Revolutionizing SCH-79797 Production: A Scalable Two-Step Synthetic Strategy for Global Supply Chains
Revolutionizing SCH-79797 Production: A Scalable Two-Step Synthetic Strategy for Global Supply Chains
The pharmaceutical landscape is constantly evolving, driven by the urgent need for more efficient and sustainable manufacturing processes for critical active pharmaceutical ingredients (APIs) and their intermediates. A prime example of this technological leap is found in the synthesis of SCH-79797, a potent non-peptide protease-activated receptor 1 (PAR1) antagonist that has recently garnered attention for its unique dual-targeting antibacterial mechanism against both Gram-negative and Gram-positive bacteria. As detailed in patent CN113620961B, a groundbreaking synthetic methodology has been developed that drastically simplifies the production of this valuable compound. This new approach not only addresses the structural complexity of the quinazolinoindole core but also aligns perfectly with the modern industry's demand for greener chemistry and reduced operational hazards. For R&D directors and supply chain managers alike, understanding this shift from legacy methods to streamlined catalytic pathways is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting future market demands.
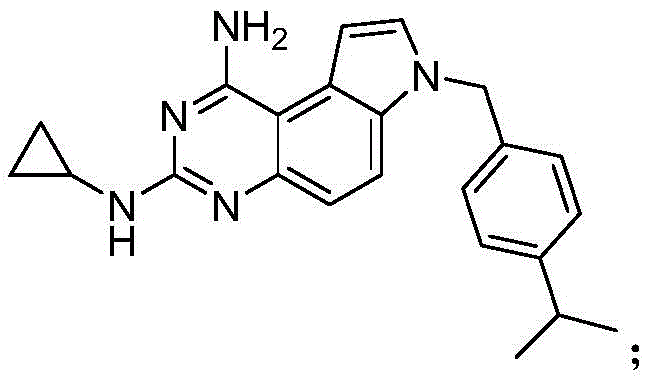
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of SCH-79797 has been plagued by significant safety and efficiency challenges that hindered its widespread commercial adoption. The seminal work published in Bioorganic & Medicinal Chemistry Letters in 1999 outlined a route that relied heavily on the use of trichloromethyl isocyanate, a highly toxic and corrosive reagent derived from phosgene. Handling such hazardous materials requires specialized containment infrastructure, rigorous safety protocols, and expensive waste treatment systems, all of which inflate the overall cost of production. Furthermore, the conventional pathway necessitated the use of liquid ammonia for the substitution of chlorine atoms, introducing苛刻 reaction conditions that are difficult to control on a large industrial scale. These factors combined to create a bottleneck in cost reduction in API manufacturing, making the final product economically less viable for broad therapeutic applications, particularly in the context of emerging antibiotic resistance where cost-effective solutions are paramount.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the methodology disclosed in CN113620961B presents a remarkably concise two-step synthesis that bypasses the need for dangerous phosgene derivatives entirely. By utilizing 1-(4-isopropylbenzyl)-1H-5-aminoindole and dimethyl cyanimide as the primary building blocks, the process constructs the critical quinazoline ring system in a single, high-yielding cyclization event. This strategic redesign eliminates the formation of chlorinated intermediates, thereby removing the subsequent need for harsh ammonolysis steps. The result is a synthetic pathway that is not only shorter but inherently safer and more environmentally benign. This innovation represents a significant leap forward for any organization seeking a reliable pharmaceutical intermediate supplier, as it translates directly into reduced regulatory burden and lower capital expenditure on safety infrastructure.
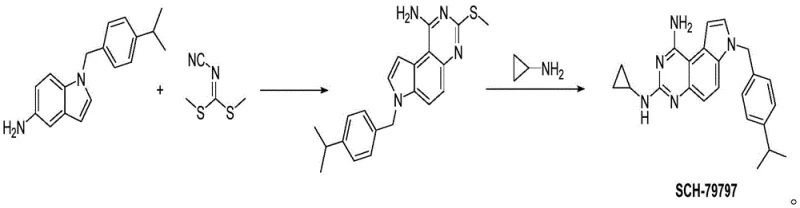
Mechanistic Insights into Dimethyl Cyanimide-Mediated Cyclization
The core of this synthetic breakthrough lies in the efficient utilization of dimethyl cyanimide as a C1 synthon for the construction of the pyrimidine ring fused to the indole scaffold. In the first step, the nucleophilic amino group of the 1-(4-isopropylbenzyl)-1H-5-aminoindole attacks the electrophilic carbon of the cyanimide functionality. This initial addition is followed by an intramolecular cyclization where the adjacent nitrogen atom participates in ring closure, facilitated by thermal energy in the range of 160°C to 170°C. The choice of solvent, such as acetonitrile or N,N-dimethylformamide, plays a crucial role in solubilizing the reactants and stabilizing the transition state, ensuring that the reaction proceeds to completion with minimal formation of side products. This mechanistic elegance allows for the direct formation of the 2-methylthio-4-aminoquinazoline intermediate (Formula I) with exceptional purity, often exceeding 98% without the need for complex chromatographic purification.
Following the formation of the intermediate, the second step involves a nucleophilic aromatic substitution where the methylthio group is displaced by cyclopropylamine. This transformation is conducted under solvent-free vacuum conditions at elevated temperatures between 190°C and 210°C. The absence of solvent in this stage is a critical design feature that drives the equilibrium towards the product by facilitating the removal of volatile byproducts, such as methanethiol, under vacuum. This solvent-free approach not only simplifies the workup procedure but also significantly reduces the environmental footprint of the process. For technical teams focused on impurity control, this high-temperature neat reaction ensures that the final SCH-79797 product is obtained with high structural fidelity, minimizing the presence of unreacted starting materials or hydrolysis byproducts that could complicate downstream formulation.
How to Synthesize SCH-79797 Efficiently
Implementing this novel synthetic route requires precise control over reaction parameters to maximize yield and purity while maintaining operational safety. The process begins with the careful mixing of the aminoindole precursor and dimethyl cyanimide in a suitable polar aprotic solvent, followed by heating to initiate the cyclization. Once the intermediate is isolated, it undergoes a solvent-free aminolysis with cyclopropylamine under vacuum to yield the final target molecule. The detailed standardized synthetic steps, including specific molar ratios, temperature ramps, and workup procedures, are outlined in the guide below to assist process chemists in replicating this high-efficiency protocol.
- React 1-(4-isopropylbenzyl)-1H-5-aminoindole with dimethyl cyanimide in a solvent like acetonitrile or DMF at 160-170°C to form Intermediate I.
- Subject Intermediate I to nucleophilic substitution with cyclopropylamine under solvent-free vacuum conditions at 190-210°C to yield SCH-79797.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this two-step synthesis offers profound strategic advantages that extend far beyond simple chemical curiosity. The elimination of trichloromethyl isocyanate removes a major supply chain risk associated with the sourcing and transport of controlled, hazardous precursors. This shift significantly simplifies the logistics of raw material acquisition and reduces the insurance and compliance costs associated with storing toxic chemicals. Furthermore, the high overall yield of the process means that less raw material is required to produce the same amount of finished goods, leading to substantial cost savings in material procurement. These efficiencies make the new route highly attractive for long-term supply contracts where price stability and continuity are critical.
- Cost Reduction in Manufacturing: The streamlined nature of this two-step process inherently lowers manufacturing costs by reducing the number of unit operations required. By avoiding the use of expensive and hazardous reagents like trichloromethyl isocyanate, the process eliminates the need for specialized scrubbing systems and extensive personal protective equipment, thereby lowering overhead expenses. Additionally, the solvent-free condition in the second step removes the energy-intensive requirement for solvent recovery and distillation, further driving down utility costs. These cumulative effects result in a significantly more economical production model compared to traditional multi-step syntheses.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials such as dimethyl cyanimide and cyclopropylamine enhances the robustness of the supply chain. Unlike legacy methods that depend on reagents with strict regulatory controls, the inputs for this novel route are commercially accessible from multiple global vendors, reducing the risk of supply disruptions. The simplicity of the process also means that production can be scaled up rapidly to meet surges in demand without the need for complex retooling of existing reactor infrastructure, ensuring consistent delivery timelines for downstream partners.
- Scalability and Environmental Compliance: From an environmental perspective, this synthesis aligns perfectly with green chemistry principles by minimizing waste generation and avoiding the use of persistent organic pollutants. The high atom economy of the cyclization step and the solvent-free nature of the final substitution reduce the volume of chemical waste that requires treatment and disposal. This not only lowers environmental compliance costs but also positions the manufacturer as a responsible partner in the pharmaceutical value chain, appealing to clients who prioritize sustainability in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of SCH-79797, based on the detailed specifications provided in the patent literature. These insights are intended to clarify the operational benefits and technical feasibility of adopting this advanced manufacturing route for commercial production.
Q: How does this new synthesis method improve safety compared to conventional routes?
A: Unlike the conventional method described in Bioorg. Med. Chem. Lett. 1999 which utilizes highly toxic trichloromethyl isocyanate and harsh liquid ammonia conditions, this novel route employs dimethyl cyanimide and cyclopropylamine. This substitution significantly reduces hazardous waste generation and eliminates the need for handling extreme phosgene derivatives, thereby enhancing operational safety and environmental compliance.
Q: What are the yield characteristics of this two-step process?
A: The patent data indicates exceptionally high efficiency for this pathway. The first step cyclization achieves yields approaching 97%, while the subsequent aminolysis step maintains yields around 95%. This cumulative high yield profile minimizes raw material loss and maximizes throughput for commercial production.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the process is explicitly designed for scalability. The second step operates under solvent-free vacuum conditions, which simplifies downstream processing by removing the need for extensive solvent recovery systems. Combined with the short two-step sequence, this facilitates easier commercial scale-up of complex heterocycles compared to multi-step traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SCH-79797 Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this optimized synthetic route for the global supply of antibacterial agents and PAR1 antagonists. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of SCH-79797 meets the highest international standards for pharmaceutical intermediates. We are committed to leveraging our technical expertise to deliver high-purity SCH-79797 that supports your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced capabilities can serve as a cornerstone for your successful product launch.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
