Advanced One-Step Synthesis of 2-Chloro-5-Aminophenol for Commercial Scale-Up
Advanced One-Step Synthesis of 2-Chloro-5-Aminophenol for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to critical building blocks, and the recent disclosure in patent CN113968792A represents a significant leap forward in the synthesis of 2-chloro-5-aminophenol. This vital intermediate, widely used in the construction of complex heterocycles and bioactive molecules, has traditionally been plagued by cumbersome multi-step processes involving hazardous reagents. The new methodology described in this patent shifts the paradigm by utilizing a direct, selective chlorination strategy starting from readily available meta-substituted phenols. By leveraging N-chlorosuccinimide (NCS) or in-situ generated chlorine in polar solvents, this approach achieves high regioselectivity and exceptional purity in a single operational step. For R&D directors and procurement specialists alike, this innovation promises not only a cleaner reaction profile but also a substantial reduction in the environmental footprint associated with legacy manufacturing routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-chloro-5-aminophenol has relied heavily on the Sandmeyer reaction, a classical but notoriously inefficient transformation. As illustrated in the traditional synthetic routes, the process typically begins with the diazotization of 2-amino-5-nitrophenol using sodium nitrite and hydrochloric acid at low temperatures, followed by a substitution reaction with cuprous chloride. 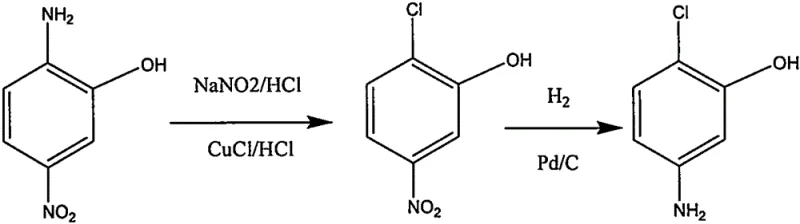 This sequence introduces significant safety hazards due to the instability of diazonium salts and generates massive quantities of heavy metal wastewater containing copper, which requires expensive treatment protocols. Furthermore, alternative routes involving electrolysis or multi-step protection-deprotection sequences often suffer from poor atom economy and low overall yields, sometimes dropping as low as 37% in electrochemical variants. These inefficiencies create bottlenecks in the supply chain, driving up costs and complicating regulatory compliance regarding heavy metal residues in the final active pharmaceutical ingredients.
This sequence introduces significant safety hazards due to the instability of diazonium salts and generates massive quantities of heavy metal wastewater containing copper, which requires expensive treatment protocols. Furthermore, alternative routes involving electrolysis or multi-step protection-deprotection sequences often suffer from poor atom economy and low overall yields, sometimes dropping as low as 37% in electrochemical variants. These inefficiencies create bottlenecks in the supply chain, driving up costs and complicating regulatory compliance regarding heavy metal residues in the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these archaic methods, the novel approach detailed in the patent utilizes a direct electrophilic aromatic substitution mechanism that bypasses the need for diazonium intermediates entirely. By reacting m-aminophenol directly with N-chlorosuccinimide (NCS) in solvents like ethanol, DMF, or tetrahydrofuran, the process achieves precise ortho-chlorination relative to the hydroxyl group. 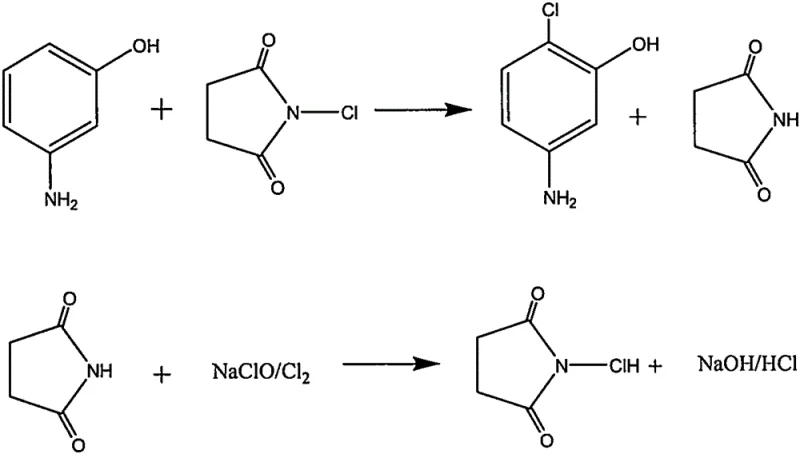 This one-step transformation is not only operationally simpler but also inherently safer, as it avoids the thermal risks associated with diazotization. The reaction conditions are mild, typically ranging between 40°C and 65°C, and the by-product, succinimide, is non-toxic and can potentially be recycled back into the chlorinating agent, creating a closed-loop system that aligns perfectly with modern green chemistry principles. This streamlined workflow drastically reduces the number of unit operations required, translating directly into lower capital expenditure and faster batch turnover times for manufacturing facilities.
This one-step transformation is not only operationally simpler but also inherently safer, as it avoids the thermal risks associated with diazotization. The reaction conditions are mild, typically ranging between 40°C and 65°C, and the by-product, succinimide, is non-toxic and can potentially be recycled back into the chlorinating agent, creating a closed-loop system that aligns perfectly with modern green chemistry principles. This streamlined workflow drastically reduces the number of unit operations required, translating directly into lower capital expenditure and faster batch turnover times for manufacturing facilities.
Mechanistic Insights into Selective Electrophilic Chlorination
The success of this synthesis hinges on the delicate balance of electronic effects governing the electrophilic aromatic substitution on the m-aminophenol substrate. Both the hydroxyl (-OH) and amino (-NH2) groups are strong activating groups that direct incoming electrophiles to the ortho and para positions. However, the specific reaction conditions and the nature of the chlorinating agent (NCS) favor attack at the position ortho to the hydroxyl group, yielding the desired 2-chloro-5-aminophenol with high regioselectivity. The use of polar aprotic or protic solvents helps stabilize the transition state and solubilize the reactants, ensuring homogeneous reaction kinetics. Crucially, the mildness of NCS prevents over-chlorination, a common side reaction in harsher chlorination protocols using molecular chlorine gas, thereby minimizing the formation of di-chlorinated impurities that are difficult to separate. This mechanistic control is essential for maintaining the high purity profiles required by stringent pharmacopeial standards.
From an impurity control perspective, the absence of copper catalysts eliminates the risk of metal-catalyzed oxidative coupling or other side reactions that often complicate the purification of aniline derivatives. The primary by-product, succinimide, is highly water-soluble and can be easily removed during the aqueous workup or crystallization steps described in the patent examples. In cases where m-nitrophenol is used as the starting material, the subsequent reduction step (using Pd/C or iron powder) is a well-established and robust transformation that further ensures the removal of any remaining nitro-containing impurities. The ability to monitor the reaction progress via HPLC until the starting material content drops below 0.1% allows for precise endpoint determination, ensuring consistent batch-to-batch quality and minimizing the presence of unreacted starting materials in the final isolate.
How to Synthesize 2-Chloro-5-Aminophenol Efficiently
The patent provides a comprehensive set of experimental embodiments that serve as a robust foundation for scaling this technology from the laboratory to the pilot plant. The general procedure involves dissolving the starting material, m-aminophenol, in a selected solvent such as tetrahydrofuran or DMF, followed by the controlled addition of the chlorinating agent. Temperature control is paramount, with optimal results observed when the reaction mixture is maintained within a specific window to balance reaction rate and selectivity. Following the reaction, the product is isolated through a straightforward workup involving solvent evaporation and precipitation with ice water, yielding a solid that can be further purified by recrystallization if necessary. The detailed standardized synthesis steps for implementing this protocol are outlined below.
- Dissolve m-aminophenol in a polar solvent such as ethanol, DMF, or tetrahydrofuran within a temperature-controlled reactor.
- Slowly add N-chlorosuccinimide (NCS) solution while maintaining the temperature between 40°C and 65°C to ensure regioselectivity.
- Monitor reaction progress via HPLC until starting material is below 0.1%, then isolate the product through crystallization and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical advantages over traditional methods. The elimination of copper catalysts and diazotization reagents removes the need for specialized waste treatment infrastructure dedicated to heavy metal removal, resulting in significant operational cost savings. Additionally, the raw materials required, specifically m-aminophenol and N-chlorosuccinimide, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague more exotic synthetic pathways. The simplicity of the post-treatment process, which relies primarily on filtration and crystallization rather than complex chromatographic separations, enhances throughput and reduces the consumption of organic solvents, further driving down the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The streamlined one-step nature of this process drastically reduces labor hours and energy consumption compared to multi-step legacy routes. By avoiding the use of expensive transition metal catalysts like palladium or copper in the chlorination step, the direct material costs are significantly lowered. Furthermore, the potential for recycling the succinimide by-product back into the chlorinating agent creates a circular economy within the plant, maximizing atom economy and minimizing waste disposal fees. These factors combine to create a highly cost-competitive manufacturing profile that allows for better margin management in volatile market conditions.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials like m-aminophenol ensures that production schedules are not held hostage by the supply constraints of niche intermediates. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without compromising yield, adds a layer of resilience to the manufacturing process. This reliability translates into shorter lead times for customers, as the reduced processing time allows for faster batch completion and quicker turnaround from order to shipment. Consequently, supply chain planners can maintain leaner inventory levels while still meeting just-in-time delivery commitments.
- Scalability and Environmental Compliance: The patent explicitly mentions the compatibility of this method with micro-reactor technology, which offers superior heat and mass transfer characteristics for exothermic reactions. This feature facilitates safe scale-up from kilogram to tonne quantities without the thermal runaway risks associated with batch reactors. Moreover, the green nature of the process, characterized by the absence of heavy metals and the use of recyclable solvents, aligns with increasingly strict environmental regulations globally. This compliance reduces the regulatory burden on the manufacturer and enhances the sustainability profile of the final product, a key differentiator for environmentally conscious downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the NCS method improve upon traditional Sandmeyer reactions?
A: The NCS method eliminates the need for hazardous diazotization and toxic copper catalysts, significantly reducing heavy metal waste and simplifying purification.
Q: What is the expected purity of 2-chloro-5-aminophenol using this route?
A: Patent data indicates that optimized conditions can achieve purities exceeding 99.5% with yields up to 88%, suitable for sensitive pharmaceutical applications.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is designed for continuous production using micro-reactors or standard kettles, offering excellent thermal control and safety for large-scale operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Aminophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercially viable process requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel chlorination method are fully realized in large-scale manufacturing. We are committed to delivering high-purity 2-chloro-5-aminophenol that meets stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent systems and temperature controls required by this process, guaranteeing a consistent and reliable supply for your critical drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this essential pharmaceutical intermediate.
