Revolutionizing Aniline Dye Intermediate Production via Conductivity-Based Separation and Continuous Washing
The chemical manufacturing landscape for fine intermediates is undergoing a significant transformation, driven by the urgent need for sustainable processes that do not compromise on yield or purity. Patent CN109608354B introduces a groundbreaking refining method for aniline dye intermediates that fundamentally rethinks the post-reaction workup. Unlike conventional approaches that rely heavily on energy-intensive crystallization and filtration, this innovation leverages a sophisticated combination of thermal layering and conductivity-based separation. By shifting from a solid-liquid separation paradigm to a liquid-liquid extraction model, the technology addresses critical pain points regarding wastewater generation and product loss. For R&D directors and process engineers, this represents a pivotal shift towards continuous, automated processing that enhances both operational efficiency and environmental compliance. The method specifically targets the production of N,N-dialkylated aniline derivatives, which are vital precursors in the dye and fine chemical industries, ensuring that the final product meets stringent quality specifications while minimizing the ecological footprint of the manufacturing facility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional refining processes for aniline dye intermediates have long been plagued by inherent inefficiencies associated with solid-state handling. Historically, the standard protocol involves diluting the reaction mixture with large volumes of water, followed by cooling to induce crystallization, and finally separating the solid product via suction filtration. This multi-step approach is not only labor-intensive but also technically flawed; the crystallization step often results in the formation of fine crystals that are difficult to trap, leading to unavoidable product loss in the mother liquor. Furthermore, the filtration process creates significant challenges for waste gas management, as the exposure of the material to the atmosphere during filtering makes the centralized absorption of volatile organic compounds extremely difficult. The sheer volume of wastewater generated during the dilution and washing of the filter cake imposes a heavy burden on downstream treatment facilities, requiring substantial capital investment in recycling infrastructure. These cumulative inefficiencies result in higher production costs, lower overall yields, and a process that is increasingly untenable under modern environmental regulations.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a liquid-liquid separation strategy that bypasses the need for crystallization entirely. The core of this innovation lies in the recognition that the target aniline dye intermediate exists as an oil that is immiscible with water under specific temperature and pH conditions. By maintaining the reaction mixture at elevated temperatures (80-110°C) in a layering kettle, the system allows the organic phase to separate naturally from the aqueous mother liquor. This physical separation is further refined through the precise monitoring of conductivity, enabling an automated switch to isolate the organic layer with high precision. The elimination of the dilution and crystallization steps not only simplifies the workflow but also drastically reduces the volume of water required. Consequently, the process achieves a much higher recovery rate of the valuable intermediate, as the product is not lost to solubility limits in cold mother liquor or mechanical trapping in filter cakes. This streamlined methodology offers a robust pathway for industrial scale-up, providing a reliable dye intermediate supplier with the capability to deliver consistent quality.
Mechanistic Insights into Conductivity-Based Phase Separation
The success of this refining method hinges on a deep understanding of the physicochemical properties of the reaction mixture, particularly the interplay between density, temperature, and ionic conductivity. During the synthesis phase, the pH is meticulously controlled between 3 and 7 using ammonia water as an acid-binding agent. This specific range is critical because a pH that is too low retards the alkylation reaction rate, while a pH that is too high promotes the hydrolysis of the acetyl protecting group, leading to deacetylated byproducts that complicate purification. Once the reaction is complete, the pH is adjusted to a neutral range of 6-8 to optimize the subsequent layering. The mixture is then heated to 80-110°C, a temperature window chosen to reduce the viscosity of the organic phase and prevent premature crystallization, which would hinder separation. To further enhance the density difference between the aqueous and organic phases, additives such as ammonium chloride or additional ammonia water are introduced. This density modulation ensures a sharp interface between the layers, facilitating a clean split that is essential for high-purity outcomes.
Following the thermal layering, the separation mechanism employs online conductivity monitoring to distinguish between the organic product and the saline mother liquor. Since the organic phase has low conductivity and the mother liquor has high conductivity due to dissolved salts, a critical conductivity threshold (typically 5000-8000 μS/cm) is set to trigger the automatic switching of valves. This automation removes human error from the separation process, ensuring that the cut point between phases is consistent across batches. The separated organic crude is then subjected to a counter-current washing process in a dedicated washing tower, where hot water is introduced from the bottom to rinse out residual impurities without dissolving the product. This washing step is also monitored by conductivity, allowing for the recycling of wash water once it reaches a certain purity level. The integration of these control loops creates a closed-loop system that maximizes resource utilization and minimizes the generation of hazardous waste streams.
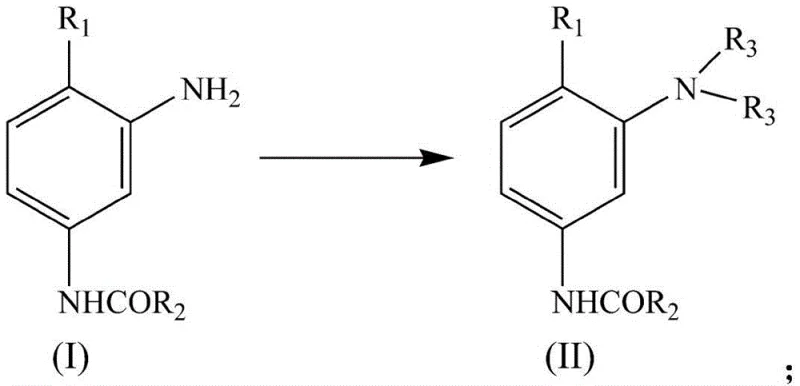
How to Synthesize Aniline Dye Intermediates Efficiently
The implementation of this advanced refining protocol requires precise adherence to the reaction parameters and separation logic outlined in the patent documentation. Operators must begin by pulping the raw aniline derivative, such as m-acetamidoaniline, in water before introducing the alkylating agent under controlled thermal conditions. The subsequent steps involve transferring the crude mixture to the layering vessel, adjusting the ionic strength to facilitate phase splitting, and executing the conductivity-guided separation. This sequence ensures that the intermediate is isolated in its oily form, ready for the final washing stage which polishes the product to commercial grade purity. For a detailed breakdown of the specific operating conditions, reagent ratios, and equipment settings required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Synthesize the crude mixture by pulping m-acetamidoaniline or 3-amino-4-methoxyacetanilide in water, heating, and adding alkylating agents while strictly controlling pH between 3 and 7 using ammonia water.
- Transfer the mixture to a layering kettle, heat to 80-110°C, and adjust density using salts or ammonia to facilitate phase separation based on conductivity differences.
- Separate the organic phase automatically via conductivity monitoring, then wash the crude product in a counter-current washing tower to remove residual mother liquor and salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this refining technology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The most immediate impact is observed in the drastic reduction of operational expenditures associated with waste management and utility consumption. By eliminating the dilution and crystallization steps, the process significantly lowers the demand for process water and the energy required for heating and cooling large volumes of solvent. This efficiency gain directly contributes to cost reduction in dye intermediate manufacturing, allowing for more competitive pricing structures without sacrificing margin. Furthermore, the simplified equipment footprint—removing the need for large crystallizers and filter presses—reduces capital expenditure for new production lines and lowers maintenance costs for existing facilities. The ability to recycle salts and water through MVR crystallization of the mother liquor further insulates the supply chain from fluctuations in raw material costs and wastewater disposal fees.
- Cost Reduction in Manufacturing: The elimination of filtration and drying steps removes some of the most energy-intensive and labor-heavy unit operations from the production line. Without the need to pump large slurry volumes through filter presses or operate vacuum dryers for extended periods, the overall energy consumption per kilogram of product is substantially decreased. Additionally, the reduction in wastewater volume means that the facility incurs lower fees for effluent treatment and discharge, which is a major cost driver in the chemical industry. The recovery of salts from the mother liquor also allows for the reuse of acid-binding agents, further driving down the variable cost of goods sold and enhancing the overall economic viability of the production campaign.
- Enhanced Supply Chain Reliability: The transition to a liquid-liquid separation process inherently improves the robustness of the supply chain by reducing the number of potential failure points. Traditional filtration processes are prone to bottlenecks, such as filter cloth blinding or pump cavitation, which can cause unplanned downtime and delay shipments. In contrast, the continuous nature of the layering and washing tower system allows for smoother throughput and more predictable batch cycle times. This reliability ensures that delivery schedules are met consistently, reducing lead time for high-purity dye intermediates and strengthening the partnership between the manufacturer and their downstream clients who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: From a regulatory perspective, this process offers a clear pathway to sustainable growth. The closed-system design minimizes the release of volatile organic compounds (VOCs) into the workplace and the environment, addressing a key concern for environmental health and safety officers. The significant reduction in wastewater discharge simplifies compliance with increasingly strict environmental regulations, mitigating the risk of fines or production shutdowns. Moreover, the process is highly scalable; the principles of conductivity-based separation and thermal layering translate effectively from pilot scale to commercial production, enabling the rapid expansion of capacity to meet surging market demand without the need for complex re-engineering of the core process logic.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this refining technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios. The answers highlight the specific mechanisms that drive the superior performance of this novel approach compared to legacy methods.
Q: How does the conductivity-based separation method improve yield compared to traditional crystallization?
A: Traditional crystallization often traps product in the mother liquor or loses fine crystals during filtration. By utilizing conductivity differences to separate the organic phase directly at elevated temperatures (80-110°C), this method avoids solid-state losses entirely, recovering the oily intermediate more efficiently and improving overall yield significantly.
Q: What role does pH control play in preventing byproduct formation during alkylation?
A: Maintaining the reaction pH between 3 and 7 is critical. If the pH is too low, the reaction rate slows down; if too high, the acetyl protecting group hydrolyzes, creating impurities. Precise control using ammonia water ensures the acetyl group remains intact while allowing efficient alkylation, resulting in a cleaner crude product.
Q: How does this refining process impact wastewater treatment costs?
A: The process eliminates the massive water volumes required for dilution and crystallization washing in traditional methods. Furthermore, the mother liquor and concentrated wash water are treated via MVR crystallization to recover salts and distilled water for recycling, drastically reducing the net discharge volume and associated treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aniline Dye Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced refining technologies requires a partner with deep technical expertise and a proven track record of execution. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN109608354B are fully realized in practice. Our state-of-the-art facilities are equipped with the necessary automation and containment systems to handle conductivity-based separations and closed-loop washing processes safely and efficiently. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aniline dye intermediate meets the exacting standards required by the global dye and fine chemical markets.
We invite you to collaborate with us to leverage these process innovations for your specific supply chain needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this refining method for your projects. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
