Revolutionizing N-Nitroazole Production via Electrocatalytic Radical Coupling for Industrial Scale
Introduction to Advanced Electrocatalytic N-Nitration Technology
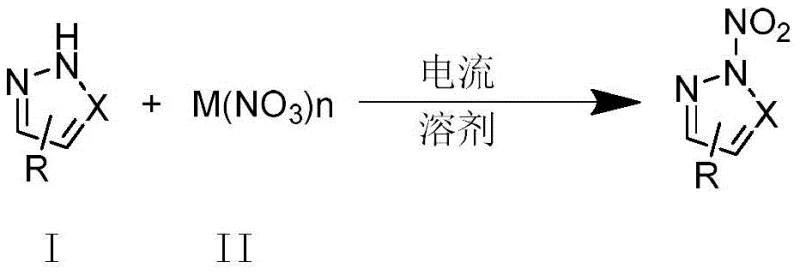
The landscape of organic synthesis is undergoing a significant transformation driven by the urgent need for greener, more sustainable manufacturing processes, particularly in the production of high-value heterocyclic intermediates. Patent CN111235597B introduces a groundbreaking methodology for the synthesis of N-nitroazole energetic compounds, shifting away from traditional, hazardous nitration protocols towards a sophisticated electrocatalytic free radical coupling reaction. This innovation leverages economical and non-toxic metal nitrates as the nitro source, facilitating the direct functionalization of the N-H bond in azoles through a one-step electrochemical process. By operating under mild, acid-free conditions, this technology not only enhances safety profiles but also significantly broadens the substrate scope, allowing for the efficient construction of N-N single bonds in complex molecular architectures that were previously difficult to access. For global procurement and R&D teams, this represents a pivotal opportunity to optimize supply chains for critical energetic material intermediates and pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formation of N-NO2 bonds via N-nitration has relied heavily on the use of aggressive nitrating agents, typically involving mixtures of concentrated nitric acid and sulfuric acid to generate the reactive nitronium ion (NO2+). While effective for simple substrates, this conventional Ingold-Hughes polar addition mechanism imposes severe limitations on synthetic flexibility, particularly when dealing with complex molecules containing acid-sensitive functional groups. The harsh acidic environment often leads to decomposition, polymerization, or unwanted side reactions, drastically reducing overall yields and complicating downstream purification processes. Furthermore, the generation of substantial quantities of acidic waste streams poses significant environmental compliance challenges and increases the operational costs associated with waste treatment and neutralization, making traditional nitration increasingly unsustainable for modern large-scale manufacturing facilities seeking to reduce their carbon footprint.
The Novel Approach
In stark contrast to these legacy methods, the novel electrocatalytic approach detailed in the patent utilizes a mild, acid-free system that harnesses the power of electricity to drive the reaction forward. By employing inexpensive metal nitrates such as iron(III) nitrate or bismuth(III) nitrate as the nitrogen source, the process avoids the corrosive hazards associated with mineral acids entirely. The reaction operates in a simple undivided cell using acetonitrile as a solvent, with a graphite anode and a platinum cathode, maintaining temperatures between 50°C and 80°C. This shift to an electrochemical paradigm not only simplifies the reaction setup but also enables a radical-based mechanism that is highly tolerant of diverse functional groups, including halogens and electron-withdrawing substituents, thereby offering a robust and versatile platform for the synthesis of a wide array of N-nitroazole derivatives with high selectivity and efficiency.
Mechanistic Insights into Electrocatalytic Biradical Coupling
The core scientific breakthrough of this technology lies in its unique mechanistic pathway, which diverges from classical ionic nitration to a radical-mediated process driven by electrochemical oxidation. As illustrated in the mechanistic proposal, the azole substrate undergoes a single-electron transfer (SET) oxidation at the anode surface, generating a reactive nitrogen-centered radical species. Simultaneously, the metal nitrate salt, upon heating in the reaction medium, serves as a thermal source for the generation of nitro radicals (NO2·). This dual-generation of radical species creates a highly reactive environment where the nitrogen radical of the azole and the nitro radical can undergo a rapid cross-coupling event to form the desired N-N single bond. The protons released during the oxidation of the azole are subsequently reduced at the cathode to evolve hydrogen gas, completing the electrical circuit and ensuring that the overall process remains redox-neutral without the need for external oxidants or reductants beyond the electrical current.

This mechanistic understanding is critical for R&D directors aiming to optimize impurity profiles and maximize yield. Because the reaction proceeds through discrete radical intermediates rather than highly electrophilic nitronium ions, the risk of over-nitration or electrophilic aromatic substitution on the carbon skeleton of the azole ring is significantly minimized. This inherent chemoselectivity ensures that the nitro group is installed exclusively at the nitrogen position, preserving the integrity of other sensitive moieties on the molecule. Furthermore, the ability to fine-tune the reaction kinetics by adjusting the applied current density (ranging from 8mA to 20mA) or voltage provides an additional layer of process control, allowing manufacturers to balance reaction rate against selectivity to achieve the highest possible purity specifications required for downstream applications in energetic materials or fine chemical synthesis.
How to Synthesize N-Nitroazoles Efficiently
The practical implementation of this electrochemical synthesis route is designed for simplicity and reproducibility, making it highly attractive for both laboratory-scale optimization and pilot plant operations. The standard protocol involves dissolving the azole substrate and the metal nitrate salt in acetonitrile, adding a supporting electrolyte such as tetrabutylammonium tetrafluoroborate, and subjecting the mixture to constant current electrolysis under a nitrogen atmosphere. The detailed standardized synthesis steps below outline the precise molar ratios, temperature controls, and workup procedures necessary to achieve optimal results, ensuring that technical teams can replicate the high yields reported in the patent data with minimal deviation.
- Mix the azole substrate with a cheap metal nitrate (such as Fe(NO3)3 or Bi(NO3)3) and a supporting electrolyte like n-Bu4NBF4 in acetonitrile solvent within an undivided cell.
- Apply a constant current (8-20mA) or voltage (2-4V) using a graphite anode and platinum cathode while heating the mixture to 50-80°C under nitrogen protection.
- After 6-12 hours of reaction, filter the mixture, extract with ethyl acetate, dry over sodium sulfate, and purify via chromatography to obtain the N-nitroazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrocatalytic nitration technology offers profound advantages for procurement managers and supply chain heads focused on cost reduction and operational resilience. The elimination of concentrated mineral acids removes the need for specialized acid-resistant reactors and extensive corrosion monitoring systems, leading to significant capital expenditure savings and reduced maintenance downtime. Additionally, the use of commodity-grade metal nitrates as reagents replaces expensive and hazardous specialized nitrating agents, driving down the raw material cost per kilogram of the final product. The simplified workup procedure, which involves basic filtration and extraction rather than complex neutralization and phase separation steps associated with acidic waste, further streamlines the manufacturing workflow, reducing labor costs and accelerating batch cycle times to improve overall throughput capacity.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly and dangerous reagents with abundant, low-cost metal salts and electricity. By removing the requirement for strong acids, the process eliminates the downstream costs associated with acid waste neutralization and disposal, which can be a substantial portion of the operating budget in traditional nitration facilities. Furthermore, the high atom economy of the radical coupling mechanism minimizes the formation of byproducts, reducing the load on purification units and increasing the overall mass efficiency of the production line, which translates directly into improved profit margins for high-volume manufacturing campaigns.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the reliance on widely available starting materials such as iron nitrate and bismuth nitrate, which are not subject to the same strict regulatory controls and supply volatility as mixed acid nitrating agents. The robustness of the reaction conditions, which tolerate a wide range of temperatures and currents, ensures consistent production output even with minor fluctuations in utility supplies. This reliability is crucial for maintaining continuous supply to downstream customers in the energetic materials and pharmaceutical sectors, where interruptions in the availability of key intermediates can have cascading effects on global production schedules and contract fulfillment obligations.
- Scalability and Environmental Compliance: The scalability of this electrochemical method is evidenced by successful gram-scale demonstrations that maintain high yields, indicating a clear path to multi-kilogram and ton-scale production without fundamental changes to the reaction chemistry. From an environmental standpoint, the acid-free nature of the process aligns perfectly with increasingly stringent global regulations regarding industrial effluent and hazardous waste management. By generating hydrogen gas as the only stoichiometric byproduct alongside the desired organic product, the technology offers a cleaner alternative that reduces the facility's environmental liability and supports corporate sustainability goals related to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical N-nitration technology, providing clarity on its operational parameters and strategic value for industrial partners. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, ensuring that decision-makers have access to accurate and actionable information for evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the advantages of using metal nitrates over traditional nitric acid for N-nitration?
A: Using metal nitrates in an electrocatalytic system eliminates the need for strong mineral acids like concentrated nitric or sulfuric acid. This avoids the generation of large amounts of acidic waste, reduces equipment corrosion risks, and allows for the nitration of substrates containing acid-labile functional groups that would otherwise decompose under traditional harsh conditions.
Q: Is this electrochemical nitration method scalable for industrial production?
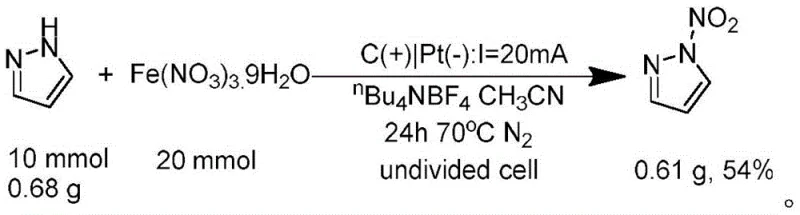
A: Yes, the patent data demonstrates successful gram-scale synthesis with consistent yields (e.g., 54% yield for N-nitropyrazole on a 10 mmol scale). The use of simple undivided cells, inexpensive electrode materials like graphite and platinum, and commercially available metal nitrates suggests high feasibility for scaling up to kilogram or ton-level production without complex reactor engineering.
Q: What is the proposed reaction mechanism for this N-N bond formation?
A: The reaction proceeds via a biradical coupling mechanism. The azole substrate undergoes anodic oxidation to form a nitrogen-centered radical, while the metal nitrate thermally generates a nitro radical (NO2·). These two radicals then couple to form the N-N single bond, with hydrogen protons being reduced at the cathode to evolve hydrogen gas, ensuring a redox-neutral and atom-economical process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Nitroazoles Supplier
As the demand for high-performance energetic materials and specialized heterocyclic intermediates continues to grow, partnering with a technically proficient CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative for securing a stable and cost-effective supply. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of N-nitroazoles meets the exacting standards required for sensitive applications in defense, aerospace, and pharmaceutical research.
We invite you to engage with our technical procurement team to discuss how this innovative electrochemical synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits and process efficiencies available through our manufacturing capabilities. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering superior value and reliability as your long-term partner in fine chemical synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
