Scalable Synthesis of 4,6,7-Trifluoro-1H-Indole-2-Carboxylic Acid via Safe Isatin Intermediates
Scalable Synthesis of 4,6,7-Trifluoro-1H-Indole-2-Carboxylic Acid via Safe Isatin Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways for fluorinated heterocycles, particularly indole derivatives which serve as privileged scaffolds in medicinal chemistry. A significant breakthrough in this domain is detailed in patent CN116396204A, which discloses a novel preparation method for 4,6,7-trifluoro-1H-indole-2-carboxylic acid (CAS: 1699249-56-3). This compound is a critical intermediate for developing antiviral precursors, specifically those targeting the Hepatitis B Virus (HBV) replication cycle. Unlike traditional methods that rely on hazardous azide chemistry, this invention leverages a safer isatin-based route. By substituting and condensing 2,3,5-trifluoroaniline with chloral and hydroxylamine, followed by ring closure with sulfuric acid, the process generates a stable 4,6,7-trifluoroisatin intermediate. This strategic shift not only mitigates severe safety risks associated with explosive reagents but also ensures a stable product quality suitable for large-scale industrial amplification, addressing the growing market demand for high-purity fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated indoles has often relied on the Hemetsberger indole synthesis, a pathway that presents formidable challenges for commercial manufacturing. As illustrated in the reaction scheme below, this conventional approach typically utilizes 2,4,5-trifluorobenzaldehyde as a starting material, which undergoes nucleophilic substitution with ethyl azidoacetate. The subsequent ring closure requires refluxing in xylene, followed by hydrolysis and acidification. The primary drawback of this methodology is the inherent danger posed by ethyl azidoacetate, a known explosive substance that creates substantial potential safety hazards in a plant environment. Furthermore, the economic viability of this route is compromised by a notoriously low total yield, reported to be approximately 18%. Such inefficiency results in significant waste generation and higher production costs, rendering it unsuitable for the reliable supply chain requirements of modern pharmaceutical manufacturing.
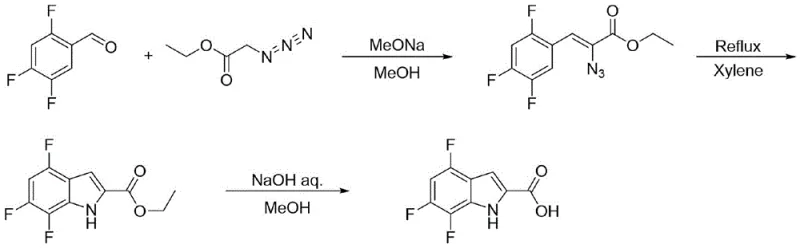
The Novel Approach
In stark contrast to the hazardous legacy methods, the novel approach outlined in the patent data offers a concise and effective synthesis route tailored for industrial safety and efficiency. This method bypasses the use of unstable azides entirely, opting instead for a condensation reaction between 2,3,5-trifluoroaniline and chloral in the presence of hydroxylamine. The resulting intermediate is cyclized using sulfuric acid to form 4,6,7-trifluoroisatin, a robust scaffold that can be easily manipulated. The process continues with N-protection using p-toluenesulfonyl chloride, followed by a selective reduction to generate the indole core. Finally, a Grignard-type carboxylation introduces the essential carboxylic acid functionality at the 2-position. This multi-step sequence is characterized by simple operational flows and the use of easily obtained raw materials, ensuring that the final product achieves a chemical purity exceeding 99%, thereby meeting the stringent specifications required for active pharmaceutical ingredient (API) intermediates.
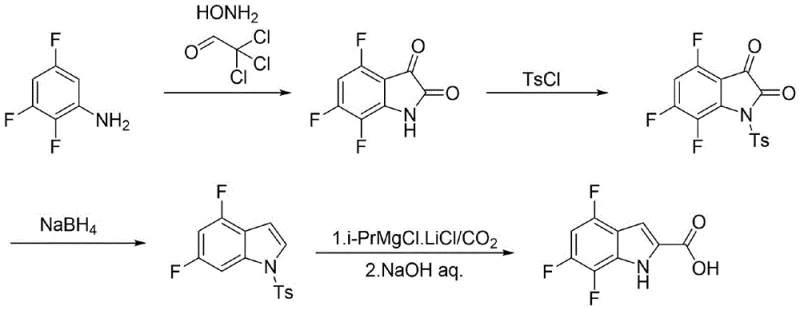
Mechanistic Insights into Isatin Cyclization and Grignard Carboxylation
The core of this synthetic innovation lies in the efficient construction of the trifluoroisatin skeleton and its subsequent transformation. The initial step involves the condensation of 2,3,5-trifluoroaniline with chloral and hydroxylamine to form an oxime intermediate, N-(2,3,5-trifluorophenyl)-2-(hydroxyimino)acetamide. This intermediate is then subjected to a heterogeneous cyclization system comprising toluene, glyoxylic acid, and concentrated sulfuric acid. The use of a heterogeneous system is mechanistically significant as it avoids excessive dehydration and carbonization that often plague homogeneous acidic conditions, thereby stabilizing the yield of the isatin product. Following cyclization, the nitrogen atom is protected with a tosyl group using a strong base such as potassium tert-butoxide or sodium tert-amylate. This protection is crucial as it prevents side reactions during the subsequent reduction step, where sodium borohydride and a boron reagent (BF3·Et2O) selectively reduce the carbonyl groups to methylene units, effectively converting the isatin into the corresponding indole structure without compromising the sensitive fluorine substituents on the aromatic ring.
The final stage of the synthesis demonstrates sophisticated control over regioselectivity and functional group tolerance. The N-protected trifluoroindole undergoes metal-halogen exchange or direct metallation at the 2-position using a complex of isopropyl magnesium chloride and lithium chloride (i-PrMgCl·LiCl). This specific Grignard reagent combination is chosen over n-butyllithium because it offers comparable yields with significantly enhanced safety profiles and easier handling characteristics. Upon exposure to carbon dioxide, the organomagnesium species is carboxylated to form the carboxylic acid moiety. The final deprotection step utilizes aqueous sodium hydroxide to remove the tosyl group, revealing the free NH of the indole ring. This sequence ensures that the highly reactive 2-position is functionalized precisely, while the electron-withdrawing fluorine atoms at the 4, 6, and 7 positions remain intact, preserving the electronic properties necessary for the molecule's biological activity as an HBV inhibitor precursor.
How to Synthesize 4,6,7-Trifluoro-1H-Indole-2-Carboxylic Acid Efficiently
Executing this synthesis requires precise control over reaction conditions, particularly temperature and stoichiometry, to maximize yield and purity. The process begins with the careful dropwise addition of the aniline mixture into the chloral solution, maintaining temperatures between 80-85°C to ensure complete conversion to the oxime intermediate. The subsequent cyclization in toluene and sulfuric acid must be heated to 95-100°C to drive the ring closure effectively. For the reduction step, strict temperature control between -5°C and 0°C is vital when adding sodium borohydride to prevent exothermic runaway, followed by a slow warm-up to room temperature. The final carboxylation demands an inert atmosphere and low temperatures (-15°C to -5°C) during the addition of the Grignard reagent to maintain the stability of the organometallic species before CO2 introduction. Detailed standardized operating procedures for these critical parameters are essential for reproducibility.
- Condense 2,3,5-trifluoroaniline with chloral and hydroxylamine, followed by sulfuric acid-mediated cyclization to form 4,6,7-trifluoroisatin.
- Protect the isatin nitrogen using p-toluenesulfonyl chloride (TsCl) and a strong base like potassium tert-butoxide.
- Reduce the protected isatin to the corresponding indole using sodium borohydride and a boron reagent such as BF3·Et2O.
- Perform lithiation at the 2-position using i-PrMgCl·LiCl, react with CO2 to form the carboxylic acid, and finally deprotect with NaOH.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the Hemetsberger route to this isatin-based methodology represents a substantial optimization of the supply chain risk profile. The elimination of ethyl azidoacetate removes a major bottleneck related to the storage and transport of explosive materials, which often incurs high insurance premiums and requires specialized hazardous goods logistics. By utilizing stable, commodity-grade raw materials like 2,3,5-trifluoroaniline and chloral, the new process drastically simplifies the sourcing strategy. This shift not only enhances supply chain reliability by reducing dependency on niche, high-risk reagents but also facilitates smoother regulatory compliance across different jurisdictions. The robustness of the process ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed quality control tests or the need for extensive reprocessing.
- Cost Reduction in Manufacturing: The economic benefits of this new route are driven primarily by the significant improvement in overall yield and the reduction of waste disposal costs. While the conventional method suffers from a low yield of approximately 18%, the optimized isatin route achieves much higher efficiency, directly lowering the cost of goods sold (COGS) per kilogram of the final API intermediate. Furthermore, the avoidance of expensive and hazardous azide reagents reduces raw material expenditure. The simplified workup procedures, such as the use of standard extraction and crystallization techniques rather than complex purification required for azide byproducts, further contribute to operational cost savings. These factors combine to offer a more competitive pricing structure for downstream pharmaceutical manufacturers.
- Enhanced Supply Chain Reliability: Stability in supply is paramount for long-term drug development projects. This synthesis method utilizes reagents that are widely available in the global chemical market, reducing the risk of shortages that can occur with specialized explosive precursors. The process is designed for large-scale production, meaning it can be readily amplified from pilot plant quantities to multi-ton commercial volumes without fundamental changes to the chemistry. This scalability ensures that as the demand for HBV inhibitors grows, the supply of this critical indole intermediate can be ramped up seamlessly. Additionally, the high purity (>99%) achieved reduces the burden on downstream purification processes, ensuring a steady flow of qualified material to the final drug substance manufacturing sites.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers distinct advantages that align with modern green chemistry principles. The removal of explosive azides significantly lowers the facility's safety risk rating, allowing for more flexible manufacturing scheduling and reduced safety infrastructure costs. The use of sulfuric acid and standard organic solvents like toluene and THF allows for established waste treatment protocols, avoiding the complex decomposition procedures required for azide waste. The process generates fewer hazardous byproducts, simplifying effluent treatment and reducing the environmental footprint of the manufacturing operation. This compliance ease accelerates the approval process for new manufacturing sites and supports sustainable production goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4,6,7-trifluoro-1H-indole-2-carboxylic acid. These answers are derived directly from the technical specifications and advantageous effects detailed in the patent literature, providing clarity on why this specific synthetic route is preferred for industrial applications. Understanding these nuances helps stakeholders make informed decisions regarding vendor selection and process validation.
Q: Why is the Hemetsberger synthesis method considered unsuitable for large-scale production of this compound?
A: The traditional Hemetsberger synthesis relies on ethyl azidoacetate, which is a highly explosive substance posing significant safety hazards. Furthermore, the total yield of this conventional method is typically low, around 18%, making it economically unviable for industrial manufacturing compared to the safer isatin-based route.
Q: What represents the key safety improvement in the new preparation method described in patent CN116396204A?
A: The new method completely eliminates the use of explosive azide reagents. Instead, it utilizes a condensation-cyclization strategy starting from 2,3,5-trifluoroaniline and chloral to form an isatin intermediate, which is then reduced and carboxylated using stable reagents like sodium borohydride and Grignard complexes.
Q: What level of chemical purity can be achieved using this optimized synthetic route?
A: The patented process is designed to achieve exceptional product quality, with the final 4,6,7-trifluoro-1H-indole-2-carboxylic acid reaching a chemical purity of greater than 99%. This high purity is critical for its application as an intermediate in antiviral drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6,7-Trifluoro-1H-Indole-2-Carboxylic Acid Supplier
As the global demand for antiviral therapeutics continues to rise, securing a dependable source of high-quality intermediates is critical for maintaining your development timelines. NINGBO INNO PHARMCHEM stands ready to support your pipeline with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team has thoroughly analyzed the isatin-based route described in CN116396204A and has optimized it for maximum efficiency and safety in our own facilities. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of 4,6,7-trifluoro-1H-indole-2-carboxylic acid meets the exacting standards required for clinical and commercial API synthesis. Our commitment to quality assurance guarantees that you receive a product that is not only chemically pure but also free from problematic impurities that could affect downstream reactions.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of scaling this specific indole derivative, we are equipped to provide comprehensive support. Please contact us today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain partner that prioritizes safety, quality, and continuous improvement, ensuring that your critical antiviral programs proceed without interruption.
