Advanced Electrocatalytic N-Nitrosation for Scalable Pharmaceutical Intermediate Manufacturing
The landscape of organic synthesis is undergoing a transformative shift towards greener, more sustainable methodologies, particularly in the construction of nitrogen-nitrogen bonds which are pivotal for pharmaceutical and agrochemical applications. Patent CN111235593B introduces a groundbreaking electrocatalytic free radical coupling reaction that fundamentally redefines the synthesis of nitrogen nitrosation products from secondary amines. This innovative approach utilizes economical and non-toxic metal nitrates as the nitro source, effectively bypassing the need for hazardous traditional nitrating agents. By employing a one-step electrocatalytic mode, the technology achieves high yields of nitrosamine compounds while ensuring a simple reaction process and excellent substrate universality. For industry leaders seeking a reliable pharmaceutical intermediates supplier, this patent represents a critical advancement in process chemistry, offering a pathway to high-purity OLED material and API precursors that aligns with modern environmental standards and operational safety requirements.
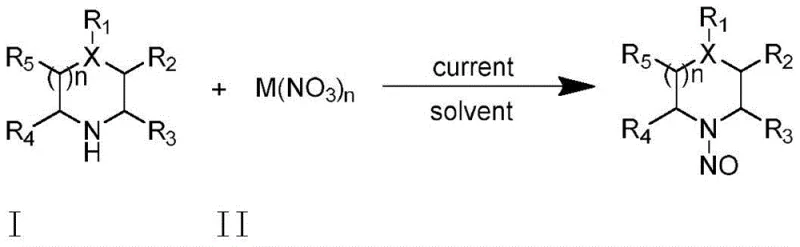
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the N-nitrosation of amines has been predominantly achieved using reagents such as sodium nitrite supported on solid acids or, more commonly in industrial settings, fuming nitric acid. These conventional pathways are fraught with significant drawbacks that pose challenges for both research and large-scale manufacturing. The use of fuming nitric acid, for instance, introduces severe corrosion issues to reactor equipment and generates substantial hazardous waste, complicating disposal and increasing the overall environmental footprint. Furthermore, reactions involving strong acids often require stringent safety protocols to manage the risks associated with exothermic events and toxic gas evolution. Additionally, traditional methods frequently suffer from harsh reaction conditions that can degrade sensitive functional groups on complex substrates, leading to lower product purity and necessitating costly downstream purification steps. The reliance on stoichiometric oxidants also contributes to poor atom economy, making these processes less attractive from a cost reduction in fine chemical manufacturing perspective.
The Novel Approach
In stark contrast, the novel electrocatalytic strategy disclosed in the patent data offers a sophisticated solution to these enduring challenges by leveraging electricity as a clean reagent to drive the transformation. This method operates under mild conditions, typically between 40-80°C, utilizing inexpensive metal nitrates like iron or bismuth nitrate as the nitrogen source instead of corrosive acids. The electrochemical cell setup, featuring a graphite anode and platinum cathode, facilitates a controlled single-electron transfer process that generates reactive radical species in situ without the need for external chemical oxidants. This not only simplifies the reaction workflow into a one-step procedure but also drastically reduces the generation of toxic byproducts. The ability to tune the reaction precisely through current or voltage control allows for exceptional selectivity, minimizing side reactions and ensuring that the resulting nitrosamine compounds are obtained with high purity. This technological leap provides a robust foundation for the commercial scale-up of complex polymer additives and pharmaceutical intermediates, addressing the critical need for safer and more efficient synthetic routes.
Mechanistic Insights into Electrocatalytic N-Nitrosation
Understanding the underlying mechanism is crucial for R&D directors evaluating the feasibility of integrating this technology into existing pipelines. The proposed mechanism involves a double-radical reaction pathway initiated by anodic oxidation. Initially, the secondary amine substrate undergoes a single-electron transfer at the anode surface to form a nitrogen radical cation intermediate. Simultaneously, the metal nitrate salt present in the solution participates in the redox cycle, where the nitrate anion is activated to generate a nitroso radical species. This dual generation of radicals is the key to the reaction's success, as it allows for a rapid cross-coupling event between the nitrogen radical cation and the nitroso radical. The subsequent recombination forms the desired N-N single bond, yielding the N-nitrosation product. At the cathode, protons generated during the oxidation process are reduced to form hydrogen gas, which serves as the only byproduct of the electron balance, further highlighting the green nature of this protocol. This mechanistic clarity ensures that the process is not merely empirical but is grounded in sound electrochemical principles that can be modeled and optimized for various substrates.
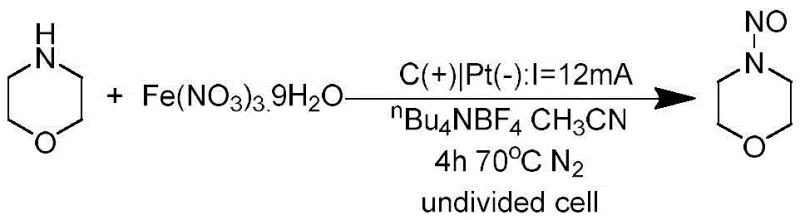
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed pathways. Traditional acid-mediated nitrosation often leads to over-nitration or oxidative degradation of the amine scaffold due to the aggressive nature of the nitronium ion or other high-energy species formed in strong acid media. In the electrocatalytic system, the concentration of reactive radical species is governed by the applied current density, allowing for a steady-state concentration that favors the desired cross-coupling over uncontrolled side reactions. Furthermore, the absence of strong mineral acids prevents acid-catalyzed decomposition of acid-sensitive functional groups that might be present on advanced intermediates. The use of supporting electrolytes such as tetrabutylammonium salts ensures sufficient conductivity without introducing nucleophilic counterions that could interfere with the radical coupling. This precise control over the reaction environment results in a cleaner crude reaction profile, which significantly reduces the burden on purification teams and enhances the overall yield of the target high-purity pharmaceutical intermediates.
How to Synthesize N-Nitrosomorpholine Efficiently
To implement this synthesis route effectively, operators must adhere to specific electrochemical parameters that ensure reproducibility and safety. The process begins with the preparation of an anhydrous reaction mixture containing the secondary amine, the metal nitrate salt, and a suitable supporting electrolyte dissolved in acetonitrile or methanol. It is imperative to maintain an inert atmosphere, typically nitrogen, to prevent interference from oxygen which could quench the radical intermediates or lead to oxidative side products. Once the solution is prepared and deoxygenated, the electrochemical cell is assembled with a graphite rod serving as the sacrificial or inert anode and a platinum sheet as the cathode. The reaction is then driven by applying a constant current, typically in the range of 8-15mA for laboratory scale, while maintaining the temperature between 40-80°C to facilitate ion mobility and reaction kinetics. Monitoring the charge passed through the system is essential to determine the endpoint, after which standard workup procedures involving aqueous washing and organic extraction are employed to isolate the product.
- Mix secondary amine and metal nitrate in acetonitrile or methanol solvent with electrolyte under nitrogen protection.
- Apply constant current (8-15mA) or voltage (2-4V) using graphite anode and platinum cathode at 40-80°C.
- Stir for 4-8 hours, then filter, extract with ethyl acetate, and purify via chromatography to obtain the target nitrosamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrocatalytic technology translates into tangible strategic benefits that extend beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material supply chain; replacing hazardous fuming nitric acid or specialized nitrosating agents with commodity metal nitrates significantly mitigates supply risk and regulatory burden. Metal nitrates such as iron nitrate are widely available, stable, and inexpensive, ensuring a consistent supply of critical reagents without the volatility associated with controlled hazardous chemicals. This shift also impacts storage and handling costs, as the safety requirements for storing metal salts are far less stringent than those for corrosive liquids, thereby reducing insurance premiums and facility maintenance expenses. Moreover, the simplified workup procedure, which avoids neutralization of large volumes of strong acid, reduces the consumption of auxiliary chemicals and lowers the cost of waste treatment, contributing to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents directly lowers the bill of materials for every batch produced. By utilizing electricity as the primary driver for the redox process, the dependency on stoichiometric chemical oxidants is removed, which traditionally accounts for a significant portion of reagent costs in nitrosation reactions. Additionally, the high selectivity of the electrochemical method minimizes the formation of byproducts, which means less material is lost during purification and higher overall throughput is achieved per unit of raw material input. The reduced need for specialized corrosion-resistant equipment also lowers capital expenditure for new production lines, making this technology economically viable for both pilot and commercial scales.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for maintaining continuous production schedules, and this method excels by relying on globally abundant metal salts rather than niche specialty chemicals. The robustness of the reaction conditions allows for flexibility in sourcing, as various metal nitrates (Fe, Co, Bi, Cu) have been shown to be effective, providing multiple options for procurement teams to mitigate single-source risks. Furthermore, the stability of the reagents ensures long shelf-life and ease of transport, reducing the likelihood of supply disruptions due to degradation or shipping restrictions often placed on dangerous goods. This resilience ensures that reducing lead time for high-purity pharmaceutical intermediates becomes a achievable goal rather than a logistical challenge.
- Scalability and Environmental Compliance: Scaling electrochemical processes is increasingly straightforward with modern flow chemistry and undivided cell technologies, as evidenced by the successful gram-scale demonstrations in the patent data. The inherent safety of the system, operating at low voltages and moderate temperatures, simplifies the hazard analysis required for scaling up, accelerating the timeline from lab to plant. Environmentally, the process aligns with strict global regulations by minimizing toxic waste generation and avoiding the release of nitrogen oxides, which are common pollutants in traditional nitrosation. This compliance reduces the administrative burden of environmental reporting and permits, facilitating smoother operations in regions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic N-nitrosation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational parameters and benefits for potential partners. Understanding these details is essential for evaluating the fit of this methodology within your specific production context and for assessing the potential return on investment associated with adopting this greener synthetic route.
Q: What are the advantages of electrocatalytic N-nitrosation over traditional methods?
A: Unlike traditional methods using fuming nitric acid or perchloric acid which are highly corrosive and polluting, this electrocatalytic method uses inexpensive metal nitrates in an acid-free system, significantly reducing environmental hazards and safety risks while maintaining high yields.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent data demonstrates successful gram-scale synthesis with consistent high yields (e.g., 84% on scale), indicating strong potential for commercial scale-up without significant loss of efficiency or selectivity.
Q: What types of secondary amines are compatible with this reaction?
A: The reaction system exhibits excellent substrate universality, successfully converting various cyclic and acyclic secondary amines including morpholine, piperidine, and their derivatives with different substituents into corresponding N-nitrosation products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Nitrosomorpholine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of patent CN111235593B and are fully equipped to leverage this advanced electrocatalytic technology for your custom synthesis needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence means that we do not just supply chemicals; we deliver validated processes that enhance your supply chain stability and product performance.
We invite you to collaborate with us to optimize your current manufacturing strategies and explore the cost efficiencies offered by this novel electrochemical approach. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us partner together to drive innovation and efficiency in the global fine chemical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
