a reduction in the overall lead time for high-purity pharmaceutical intermediates. Furthermore, the shift from a high-temperature melt process to a moderate-temperature aqueous process fundamentally changes the equipment profile required. Standard reactors can be employed instead of specialized high-heat vessels, reducing both capital investment and maintenance costs associated with equipment degradation.
- Cost Reduction in Manufacturing: The economic implications of this process are substantial, driven primarily by energy efficiency and labor optimization. Operating at 95°C instead of 190°C results in a dramatic decrease in steam or thermal oil consumption, directly lowering utility costs per kilogram of product. Additionally, the telescoped nature of the synthesis means that solvents and water do not need to be evaporated to dryness between steps, saving significant energy on distillation and drying operations. The avoidance of complex filtration steps for the intermediate also reduces the consumption of filter aids and the labor costs associated with plate-and-frame filter press operations. Qualitatively, these factors combine to create a leaner cost structure, enabling competitive pricing for the final API intermediate without compromising on quality margins.
- Enhanced Supply Chain Reliability: From a supply continuity perspective, the robustness of this method mitigates several common production risks. The use of readily available raw materials like sodium cyanate and standard hydrogenation catalysts ensures that the supply chain is not dependent on niche or volatile reagents. The milder reaction conditions reduce the likelihood of batch failures due to equipment malfunction or thermal runaway, ensuring consistent output. Moreover, the simplified workflow shortens the production cycle from roughly 32 hours to 16 hours, effectively doubling the theoretical capacity of existing infrastructure. This increased throughput capability allows suppliers to respond more rapidly to fluctuating market demands, providing a buffer against supply shocks and ensuring steady availability for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this solution-phase method scales linearly with fewer engineering hurdles than melt condensations. Heat transfer is far more efficient in a liquid slurry than in a viscous melt, ensuring uniform temperature distribution even in large-scale reactors, which is critical for maintaining product consistency. Environmentally, the process generates less waste; the elimination of intermediate isolation reduces solvent usage and wastewater load. The aqueous nature of the reaction medium simplifies effluent treatment compared to organic solvent-heavy processes. These attributes make the technology highly attractive for commercial scale-up of complex pharmaceutical intermediates in regions with stringent environmental regulations, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for large-scale production. The answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making.
Q: How does the new sodium cyanate method improve upon the traditional Wöhler synthesis?
A: The new method eliminates the need for high-temperature urea melting (190°C), operating instead at a mild 95°C in aqueous solution. This significantly reduces energy consumption, equipment stress, and reaction time while improving yield from roughly 62% to over 70%.
Q: Is intermediate isolation required in this patented process?
A: No, a key advantage of this process is the 'telescoped' nature of the first step. The amino orotic acid generated via hydrogenation is used directly in the next reaction without filtration or drying, saving substantial labor and time.
Q: What are the purity specifications achievable with this route?
A: The optimized process yields product with a content greater than 90.0%, with moisture and ignition residue tightly controlled below 1.0% and 0.3% respectively, meeting stringent pharmaceutical intermediate standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,6,8-Tetrahydroxypyrimido[5,4-d]pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101899046B are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,4,6,8-tetrahydroxypyrimido[5,4-d]pyrimidine meets the exacting standards required for cardiovascular drug synthesis. Our commitment to quality assurance means that we can consistently deliver material with >90% content and minimal impurities, supporting your regulatory filings and clinical trials with reliable data.
We invite global pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing protocols, we can help you achieve significant reductions in COGS while securing a stable supply of critical intermediates. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to engineer a supply chain that is not only cost-effective but also resilient and compliant with the highest international standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
