Scalable Synthesis of 4-Amino-3-Hydrazino-5-Methyl-1,2,4-Triazole Energetic Salts for Defense Applications
Introduction to Next-Generation Energetic Materials
The development of high-energy density materials (HEDMs) that balance superior performance with enhanced safety remains a paramount challenge in the field of energetic chemistry. Patent CN108148007B introduces a groundbreaking methodology for synthesizing a series of 4-amino-3-hydrazino-5-methyl-1,2,4-triazole energetic ionic salts, addressing critical limitations in current propellant and explosive formulations. These novel compounds leverage a unique cationic backbone derived from triaminoguanidine, functionalized to maximize nitrogen content and positive enthalpy of formation while maintaining structural integrity under thermal stress. The patent highlights a versatile synthetic platform capable of generating a wide array of salts by varying the counter-anion, including nitrates, perchlorates, and complex tetrazole derivatives. This technological advancement offers a reliable energetic material supplier pathway for industries seeking to replace traditional hazardous explosives like RDX and HMX with greener, more stable alternatives that do not compromise on detonation velocity or pressure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for high-nitrogen energetic compounds often rely on aggressive nitration processes using mixed acids, which pose significant safety risks due to exothermic runaway potential and the generation of toxic nitrogen oxide byproducts. Furthermore, conventional methods frequently struggle with poor atom economy and difficult purification steps, leading to lower overall yields and higher production costs associated with waste treatment. The instability of intermediate species in acidic media can also result in unpredictable decomposition, complicating scale-up efforts for commercial manufacturing. Many existing protocols require harsh reaction conditions, such as extreme temperatures or pressures, which demand specialized equipment and rigorous safety protocols, thereby increasing the capital expenditure for production facilities. Additionally, the environmental footprint of these legacy processes is substantial, often involving chlorinated solvents and generating large volumes of acidic wastewater that require extensive neutralization before disposal.
The Novel Approach
In stark contrast, the methodology described in CN108148007B utilizes a mild silver salt metathesis strategy that fundamentally shifts the safety and efficiency profile of energetic salt production. By reacting the pre-formed 4-amino-3-hydrazino-5-methyl-1,2,4-triazole hydrochloride with various silver salts in solvents like water or ethanol, the process drives the reaction forward through the precipitation of insoluble silver chloride. This approach allows for precise control over reaction conditions, typically operating between 0°C and 100°C, which drastically reduces energy consumption and thermal hazards. As illustrated in the synthesis of the nitrate derivative, the reaction proceeds rapidly, often completing within 0.5 hours at 40°C, demonstrating exceptional kinetic efficiency.
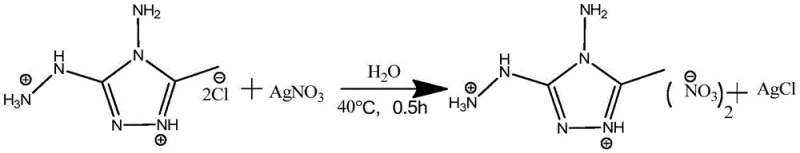
The versatility of this method is evident in its ability to accommodate a diverse range of anions without altering the core reaction parameters, enabling the rapid screening of new energetic formulations. The use of benign solvents and the elimination of strong oxidizers during the salt formation step significantly lowers the environmental impact, aligning with modern green chemistry principles. This novel approach not only simplifies the downstream processing by facilitating easy filtration of the silver chloride byproduct but also ensures high purity of the final ionic salt through straightforward recrystallization techniques.
Mechanistic Insights into Silver Salt Metathesis and Ionic Assembly
The core chemical transformation relies on the thermodynamic driving force provided by the formation of the silver chloride lattice, which effectively removes chloride ions from the solution and shifts the equilibrium towards the desired energetic salt. The cationic component, 4-amino-3-hydrazino-5-methyl-1,2,4-triazole, possesses a highly conjugated heterocyclic system that stabilizes the positive charge through resonance delocalization across the nitrogen-rich ring structure. This structural feature is critical for achieving the high thermal stability observed in the final products, with decomposition temperatures frequently exceeding 150°C. The presence of multiple amino and hydrazino substituents further enhances the hydrogen bonding network within the crystal lattice, contributing to increased density and mechanical stability.
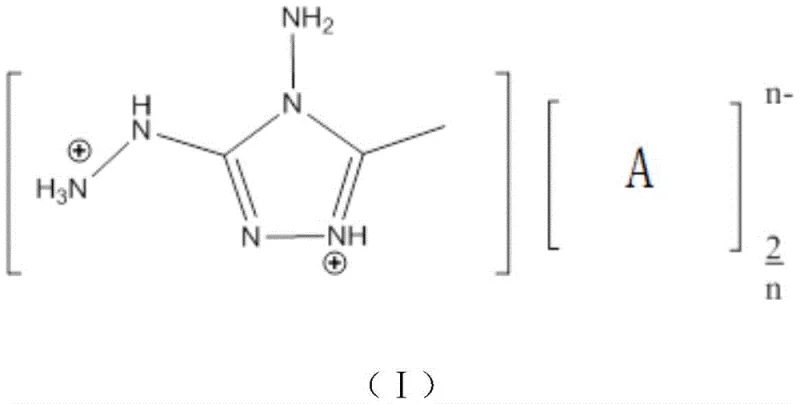
From an impurity control perspective, the metathesis mechanism is inherently self-purifying; since the silver salt reactants are typically high-purity reagents and the only byproduct is solid AgCl, the liquid phase contains predominantly the target ionic species. Any unreacted starting material can be easily separated due to solubility differences in the chosen recrystallization solvent, usually methanol or ethanol. The selection of the anion [A]n- plays a pivotal role in tuning the oxygen balance and detonation performance, allowing chemists to optimize the formulation for specific applications ranging from gas generators to low-signature propellants. The ability to incorporate oxygen-rich anions like perchlorate or nitrate improves the oxygen balance, ensuring more complete combustion and reduced smoke signature, which is a critical requirement for tactical missile systems.
How to Synthesize 4-Amino-3-Hydrazino-5-Methyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these high-value intermediates, beginning with the cyclization of triaminoguanidine hydrochloride in acetic acid followed by the critical metathesis step. This standardized approach ensures reproducibility and safety, making it ideal for transfer from laboratory bench to pilot plant operations. The detailed标准化 synthesis steps see the guide below for specific molar ratios and temperature profiles that guarantee optimal yield and purity.
- Condense triaminoguanidine hydrochloride with acetic acid under reflux, then react with hydrochloric acid to form the triazole hydrochloride intermediate.
- Disperse the hydrochloride intermediate in a solvent such as water or ethanol and add the corresponding silver salt (e.g., silver nitrate, silver perchlorate).
- Stir the mixture at mild temperatures (0°C to 100°C) to precipitate silver chloride, then filter and recrystallize the product to obtain the pure energetic salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology offers transformative benefits in terms of cost structure and logistical reliability. The process eliminates the need for expensive and hazardous nitration infrastructure, replacing it with standard glass-lined or stainless steel reactors capable of handling mild aqueous or alcoholic solutions. This reduction in specialized equipment requirements translates directly into lower capital expenditure and faster deployment of new production lines. Furthermore, the high yields reported, such as 88% for the nitrate salt and 85% for the perchlorate variant, indicate a highly efficient use of raw materials, minimizing waste and maximizing output per batch.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of inexpensive solvents like water and ethanol significantly lower the variable costs associated with production. By avoiding complex purification chromatography and relying on simple filtration and recrystallization, the operational expenses are drastically simplified, leading to substantial cost savings over the product lifecycle. The high atom economy of the metathesis reaction ensures that the majority of the input mass is converted into valuable product rather than waste streams requiring costly disposal.
- Enhanced Supply Chain Reliability: The starting materials, including triaminoguanidine hydrochloride and common silver salts, are commercially available from multiple global suppliers, reducing the risk of single-source bottlenecks. The mild reaction conditions mean that production is less susceptible to interruptions caused by utility failures or cooling system limitations, ensuring consistent delivery schedules. Additionally, the stability of the intermediate hydrochloride salt allows for stockpiling, providing a buffer against raw material fluctuations and enabling just-in-time manufacturing strategies.
- Scalability and Environmental Compliance: The process generates basically no three wastes, with the primary byproduct being silver chloride, which can be recycled to recover silver, further enhancing the economic and environmental profile. The use of non-chlorinated solvents and the absence of toxic gas emissions simplify regulatory compliance and reduce the burden on environmental health and safety teams. This green manufacturing profile facilitates easier permitting for new facilities and aligns with the sustainability goals of major defense and aerospace contractors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this energetic salt technology, derived directly from the experimental data and claims within the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating these materials into existing formulations.
Q: What are the thermal stability characteristics of these energetic salts?
A: According to patent CN108148007B, the synthesized salts exhibit excellent thermal stability with decomposition temperatures mostly above 150°C, making them suitable for demanding applications.
Q: How does the silver metathesis method improve safety compared to traditional nitration?
A: The silver metathesis route avoids direct nitration hazards, operates at mild temperatures (often below 50°C), and utilizes water or alcohol solvents, significantly reducing the risk of runaway reactions.
Q: What is the impact sensitivity range for these new compounds?
A: Testing indicates impact energies between 4-40J, classifying several of these salts as insensitive explosives, which enhances handling safety during logistics and manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-3-Hydrazino-5-Methyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of advanced energetic intermediates for next-generation defense and aerospace applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to full-scale manufacturing is seamless and risk-mitigated. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-amino-3-hydrazino-5-methyl-1,2,4-triazole salts meets the exacting standards required for high-performance munitions and propellants.
We invite you to engage with our technical procurement team to discuss how this patented synthesis route can optimize your current supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener, more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique project requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
