Advanced Zero-Discharge Synthesis of Sodium Dimethyl Isophthalate-5-sulfonate for High-Performance Polyester Applications
The global demand for high-performance cationic dyeable polyester (CDP) continues to drive the need for superior third-mononer intermediates, specifically Sodium Dimethyl Isophthalate-5-sulfonate (SIPM-SO3Na). A pivotal advancement in this sector is detailed in Chinese Patent CN102617413B, which outlines a robust method for preparing this critical intermediate using high-concentration fuming sulfuric acid. This technology addresses long-standing inefficiencies in traditional sulfonation processes by optimizing reaction conditions to prevent substrate degradation while implementing a comprehensive mother liquor recycling system. For R&D directors and procurement specialists, this patent represents a significant leap towards sustainable manufacturing, offering a pathway to reduce waste disposal costs while enhancing product consistency. The methodology shifts away from hazardous SO3 gas usage towards a more controllable liquid-phase sulfonation, fundamentally altering the economic and environmental landscape of polyester intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of SIPM-SO3Na has been plagued by significant technical hurdles that impact both yield and environmental compliance. Conventional methods often rely on sulfur trioxide (SO3) gas for sulfonation, a process that is notoriously difficult to control due to its highly exothermic nature. This lack of thermal control frequently leads to the coking of m-phthalic acid, resulting in dark-colored byproducts that are difficult to purify and drastically lowering the overall yield. Alternatively, processes utilizing low-concentration sulfuric acid generate excessive amounts of sodium sulfate as a byproduct during the neutralization phase. This not only complicates the downstream purification steps but also creates a massive burden of high-salinity wastewater that is expensive and challenging to treat, often leading to direct discharge issues that violate modern environmental regulations.
The Novel Approach
The innovative approach described in the patent circumvents these issues by employing 60-70% fuming sulfuric acid (oleum) as the sulfonating agent. By carefully controlling the weight ratio of m-phthalic acid to oleum between 1:0.92 and 1:1.08, the reaction proceeds smoothly at temperatures between 150°C and 220°C without the violent exotherms associated with SO3 gas. This precise control minimizes side reactions and coking, directly translating to higher crude purity. Furthermore, the integration of a closed-loop water system allows for the recycling of process water from crystallization and washing steps back into the neutralization stage. This eliminates the generation of high-concentration wastewater, effectively achieving zero discharge and significantly reducing the operational expenditure related to effluent treatment and raw water consumption.
Mechanistic Insights into Oleum-Mediated Sulfonation and Esterification
The core of this synthesis lies in the sequential transformation of m-phthalic acid through sulfonation and esterification. In the initial step, the electrophilic aromatic substitution is driven by the SO3 present within the fuming sulfuric acid. The reaction conditions are critical; maintaining the temperature at 150-220°C for 8-10 hours ensures complete conversion to the sulfonated intermediate while avoiding the degradation of the aromatic ring. Following sulfonation, the introduction of methanol initiates the esterification process. Here, the carboxylic acid groups are converted to methyl esters. The patent specifies adding methanol at 1.16 to 1.40 times the weight of the starting acid, reacting at 50-90°C for 6-9 hours. This excess methanol drives the equilibrium towards the diester formation, ensuring that the final product possesses the necessary solubility and reactivity characteristics for subsequent polymerization into CDP fibers.
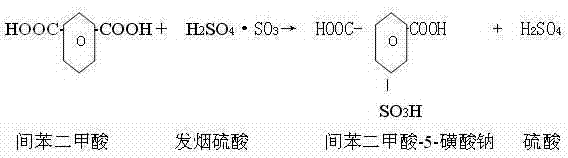
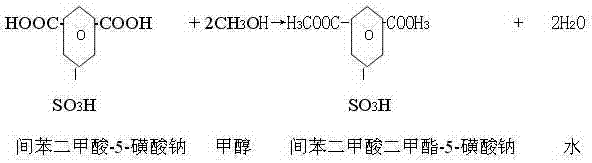
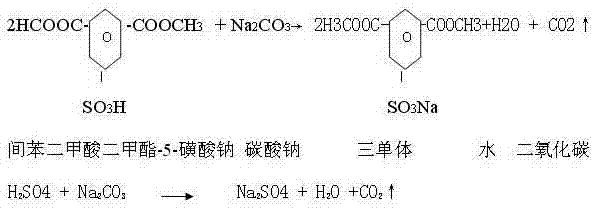
Following esterification, the crude mixture undergoes a critical neutralization and purification phase. The addition of water and sodium carbonate at controlled temperatures (20-55°C) serves a dual purpose: it neutralizes the residual sulfuric acid and converts the sulfonic acid group into its sodium salt form. The stoichiometry here is vital to prevent the hydrolysis of the newly formed ester groups while ensuring complete salt formation. The resulting slurry is then subjected to a rigorous two-step crystallization process. By dissolving the crude solid in hot water (85-95°C) and slowly cooling to below 50°C, impurities such as sodium sulfate and mono-esters remain in the mother liquor while the pure SIPM-SO3Na crystallizes out. This fractional crystallization is the key mechanism for achieving the high saponification values (376-380 mgKOH/g) required for top-tier textile applications.
How to Synthesize Sodium Dimethyl Isophthalate-5-sulfonate Efficiently
Implementing this synthesis route requires precise adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful mixing of m-phthalic acid and oleum, followed by a prolonged heating period to effect sulfonation. Once the sulfonated intermediate is confirmed, methanol is introduced for esterification under reflux conditions. The subsequent neutralization step must be monitored closely for pH levels between 5 and 7.5 to optimize salt formation without degrading the ester functionality. Finally, the purification via hot melt crystallization is essential for removing inorganic salts. For a detailed breakdown of the standardized operating procedures, including specific equipment requirements and safety protocols for handling fuming sulfuric acid, please refer to the technical guide below.
- Mix m-phthalic acid with 60-70% fuming sulfuric acid at a weight ratio of 1: (0.92-1.08) and react at 150-220°C for 8-10 hours to obtain the sulfonated intermediate.
- Add methanol (1.16-1.40 times the weight of the acid) to the sulfonated product and maintain at 50-90°C for 6-9 hours to complete the esterification reaction.
- Dilute the esterified product with water (5-7 times the acid weight) and neutralize with sodium carbonate at 20-55°C until the pH reaches 5-7.5.
- Separate the crude product via centrifugation, purify through two-step hot melt crystallization (dissolving at 85-95°C and cooling below 50°C), and dry to obtain the final high-purity powder.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic advantages beyond mere technical specification compliance. The most significant benefit is the drastic reduction in waste management liabilities. By achieving zero wastewater discharge through the recycling of mother liquors and process water, manufacturers can significantly lower their environmental compliance costs and avoid the risks associated with effluent treatment plant failures. This closed-loop system ensures a more stable production schedule, unaffected by seasonal variations in water availability or tightening local discharge regulations. Furthermore, the elimination of complex desalination steps reduces the energy intensity of the manufacturing process, contributing to a lower overall carbon footprint for the supply chain.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive SO3 gas generation equipment and the associated safety infrastructure, replacing it with standard reactors capable of handling liquid oleum. Additionally, the high yield of 85%, compared to the industry average of 78%, means that less raw material is required to produce the same amount of finished goods. The recycling of methanol from the mother liquor further reduces the consumption of this volatile solvent, leading to substantial savings in raw material procurement costs over the lifecycle of the production campaign.
- Enhanced Supply Chain Reliability: The robustness of the oleum-based sulfonation method reduces the incidence of batch failures caused by coking or runaway reactions. This reliability translates directly into more predictable lead times for customers. Since the process does not rely on the continuous supply of gaseous SO3, which can be logistically challenging to transport and store, the supply chain is less vulnerable to disruptions. The ability to recycle internal streams also buffers the process against minor fluctuations in raw material quality, ensuring consistent output even when feedstock specifications vary slightly.
- Scalability and Environmental Compliance: The technology is inherently scalable, moving seamlessly from pilot batches to multi-ton commercial production without requiring fundamental changes to the reaction chemistry. The zero-discharge capability future-proofs the manufacturing site against increasingly stringent environmental laws regarding industrial wastewater. This compliance advantage is critical for maintaining long-term contracts with multinational corporations that have strict sustainability mandates for their suppliers, thereby securing the supplier's position in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Sodium Dimethyl Isophthalate-5-sulfonate using this advanced fuming sulfuric acid method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for partners evaluating the feasibility of integrating this intermediate into their own polymer synthesis workflows or assessing the long-term viability of the supply source.
Q: How does the high-concentration oleum method improve yield compared to traditional SO3 gas sulfonation?
A: Traditional SO3 gas sulfonation often leads to uncontrollable exothermic reactions and coking of the m-phthalic acid substrate, significantly reducing yield. The patented method utilizes 60-70% fuming sulfuric acid at controlled temperatures (150-220°C), which moderates the reaction kinetics, prevents carbonization, and achieves an average yield of 85%, surpassing the industry average of 78%.
Q: What represents the primary environmental advantage of this manufacturing process?
A: The core environmental breakthrough is the implementation of a closed-loop water circulation system. By recycling mother liquors from crystallization and washing steps back into the neutralization and esterification stages, the process achieves zero wastewater discharge. Additionally, methanol is recovered via distillation and reused, minimizing volatile organic compound (VOC) emissions and raw material waste.
Q: Why is the two-step crystallization purification critical for textile grade applications?
A: Textile applications, specifically cationic dyeable polyester (CDP), require extremely high purity to ensure consistent dye uptake and color fastness. The two-step crystallization process, involving dissolution at 85-95°C followed by cooling below 50°C, effectively removes residual inorganic salts like sodium sulfate and unreacted intermediates, ensuring the final saponification value meets top-grade specifications (376-380 mgKOH/g).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Dimethyl Isophthalate-5-sulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient chemical processes is not just a regulatory requirement but a competitive necessity. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102617413B are fully realized in practical manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of SIPM-SO3Na meets the exacting standards required for high-end cationic dyeable polyester production. Our commitment to quality assurance ensures that the saponification values and impurity profiles remain consistent, safeguarding your downstream polymerization processes.
We invite you to collaborate with us to leverage these advanced synthesis technologies for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to our optimized supply chain. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and sustainability in your operations.
