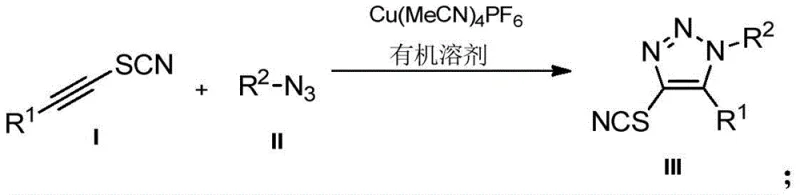
Scalable Synthesis of 4-Thiocyano-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN110746365B introduces a significant advancement in this domain by detailing a highly efficient preparation method for 4-thiocyano-1,4,5-trisubstituted 1,2,3-triazoles. This specific class of compounds is gaining traction due to the unique reactivity of the thiocyano group, which serves as a versatile handle for further derivatization into thioethers, tetrazoles, or other sulfur-containing pharmacophores. The disclosed technology leverages a copper-catalyzed cycloaddition strategy that operates under remarkably mild conditions, addressing long-standing challenges in triazole synthesis regarding regiocontrol and reaction severity. For R&D directors and process chemists, this patent represents a pivotal shift towards more predictable and scalable synthetic routes for generating high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles relied heavily on the thermal Huisgen 1,3-dipolar cycloaddition between azides and alkynes. While conceptually straightforward, this classical approach suffers from severe practical limitations that hinder its utility in modern process chemistry. The reaction typically requires elevated temperatures and extended reaction times, often leading to thermal decomposition of sensitive functional groups present in complex drug candidates. Furthermore, the thermal variant lacks regioselectivity, invariably producing a difficult-to-separate mixture of 1,4-disubstituted and 1,5-disubstituted isomers. This lack of control necessitates cumbersome purification protocols, such as repeated recrystallization or preparative HPLC, which drastically reduce overall throughput and increase manufacturing costs. Additionally, the introduction of a thiocyano group via traditional methods often involves harsh thiocyanation reagents that are incompatible with the delicate balance required for multi-step synthesis.
The Novel Approach
In stark contrast, the methodology described in CN110746365B utilizes a copper(I) catalytic system to drive the cycloaddition with exceptional precision. By employing Cu(MeCN)4PF6 as the catalyst, the reaction proceeds through a concerted mechanism that inherently favors the formation of the 1,4,5-trisubstituted regioisomer. This eliminates the formation of unwanted 1,5-isomers, thereby streamlining the isolation process and enhancing the purity profile of the final API intermediate. The reaction conditions are notably mild, operating effectively at temperatures around 80°C in common organic solvents like chloroform or 1,2-dichloroethane. This thermal gentleness preserves the integrity of sensitive substituents on the aromatic rings, such as methoxy or halogen groups, which might otherwise degrade under the harsh conditions of thermal cycloaddition. The result is a streamlined process that delivers yields ranging from 63% to 81%, providing a reliable pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cu(MeCN)4PF6-Catalyzed Cycloaddition
The core of this technological breakthrough lies in the specific activation of the alkyne moiety by the copper catalyst. The Cu(MeCN)4PF6 complex acts as a source of soluble Cu(I) species, which coordinates with the thiocyanato-internal alkyne to form a reactive copper-acetylide intermediate. This activation lowers the energy barrier for the subsequent attack by the organic azide, facilitating the ring closure at significantly lower temperatures than thermal methods. The presence of the thiocyano group on the alkyne does not inhibit the catalysis; rather, the system tolerates this functionality beautifully, allowing for the direct incorporation of the NCS group into the triazole ring at the 4-position. This mechanistic pathway ensures that the thiocyano group remains intact and correctly positioned for downstream transformations, which is critical for synthesizing libraries of sulfur-rich heterocycles used in medicinal chemistry.
From an impurity control perspective, this catalytic cycle offers distinct advantages over non-catalyzed variants. The high regioselectivity means that the primary impurity profile is dominated by unreacted starting materials rather than structural isomers, which are notoriously difficult to remove. The use of a defined copper complex also minimizes the risk of side reactions such as alkyne polymerization or azide reduction, which can plague less optimized systems. Furthermore, the catalyst loading can be tuned between 0.5 mol% and 50 mol%, with optimal results observed around 5 mol%, allowing process engineers to balance reaction rate against residual metal content. This control is essential for meeting stringent regulatory limits on heavy metals in final drug substances, reducing the burden on scavenging resins or complex workup procedures.
How to Synthesize 4-Thiocyano-1,4,5-Trisubstituted 1,2,3-Triazoles Efficiently
The implementation of this synthesis route is designed for operational simplicity, making it highly attractive for both laboratory scale-up and industrial production. The protocol involves dissolving the thiocyanate-based internal alkyne in a suitable solvent, followed by the addition of the organic azide and the copper catalyst. The mixture is then heated to 80°C and stirred for a period ranging from 12 to 24 hours, depending on the electronic nature of the substrates. Electron-rich aromatics may react slightly faster, while electron-deficient systems might require the full duration to reach completion. Following the reaction, standard workup procedures involving solvent removal and column chromatography yield the pure triazole product. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Dissolve the thiocyanate-based internal alkyne compound in an organic solvent such as chloroform or 1,2-dichloroethane.
- Add the organic azide compound and the Cu(MeCN)4PF6 catalyst to the reaction mixture under air atmosphere.
- Stir the mixture at 80°C for 12 to 24 hours, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation. The reliance on readily available starting materials, such as substituted phenylacetylenes and benzyl azides, ensures a stable supply chain that is not dependent on exotic or single-source reagents. The mild reaction conditions reduce energy consumption compared to high-temperature thermal processes, contributing to lower utility costs and a smaller carbon footprint. Moreover, the high selectivity of the reaction minimizes the generation of hazardous waste associated with separating isomeric byproducts, aligning with increasingly strict environmental compliance standards in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps required to separate regioisomers leads to substantial cost savings in downstream processing. By avoiding the need for high-pressure reactors or extreme temperatures, the capital expenditure for equipment is also reduced, as standard glass-lined or stainless steel reactors can be utilized. The high atom economy of the cycloaddition reaction ensures that raw material costs are optimized, with minimal waste generated from side reactions. Additionally, the ability to run the reaction under air atmosphere removes the need for costly inert gas purging systems, further simplifying the operational setup and reducing overhead expenses.
- Enhanced Supply Chain Reliability: The robustness of the Cu(MeCN)4PF6 catalyst system means that the process is less susceptible to variations in raw material quality or minor fluctuations in reaction parameters. This reliability ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The use of common organic solvents like chloroform and dichloroethane, which are widely available in the global chemical market, mitigates the risk of solvent shortages disrupting production schedules. This stability allows for more accurate forecasting and inventory management, reducing the need for safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The transition from gram-scale to multi-ton production is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards typical of uncatalyzed azide reactions. The mild thermal profile allows for safer scale-up without the need for specialized cooling infrastructure. Furthermore, the high yields and selectivity reduce the volume of solvent and silica gel required for purification, significantly lowering the E-factor (environmental factor) of the process. This green chemistry profile supports corporate sustainability goals and simplifies the permitting process for new manufacturing lines in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential licensees or manufacturing partners.
Q: What are the regioselectivity advantages of this Cu-catalyzed method?
A: Unlike traditional Huisgen cycloadditions which produce mixtures of 1,4- and 1,5-isomers, this Cu(MeCN)4PF6 catalyzed protocol exclusively yields the 1,4,5-trisubstituted 1,2,3-triazole scaffold, significantly simplifying downstream purification.
Q: Can this synthesis tolerate diverse functional groups on the aromatic rings?
A: Yes, the patent data demonstrates excellent tolerance for electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like bromo and chloro, ensuring broad applicability for library synthesis.
Q: Is the catalyst loading economically viable for large-scale production?
A: The process operates efficiently with catalyst loadings as low as 5 mol%, and potentially down to 0.5 mol%, which minimizes heavy metal contamination risks and reduces the cost burden associated with catalyst removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Thiocyano-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced heterocyclic intermediates in accelerating drug discovery pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop innovation to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-thiocyano-1,4,5-trisubstituted 1,2,3-triazoles meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance means that you can rely on us for consistent supply without compromising on the chemical integrity of your critical building blocks.
We invite you to collaborate with us to optimize your supply chain for these high-value intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By leveraging our expertise in copper-catalyzed click chemistry, we can help you reduce lead time for high-purity pharmaceutical intermediates and secure a competitive advantage in the market. Please contact us to request specific COA data and route feasibility assessments for your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
