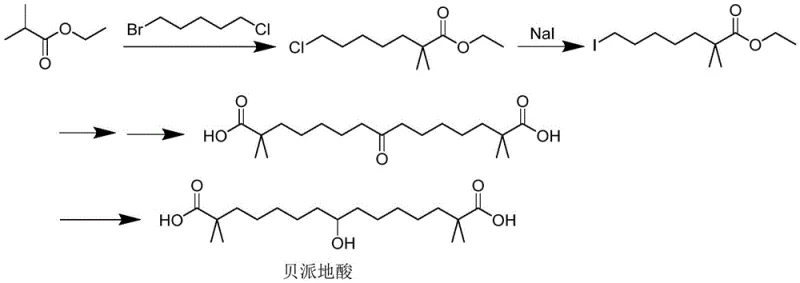
Revolutionizing Bempedoic Acid Production: From Oily Residues to High-Purity Crystals
The pharmaceutical landscape is constantly evolving, driven by the need for more efficient and scalable synthetic routes for critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is documented in patent CN115745778A, which details a novel preparation method for Bempedoic Acid. This patent addresses a longstanding bottleneck in the synthesis of this compound, specifically the difficulty in isolating the final product in a solid, high-purity form. Traditionally, the synthesis of Bempedoic Acid via conventional routes involving ethyl isobutyrate often results in an oily substance that is notoriously difficult to purify. The inventors have discovered that by utilizing a specific reduction pathway followed by a meticulously controlled seed-induced crystallization process, it is possible to bypass these purification hurdles entirely. This technological leap not only ensures the quality of the final API but also dramatically shortens the overall preparation time, offering a compelling value proposition for manufacturers seeking to optimize their production lines for cost and efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Bempedoic Acid has relied on synthetic routes starting from raw materials like ethyl isobutyrate. While chemically feasible, these traditional pathways suffer from severe downstream processing limitations. As highlighted in the background art of the patent, the final product obtained through these conventional methods invariably presents as an oily substance rather than a crystalline solid. This physical state poses a monumental challenge for purification, necessitating the use of column chromatography. In a commercial manufacturing context, column chromatography is highly undesirable due to its low throughput, high solvent consumption, and the significant labor and material costs associated with silica gel handling. Furthermore, even after undergoing this tedious purification process, the resulting product often remains an oil or possesses suboptimal purity levels, typically hovering around 89%. This inefficiency leads to extended production cycles, increased waste generation, and ultimately, a higher cost of goods sold (COGS), making the conventional route economically unsustainable for large-scale supply chains.
The Novel Approach
In stark contrast to the legacy methods, the innovative approach described in patent CN115745778A introduces a paradigm shift in how Bempedoic Acid is isolated and purified. The core of this novelty lies in the transition from a chromatography-dependent workflow to a crystallization-driven one. By synthesizing a specific keto-intermediate and subjecting it to a controlled reduction reaction, the process generates a crude oily substance that is immediately amenable to a specialized crystallization protocol. Instead of relying on the slow separation capabilities of a chromatography column, this method utilizes a binary solvent system comprising ethylene glycol monomethyl ether (or ethylene glycol dimethyl ether) and purified water. Through precise thermal management—heating to dissolve followed by controlled cooling and the strategic addition of seed crystals—the method induces rapid and massive crystallization of the target molecule. This transformation from oil to crystal is not merely a physical change; it represents a fundamental improvement in process robustness, allowing for simple filtration and drying steps that yield a product with exceptional purity levels exceeding 99.5%, thereby eliminating the need for complex and costly chromatographic purification entirely.
Mechanistic Insights into NaBH4 Reduction and Seed-Induced Crystallization
The chemical foundation of this improved synthesis rests on two critical mechanistic pillars: the selective reduction of the carbonyl group and the thermodynamics of nucleation. The process begins with the reduction of a specific intermediate, the structure of which is crucial for the subsequent crystallization behavior. This intermediate, characterized by a central ketone functionality flanked by carboxylic acid groups, is treated with sodium borohydride (NaBH4) in an alkaline aqueous environment. The use of an ice-water bath during this reduction step is vital to control the exothermic nature of the reaction and prevent side reactions that could generate impurities. The hydride ion selectively attacks the ketone carbonyl, converting it into a secondary alcohol, which is the defining structural feature of the final Bempedoic Acid molecule. This reduction step must be executed with precision to ensure that the resulting crude oil contains the correct stereochemical and chemical profile necessary for successful crystallization in the subsequent stage.
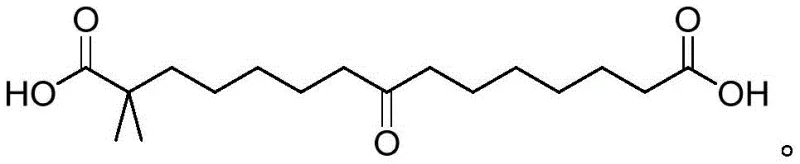
Following the reduction, the mechanism shifts from chemical transformation to physical separation via crystallization. The choice of the solvent system is the linchpin of this mechanism. The mixture of ethylene glycol ethers and water creates a unique solubility profile where the Bempedoic Acid is soluble at elevated temperatures (60-80°C) but becomes sparingly soluble upon cooling. However, supersaturation alone is often insufficient to trigger crystallization in complex organic molecules, frequently leading to 'oiling out' where the compound separates as an amorphous liquid. To overcome this kinetic barrier, the process employs seed-induced nucleation. By introducing a small quantity of pure Bempedoic Acid crystals (0.5-1.5% by mass) into the supersaturated solution at a specific temperature window (10-20°C), the system is provided with a template for crystal growth. This lowers the activation energy required for nucleation, forcing the molecules to align into a rigid crystal lattice rather than forming an disordered oil. This controlled precipitation effectively excludes impurities from the crystal lattice, resulting in the observed high purity of ≥99.5% without the need for further chromatographic refinement.
How to Synthesize Bempedoic Acid Efficiently
Implementing this novel synthesis route requires strict adherence to the parameters defined in the patent to ensure reproducibility and high yield. The process is designed to be operationally simple yet chemically precise, moving away from the artisanal nature of column chromatography towards a standardized manufacturing protocol. The following guide outlines the critical operational phases, from the initial reduction of the intermediate to the final isolation of the crystalline product. Operators must pay close attention to temperature gradients and solvent ratios, as these variables directly influence the nucleation kinetics and the physical form of the final product. For a comprehensive understanding of the exact stoichiometric quantities and equipment specifications required for GMP manufacturing, please refer to the detailed technical documentation provided below.
- Perform reduction of the keto-intermediate using sodium borohydride in an alkaline aqueous solution under ice-water bath conditions to generate the crude oily product.
- Dissolve the resulting oily substance in a heated mixture of ethylene glycol monomethyl ether and purified water, maintaining a temperature between 60°C and 80°C.
- Cool the solution to 10-20°C, introduce Bempedoic Acid seed crystals to induce nucleation, and further lower the temperature to 0-5°C to complete crystallization before filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN115745778A offers tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the column chromatography step, manufacturers can significantly reduce the consumption of expensive organic solvents and silica gel, which are major cost drivers in fine chemical production. This reduction in material usage translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market. Furthermore, the removal of chromatography removes a significant bottleneck in production scheduling. Chromatography is inherently a batch-limited process with long cycle times; replacing it with a continuous-friendly crystallization and filtration sequence allows for much faster turnover of reactor vessels, thereby increasing the overall capacity of the manufacturing facility without the need for capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The economic impact of switching to this crystallization-based method is profound. Traditional chromatography requires vast amounts of eluents and stationary phases, the disposal of which incurs additional environmental and waste management costs. By shifting to a solvent system based on ethylene glycol ethers and water, the process utilizes cheaper and more readily available materials. Additionally, the high purity achieved directly from crystallization (≥99.5%) reduces the need for re-processing or re-crystallization loops, which often result in yield losses. This streamlined approach ensures that a higher percentage of raw materials are converted into saleable product, optimizing the overall material balance and delivering substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the predictability of production lead times. The conventional oily residue method is prone to variability; slight deviations in reaction conditions can make the oil impossible to purify, leading to batch failures and delayed shipments. The new seed-induced crystallization method provides a robust and reproducible endpoint. The formation of a solid precipitate is a clear, binary indicator of reaction success, simplifying quality control (QC) checks. This reliability allows suppliers to commit to tighter delivery schedules and maintain consistent inventory levels. For buyers, this means a reduced risk of stockouts and a more stable supply of high-purity Bempedoic Acid, which is critical for maintaining uninterrupted API production lines.
- Scalability and Environmental Compliance: Scaling up a chromatographic process is notoriously difficult and often impractical for multi-ton production due to the sheer volume of silica and solvents required. In contrast, crystallization is a unit operation that scales linearly and efficiently from the laboratory to the plant floor. The ability to produce Bempedoic Acid via filtration and drying aligns perfectly with standard chemical engineering practices for large-scale manufacturing. Moreover, the reduction in hazardous waste generation (spent silica and mixed solvent wastes) supports stricter environmental compliance standards. This 'green chemistry' aspect not only reduces regulatory burdens but also enhances the sustainability profile of the supply chain, a factor increasingly valued by downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Bempedoic Acid synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in patent CN115745778A, providing clarity on how this technology resolves historical manufacturing pain points. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: How does the new crystallization method improve purity compared to conventional column chromatography?
A: The patented seed-induced crystallization method achieves a purity of ≥99.5%, significantly surpassing the ~89% purity typically obtained via column chromatography, while eliminating the risk of silica gel contamination.
Q: What is the critical role of the solvent system in this synthesis?
A: The specific mixture of ethylene glycol monomethyl ether (or dimethyl ether) and purified water creates a thermodynamic environment that favors rapid nucleation upon seeding, preventing the formation of intractable oils.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, by replacing batch-limited column chromatography with continuous-friendly crystallization and filtration, the process drastically reduces cycle time and solvent consumption, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bempedoic Acid Supplier
The technological advancements detailed in patent CN115745778A represent a significant milestone in the efficient production of Bempedoic Acid, transforming a challenging purification process into a streamlined, high-yield operation. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such chemical innovations. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and stringent purity specifications, ensuring that every batch of Bempedoic Acid we produce meets the highest international standards. We understand that transitioning to a new synthetic route requires confidence in the supplier's capability to execute flawlessly, and our track record in handling complex pharmaceutical intermediates serves as a testament to our operational excellence.
We invite procurement leaders and R&D directors to explore how our adoption of this advanced crystallization technology can optimize your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to process innovation can drive value, reduce lead times, and secure your supply of high-purity Bempedoic Acid for the long term.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
