Scalable Production of High-Purity 5-Aminofluorescein via Novel Reduction Technology
Scalable Production of High-Purity 5-Aminofluorescein via Novel Reduction Technology
The pharmaceutical and biotechnology sectors continuously demand high-performance fluorescent markers for diagnostic and research applications, with 5-aminofluorescein standing out as a critical intermediate for covalent binding to human serum albumin. Patent CN113444104A introduces a groundbreaking preparation process that fundamentally reshapes the manufacturing landscape for this valuable compound. By shifting away from hazardous traditional reduction methods, this innovation delivers a pathway characterized by mild reaction conditions, exceptional yield, and superior purity profiles without the need for complex chromatographic purification. For global procurement teams and R&D directors, this patent represents a pivotal opportunity to secure a more reliable 5-aminofluorescein supplier capable of meeting stringent quality standards while mitigating supply chain risks associated with dangerous chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
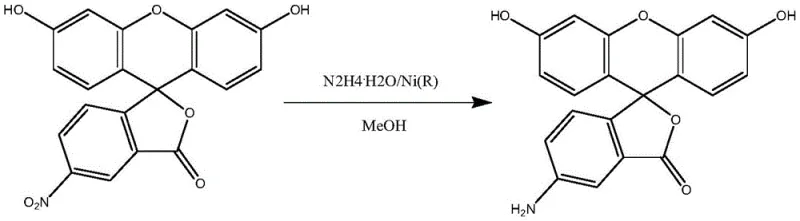
Historically, the synthesis of 5-aminofluorescein has relied heavily on the reduction of 5-nitrofluorescein using catalytic systems involving Raney nickel and hydrazine hydrate. While chemically effective in small-scale laboratory settings, this conventional approach presents severe limitations when translated to industrial manufacturing environments. The reaction is notoriously violent and highly exothermic, generating substantial amounts of heat that create a significant danger of material runaway and even potential explosion hazards if not meticulously controlled. Furthermore, the use of hydrazine hydrate introduces a critical impurity profile issue, as it tends to react with the product to form acyl hydrazine derivatives that are exceptionally difficult to remove during downstream processing. These persistent impurities compromise the final quality of the fluorescent marker, rendering the material unsuitable for sensitive biological applications and effectively blocking its path to mass production.
The Novel Approach
In stark contrast to the perilous legacy methods, the technology disclosed in patent CN113444104A employs a sophisticated array of alternative reducing agents, including sodium hydrosulfite, sodium sulfide, sodium hydrosulfide, or cesium chloride, to drive the transformation safely and efficiently. This novel approach operates under significantly milder conditions, typically utilizing temperatures between 50°C and 200°C, which drastically reduces the thermal load on the reactor and eliminates the risk of explosive exotherms. The process flow is streamlined to include a strategic acid-base workup and a double recrystallization step using dilute acid solutions, which effectively purifies the crude product to high standards. By avoiding the formation of stubborn acyl hydrazine byproducts and eliminating the need for silica gel column chromatography, this method ensures that the final 5-aminofluorescein possesses the high purity required for direct use in subsequent biological conjugation reactions, thereby unlocking true commercial scalability.
Mechanistic Insights into Chemical Reduction and Purification
The core of this innovative synthesis lies in the selective reduction of the aromatic nitro group on the fluorescein scaffold to a primary amine, a transformation that requires precise control over redox potentials to prevent degradation of the sensitive xanthene ring system. When utilizing reducing agents such as sodium hydrosulfite or sulfides in aqueous or alcoholic media, the mechanism proceeds through a series of electron transfer steps that convert the nitro functionality first to nitroso and hydroxylamine intermediates before finally yielding the stable amino group. The presence of specific solvents like methanol or water, often in combination with bases like sodium hydroxide, facilitates the solubility of the intermediate species and stabilizes the transition states, ensuring a smooth progression of the reaction over extended periods ranging from 10 to 30 hours. This prolonged reaction time at moderate temperatures allows for complete conversion of the starting material, minimizing the presence of unreacted 5-nitrofluorescein which could otherwise act as a fluorescent contaminant in the final application.
Beyond the reduction mechanism itself, the purification strategy employed in this patent is equally critical for achieving the reported purity levels of up to 97%. The process leverages the differential solubility of the product and impurities in acidic versus basic media to effect a highly efficient separation. After the reduction is complete, the reaction mixture is cooled and treated with acids such as hydrochloric or sulfuric acid to precipitate insoluble impurities, which are then removed via filtration. The subsequent recrystallization from dilute acid solutions serves as a powerful polishing step, excluding structurally similar byproducts and residual salts from the crystal lattice of the 5-aminofluorescein. Finally, dissolving the purified solid in sodium hydroxide followed by re-acidification ensures that the product is isolated in its desired zwitterionic or neutral form, free from the metallic residues or organic contaminants that typically plague catalytic hydrogenation routes, thus guaranteeing a clean impurity profile suitable for regulatory compliance.
How to Synthesize 5-Aminofluorescein Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing 5-aminofluorescein that balances operational simplicity with high chemical fidelity. The procedure begins with the careful selection of a reducing agent compatible with the chosen solvent system, followed by a controlled heating phase to drive the reduction to completion. Detailed standardized synthetic steps, including specific molar ratios, temperature gradients, and workup parameters, are essential for replicating the high yields observed in the patent examples. For technical teams looking to implement this route, adherence to the specified recrystallization cycles is paramount to achieving the target purity without resorting to chromatography. The detailed standardized synthesis steps are provided in the guide below.
- Mix 5-nitrofluorescein with a selected reducing agent (e.g., sodium hydrosulfite or cesium chloride) and a solvent in a reaction vessel.
- Stir the mixture at elevated temperatures (50-200°C) for 10 to 30 hours to ensure complete reduction of the nitro group.
- Cool the reaction, adjust pH with acid, filter insoluble substances, and perform double recrystallization with dilute acid for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers profound strategic benefits that extend far beyond simple chemical transformation. By replacing hazardous reagents with common, commodity-grade chemicals, the process fundamentally alters the cost structure and risk profile of manufacturing 5-aminofluorescein. The elimination of expensive noble metal catalysts and the removal of silica gel chromatography steps result in a drastic simplification of the production workflow, which directly translates to substantial cost savings in both raw material expenditure and labor hours. Furthermore, the inherent safety of the mild reaction conditions reduces the need for specialized high-pressure equipment or extensive safety containment measures, lowering the capital expenditure barrier for scaling production capacity.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of costly and dangerous hydrazine hydrate with inexpensive reducing agents like sodium hydrosulfite or sodium sulfide, which are widely available in the global chemical market. Additionally, the ability to achieve high purity through simple filtration and recrystallization removes the most expensive unit operation in fine chemical synthesis—column chromatography—thereby significantly reducing solvent consumption and waste disposal costs. This streamlined approach ensures that the cost reduction in pharmaceutical intermediates manufacturing is realized through both lower input costs and higher overall process efficiency, allowing for more competitive pricing structures in the final supply contract.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized catalysts or reagents with long lead times, but this new method utilizes raw materials that are ubiquitous in the chemical industry. The use of common acids, bases, and solvents means that production is not vulnerable to the bottlenecks associated with niche reagent availability. Moreover, the robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond rapidly to fluctuations in demand without compromising on quality. This reliability ensures that downstream customers receive their orders of high-purity 5-aminofluorescein consistently, reducing the lead time for high-purity pharmaceutical intermediates and stabilizing the entire value chain.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is exceptionally well-suited for commercial scale-up of complex fluorescent markers. The absence of heavy metal catalysts like Raney nickel simplifies wastewater treatment and eliminates the regulatory burden associated with heavy metal residue limits in pharmaceutical products. The mild thermal profile of the reaction reduces energy consumption compared to high-temperature or high-pressure alternatives, aligning with modern green chemistry principles. Consequently, manufacturers can scale production from pilot batches to multi-ton annual capacities with confidence, knowing that the process meets rigorous environmental standards and minimizes the generation of hazardous waste streams.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial aspects of the 5-aminofluorescein preparation process as detailed in the patent documentation. These insights are derived directly from the experimental data and comparative analysis provided in the intellectual property filing, offering clarity on safety, purity, and scalability. Understanding these factors is essential for stakeholders evaluating the feasibility of integrating this technology into their existing supply networks.
Q: What are the safety advantages of this new 5-aminofluorescein process compared to traditional methods?
A: Unlike conventional methods using hydrazine hydrate and Raney nickel which pose explosion risks and generate difficult-to-remove acyl hydrazine impurities, this novel process utilizes milder reducing agents like sodium hydrosulfite or cesium chloride, significantly enhancing operational safety and product purity.
Q: Does this synthesis route require silica gel column chromatography for purification?
A: No, one of the key commercial advantages of this patent technology is that it achieves high purity (up to 97%) through simple filtration and recrystallization steps, completely eliminating the need for costly and time-consuming silica gel column chromatography.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is specifically designed for mass production. It uses common, low-cost raw materials and avoids violent exothermic reactions, making it highly scalable and economically viable for commercial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aminofluorescein Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in patent CN113444104A for the production of high-quality fluorescent markers. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this mild reduction process are fully realized in an industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-aminofluorescein meets the exacting standards required for biological and pharmaceutical applications. We are committed to leveraging this advanced technology to deliver a product that is not only chemically superior but also commercially viable for our global partners.
We invite procurement leaders and R&D teams to engage with us to explore how this optimized synthesis route can enhance your project economics and supply security. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the specific financial benefits tailored to your volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this safer, more efficient manufacturing method is seamless and strategically sound.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
