Advanced Copper-Catalyzed Synthesis of Hydroxyisoindole Diketone for Scalable Pharmaceutical Production
Advanced Copper-Catalyzed Synthesis of Hydroxyisoindole Diketone for Scalable Pharmaceutical Production
The pharmaceutical industry constantly seeks robust synthetic routes for complex immunomodulatory agents, particularly precursors for drugs like lenalidomide and pomalidomide. A significant breakthrough in this domain is detailed in patent CN110724125A, which discloses a novel synthesis method for 3-(7-hydroxy-1-oxoisoindole-2-yl)piperidine-2,6-diketone. This compound serves as a critical intermediate in the construction of diamine-based therapeutics that exhibit potent antitumor and immunoregulatory activities. The traditional reliance on harsh acidic conditions often compromises the structural integrity of these sensitive heterocyclic systems, leading to significant yield losses and purification challenges. By shifting the paradigm towards a copper-catalyzed protocol utilizing hydrochloric acid, this technology offers a pathway to high-purity intermediates with exceptional structural fidelity. For R&D directors and procurement specialists alike, understanding this mechanistic shift is vital for securing a reliable supply chain of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxy-substituted isoindole derivatives has been plagued by the use of concentrated sulfuric acid as the primary reaction medium. While sulfuric acid is a potent dehydrating agent, its strong oxidizing properties and extreme acidity create a hostile environment for the delicate isoindole-1,3-dione ring system. Under these vigorous conditions, the imide bond is highly susceptible to hydrolytic cleavage, resulting in the catastrophic ring-opening of the core structure. As demonstrated in comparative studies within the patent data, reacting the amino-precursor in sulfuric acid leads predominantly to glutaric acid derivatives rather than the desired cyclic product. This side reaction not only decimates the yield of the target molecule but also generates difficult-to-remove impurities that complicate downstream processing. The formation of these ring-opened byproducts represents a fundamental failure of selectivity, rendering the conventional sulfuric acid route economically unviable for high-purity API manufacturing.
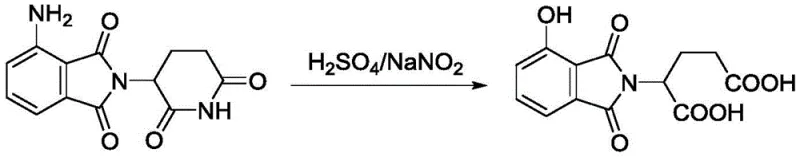
The Novel Approach
In stark contrast to the destructive nature of sulfuric acid, the innovative method described in patent CN110724125A utilizes a combination of hydrochloric acid and a copper catalyst to achieve the transformation with remarkable precision. This approach replaces the violent oxidative environment with a controlled, mild acidic medium that preserves the integrity of the isoindole ring. The reaction proceeds through a diazotization mechanism where the amino group is converted to a diazonium salt, which is subsequently hydrolyzed to the hydroxyl group under the influence of the copper catalyst. This specific catalytic system allows the reaction to proceed at moderate temperatures between 60°C and 80°C, significantly reducing thermal stress on the molecule. The result is a clean conversion that avoids the ring-opening pitfalls of the prior art, delivering the target 3-(7-hydroxy-1-oxoisoindole-2-yl)piperidine-2,6-diketone in high yields. This technological leap ensures that the complex bicyclic framework remains intact, providing a stable foundation for subsequent synthetic steps in the production of immunomodulatory drugs.
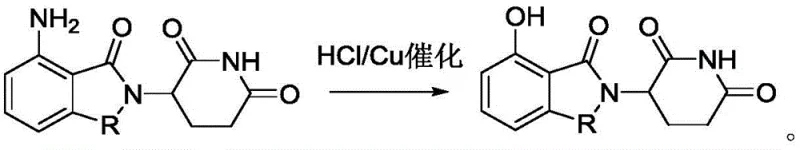
Mechanistic Insights into Copper-Catalyzed Diazotization and Hydrolysis
The core of this synthetic advancement lies in the precise orchestration of the Sandmeyer-type reaction facilitated by copper species. In this mechanism, sodium nitrite reacts with the protonated amine in the presence of hydrochloric acid to generate a reactive diazonium intermediate. Unlike uncatalyzed hydrolysis which can be erratic, the introduction of copper catalysts such as Cu, CuCl, or CuCl2 stabilizes the transition state for the nucleophilic attack by water. The copper center acts as a Lewis acid, coordinating with the diazonium group and lowering the activation energy required for the displacement of nitrogen gas by the hydroxyl moiety. This catalytic cycle is crucial because it allows the reaction to proceed efficiently at lower temperatures, thereby minimizing the kinetic energy available for competing degradation pathways like imide hydrolysis. The choice of copper is also strategic; it is abundant, inexpensive, and easily removed during workup, aligning perfectly with green chemistry principles and cost-reduction strategies in fine chemical manufacturing.
Furthermore, the selectivity of this process is governed by the specific interaction between the catalyst and the substrate's electronic environment. The electron-withdrawing nature of the adjacent carbonyl groups in the isoindole ring typically deactivates the aromatic system, making electrophilic substitution or harsh hydrolysis more likely to attack the imide bond itself. However, the copper-catalyzed pathway directs the reactivity specifically to the diazonium site. By maintaining the reaction temperature strictly between 60°C and 80°C, the process avoids the thermal threshold where the imide bond becomes labile. This kinetic control is essential for preventing the formation of ring-opened glutaric acid impurities. For quality control teams, this means the impurity profile is drastically simplified, with the primary concern being unreacted starting material rather than structurally degraded byproducts. Such high selectivity is a hallmark of a mature, scalable process suitable for GMP production environments.
How to Synthesize 3-(7-hydroxy-1-oxoisoindole-2-yl)piperidine-2,6-diketone Efficiently
Implementing this synthesis requires careful attention to reagent addition and temperature control to maximize the efficiency of the diazotization step. The process begins with the dissolution of the amino-precursor in concentrated hydrochloric acid under ice-bath conditions to stabilize the initial protonation. Following this, the copper catalyst and sodium nitrite are introduced gradually to manage the exothermic formation of the diazonium salt. Once the diazotization is complete, the mixture is heated to the optimal range of 70-80°C to drive the hydrolysis to completion. The simplicity of the workup, involving ether extraction and silica gel chromatography, further underscores the practicality of this method for laboratory and pilot-scale operations. Detailed standardized operating procedures for this synthesis are provided below to ensure reproducibility and safety.
- Dissolve the amino-isoindole precursor in concentrated hydrochloric acid (10-20N) under ice-bath conditions and stir for approximately 1 hour to ensure complete protonation.
- Add the copper catalyst (such as Cu powder, CuCl, or CuCl2) and sodium nitrite slowly to the reaction mixture while maintaining low temperature to control diazotization.
- Heat the reaction mixture to 60-80°C for 1 hour to facilitate the hydrolysis of the diazonium salt, followed by extraction and purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from sulfuric acid-based methods to this copper-catalyzed hydrochloric acid protocol represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The elimination of harsh sulfuric acid not only reduces the corrosion burden on reactor equipment, extending the lifespan of capital assets, but also simplifies waste treatment protocols. Handling concentrated sulfuric acid requires specialized containment and neutralization procedures that add substantial operational overhead. By switching to hydrochloric acid, facilities can leverage existing infrastructure more effectively and reduce the environmental compliance costs associated with sulfate waste disposal. Additionally, the use of inexpensive copper catalysts instead of precious metals ensures that raw material costs remain low and stable, shielding the supply chain from the volatility often seen in the noble metal markets.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high selectivity and yield of the desired product. In the conventional method, the formation of ring-opened byproducts represents a direct loss of valuable starting materials, effectively throwing away nearly all input costs. By preventing this degradation, the new method ensures that the majority of the raw material is converted into saleable product, drastically improving the mass balance of the process. Furthermore, the simplified purification process, which avoids the need to separate complex mixtures of ring-opened acids, reduces solvent consumption and labor hours. These factors combine to lower the overall cost of goods sold (COGS) without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that are sensitive to minor fluctuations in conditions. The robustness of the copper-catalyzed method, with its wide operating temperature window of 60-80°C and tolerance for standard industrial reagents, makes it highly reliable for continuous production. The raw materials, including hydrochloric acid, sodium nitrite, and copper powder, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. This availability ensures that production schedules can be maintained consistently, meeting the rigorous delivery timelines required by downstream API manufacturers. The stability of the synthesis also means fewer batch failures, leading to a more predictable and dependable supply of critical pharmaceutical building blocks.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, particularly when dealing with strong oxidizers and high heat. This novel method operates under mild conditions that are inherently safer to scale from kilogram to tonne quantities. The absence of violent exotherms and the use of less corrosive acids reduce the engineering controls required for large-scale reactors. From an environmental perspective, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on chemical manufacturing. The ability to produce high-purity intermediates with a smaller environmental footprint enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical companies seeking to minimize their Scope 3 emissions and meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 3-(7-hydroxy-1-oxoisoindole-2-yl)piperidine-2,6-diketone. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature. They are designed to provide clarity on the mechanistic advantages and practical implementation of this superior synthetic route. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their own production lines.
Q: Why is hydrochloric acid preferred over sulfuric acid for this synthesis?
A: Sulfuric acid creates overly violent reaction conditions that lead to the hydrolytic ring-opening of the isoindole core, producing unwanted glutaric acid derivatives. Hydrochloric acid provides a milder environment that preserves the cyclic structure.
Q: What is the role of the copper catalyst in this transformation?
A: The copper catalyst facilitates the Sandmeyer-type hydrolysis of the in-situ generated diazonium salt, allowing the replacement of the amino group with a hydroxyl group under温和 conditions without degrading the sensitive imide ring.
Q: Can this process be scaled for commercial API production?
A: Yes, the process operates at moderate temperatures (60-80°C) and uses inexpensive, readily available reagents like HCl and copper powder, making it highly suitable for large-scale commercial manufacturing with robust safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(7-hydroxy-1-oxoisoindole-2-yl)piperidine-2,6-diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced immunomodulators depends on the availability of high-quality, stable intermediates. Our technical team has extensively analyzed the copper-catalyzed synthesis route described in CN110724125A and validated its potential for large-scale application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-(7-hydroxy-1-oxoisoindole-2-yl)piperidine-2,6-diketone meets the exacting standards required for pharmaceutical synthesis. We are committed to delivering products that enable our partners to innovate without compromise.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for next-generation therapeutics. By leveraging our expertise in this specific copper-catalyzed chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a sustainable and cost-effective source of this critical pharmaceutical intermediate, driving your projects forward with confidence and reliability.
