Industrial Scale-Up of Aminoquinoxaline Diamines for High-Performance Electronic Polymers
The landscape of advanced polymer synthesis is undergoing a significant transformation driven by the need for cost-effective yet high-performance monomers. Patent CN107089954B introduces a groundbreaking methodology for the synthesis of a specific mixture comprising 2-(4-aminophenyl)-3-phenyl-6-aminoquinoxaline and 3-(4-aminophenyl)-2-phenyl-6-aminoquinoxaline. These compounds serve as vital diamine monomers for the production of next-generation polyimides and polyethers, which are essential in the fabrication of flexible electronics and high-temperature resistant materials. The core innovation lies in the strategic selection of 4-nitrophenylacetic acid as the starting raw material, a departure from the traditionally expensive 4-nitrobenzoyl derivatives. This shift not only optimizes the economic feasibility of the process but also streamlines the supply chain by utilizing widely available commodity chemicals. For R&D directors and procurement specialists alike, this patent represents a pivotal opportunity to secure a reliable aminoquinoxaline supplier capable of delivering high-purity intermediates at a fraction of the historical cost.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminoquinoxaline derivatives has been hindered by the reliance on 4-nitrobenzoyl-based starting materials, which present substantial logistical and financial challenges. As detailed in the background art of the patent, previous methods often necessitated the use of these costly precursors, which are not only expensive to procure but also suffer from limited market availability, creating bottlenecks in production scheduling. Furthermore, conventional reduction steps frequently employed hydrazine hydrate, a highly toxic and hazardous reagent that requires stringent safety protocols and complex waste treatment systems. The combination of high raw material costs, supply chain volatility, and significant environmental, health, and safety (EHS) liabilities rendered these traditional pathways unsuitable for sustainable industrial mass production. Consequently, manufacturers faced inflated production costs and restricted scalability, limiting the widespread adoption of these high-performance materials in cost-sensitive applications.
The Novel Approach
The novel approach disclosed in CN107089954B effectively dismantles these barriers by introducing a robust four-step synthetic route anchored by 4-nitrophenylacetic acid. This starting material is characterized by its abundance and low cost, immediately addressing the primary economic constraint of previous methodologies. The process employs a sequence of chlorination, Friedel-Crafts acylation, oxidative condensation, and finally, catalytic hydrogenation. By replacing the hazardous hydrazine reduction with catalytic hydrogenation using standard catalysts like palladium on carbon, the new method significantly enhances operational safety and simplifies the purification workflow. This strategic redesign ensures that the production of high-purity aminoquinoxaline mixtures is not only chemically efficient but also commercially viable for large-scale operations, offering a compelling solution for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into the Multi-Step Synthetic Route
The chemical elegance of this process is rooted in its precise control over reaction conditions to maximize yield and purity while minimizing byproduct formation. The initial chlorination step converts 4-nitrophenylacetic acid into 4-nitrophenylacetyl chloride using reagents such as oxalyl chloride or thionyl chloride, often facilitated by a catalytic amount of DMF to enhance reactivity. This acid chloride intermediate then undergoes a Lewis acid-catalyzed Friedel-Crafts acylation with benzene. The choice of Lewis acid, ranging from aluminum trichloride to zinc chloride, allows for fine-tuning the electrophilic aromatic substitution to ensure high regioselectivity and conversion to 2-(4-nitrophenyl)-1-acetophenone. Following this, the acetophenone derivative reacts with 4-nitro-o-phenylenediamine in the presence of an oxidant (oxygen or sulfur) and an alkaline catalyst. This oxidative condensation is critical for forming the quinoxaline ring system, generating the nitro-substituted intermediate mixture with exceptional purity levels exceeding 99%.
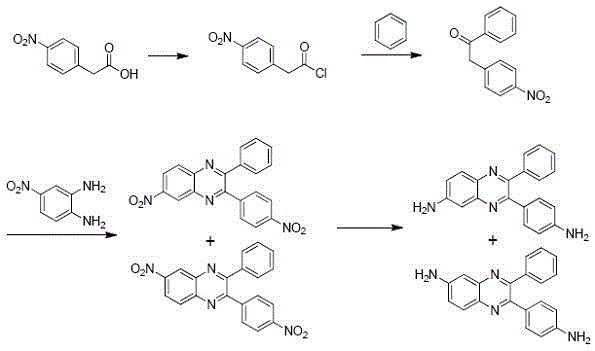
The final transformation involves the reduction of the nitro groups to amino groups via catalytic hydrogenation. This step is meticulously controlled, typically operating at temperatures between 50°C and 65°C and pressures of 0.3 to 0.8 MPa. The use of heterogeneous catalysts such as Pd/C or active nickel ensures that the reduction proceeds cleanly without affecting the sensitive quinoxaline core. From an impurity control perspective, the sequential nature of the synthesis, combined with the high selectivity of the hydrogenation step, prevents the formation of complex side products that are difficult to separate. This mechanistic robustness guarantees that the final diamine mixture meets the stringent purity specifications required for polymerization into high-performance engineering plastics, thereby satisfying the rigorous quality demands of R&D teams focused on material consistency.
How to Synthesize Aminoquinoxaline Mixtures Efficiently
Implementing this synthesis route in a commercial setting requires careful attention to reaction parameters and reagent quality to replicate the high yields reported in the patent examples. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to multi-ton production batches without losing efficiency. Operators must ensure strict temperature control during the exothermic acylation and condensation steps to maintain safety and product integrity. The following guide outlines the standardized operational framework derived from the patent data, providing a clear roadmap for technical teams aiming to integrate this chemistry into their manufacturing portfolio. Detailed standardized synthesis steps are provided in the guide below.
- Chlorinate 4-nitrophenylacetic acid using oxalyl chloride or thionyl chloride to form 4-nitrophenylacetyl chloride.
- Perform Friedel-Crafts acylation with benzene using a Lewis acid catalyst to yield 2-(4-nitrophenyl)-1-acetophenone.
- Condense the acetophenone intermediate with 4-nitro-o-phenylenediamine under oxidative conditions to form the nitroquinoxaline mixture.
- Execute catalytic hydrogenation using Pd/C or Raney Nickel to reduce nitro groups to amines, yielding the final diamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend far beyond simple chemical conversion. The fundamental shift to 4-nitrophenylacetic acid as a feedstock decouples production from the volatile pricing and scarcity issues associated with specialized nitrobenzoyl compounds. This stability in raw material sourcing translates directly into more predictable budgeting and reduced risk of production stoppages due to supply shortages. Furthermore, the elimination of hydrazine hydrate removes a significant regulatory burden and disposal cost, streamlining the overall operational expenditure. The process is inherently designed for industrial mass production, utilizing common solvents and catalysts that are easily sourced globally, ensuring a resilient and agile supply chain capable of meeting fluctuating market demands.
- Cost Reduction in Manufacturing: The economic impact of switching to 4-nitrophenylacetic acid cannot be overstated, as it replaces a high-cost specialty chemical with a widely available commodity. This substitution dramatically lowers the direct material costs per kilogram of the final product. Additionally, the high yields achieved in each step, particularly the near-quantitative conversion in the hydrogenation phase, minimize waste and maximize the output from every batch. The avoidance of expensive metal scavengers or complex purification trains further contributes to substantial cost savings, making the final aminoquinoxaline mixture highly competitive in the global market.
- Enhanced Supply Chain Reliability: By relying on a synthetic pathway built upon common industrial reagents like benzene, thionyl chloride, and standard Lewis acids, the manufacturing process becomes immune to the supply disruptions often seen with niche intermediates. The robustness of the catalytic hydrogenation step also means that the process can be run continuously or in large batches with minimal downtime for catalyst regeneration. This reliability ensures consistent delivery schedules for downstream polymer manufacturers, fostering stronger long-term partnerships and reducing the need for safety stock inventory.
- Scalability and Environmental Compliance: The process is exceptionally well-suited for scale-up, as evidenced by the use of standard unit operations such as distillation, crystallization, and filtration. The replacement of hydrazine with catalytic hydrogenation significantly reduces the toxicity profile of the effluent, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. This green chemistry approach not only mitigates regulatory risk but also aligns with the sustainability goals of modern chemical enterprises, enhancing the brand value of the final polymer products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in CN107089954B, offering clarity on the practical advantages of this route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing production lines.
Q: What is the primary cost advantage of this new synthesis route compared to conventional methods?
A: The primary advantage lies in the substitution of expensive and scarce 4-nitrobenzoyl with 4-nitrophenylacetic acid, a commodity chemical that is significantly cheaper and more readily available in bulk quantities, thereby drastically lowering the raw material input costs for large-scale manufacturing.
Q: How does the catalytic hydrogenation step improve process safety over hydrazine reduction?
A: Unlike traditional methods that rely on hazardous hydrazine hydrate for reduction, this patent utilizes catalytic hydrogenation with standard catalysts like palladium on carbon. This eliminates the handling risks associated with toxic hydrazine and simplifies the downstream purification process, enhancing overall operational safety and environmental compliance.
Q: What are the key applications for the resulting aminoquinoxaline mixture?
A: The resulting diamine monomers are critical precursors for synthesizing high-performance polymers such as quinoxalinyl polyimides, polyethers, and polyesters. These materials are valued in the electronics industry for their exceptional thermal stability, low dielectric constants, and excellent solubility, making them ideal for advanced display and semiconductor applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminoquinoxaline Mixture Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the development of advanced electronic materials. Our technical team has thoroughly analyzed the synthetic pathway described in CN107089954B and is fully prepared to execute this chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aminoquinoxaline mixture delivers the performance required for your high-end polymer applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
