Advanced Manufacturing of Sulfisoxazole Sodium: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing of Sulfisoxazole Sodium: Technical Breakthroughs and Commercial Scalability
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and high-purity synthetic routes for active ingredients, particularly in the veterinary sector where aquaculture demands reliable antibiotic solutions. Patent CN113816916A introduces a highly efficient preparation method for Sulfisoxazole Sodium, a critical sulfonamide derivative used extensively for treating bacterial infections in fish species such as rainbow trout and carp. This technical disclosure outlines a streamlined three-step synthesis involving condensation, hydrolysis, and salt formation, which collectively address longstanding challenges in yield optimization and operational simplicity. By leveraging specific solvent systems and precise pH control mechanisms, this process ensures the production of high-purity intermediates and final API grades suitable for stringent regulatory environments. For global procurement teams and R&D directors, understanding the nuances of this patented pathway is essential for securing a stable supply of this vital veterinary compound.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for sulfonamide derivatives often suffer from cumbersome operational procedures that hinder large-scale commercial viability. Conventional methods frequently rely on harsh reaction conditions, excessive use of volatile organic solvents, and complex work-up procedures involving multiple extraction and recrystallization steps. These inefficiencies not only drive up manufacturing costs due to solvent recovery requirements but also introduce variability in product quality, leading to inconsistent impurity profiles that complicate regulatory approval. Furthermore, older techniques may struggle with the complete removal of unreacted starting materials or side products, necessitating additional purification stages that reduce overall throughput. The environmental footprint of these legacy processes is also significant, generating substantial chemical waste that requires costly disposal protocols, thereby impacting the total cost of ownership for manufacturers.
The Novel Approach
In stark contrast, the methodology described in CN113816916A offers a refined and operationally simple alternative that mitigates many of these historical pain points. The novel approach utilizes a direct condensation strategy followed by a controlled hydrolysis step, eliminating the need for complex intermediate isolations or hazardous reagents. By employing a specific ratio of toluene and pyridine, the reaction environment is optimized to maximize conversion rates while minimizing side reactions. The subsequent hydrolysis is conducted under mild thermal conditions, preserving the integrity of the isoxazole ring while efficiently cleaving the protecting group. This streamlined workflow not only reduces the number of unit operations required but also enhances the overall safety profile of the manufacturing process, making it an attractive option for facilities aiming to modernize their production capabilities for veterinary antibiotics.
Mechanistic Insights into Sulfonamide Condensation and Hydrolysis
The core of this synthesis lies in the initial nucleophilic substitution reaction where 3-aminoisoxazole acts as the nucleophile attacking the sulfonyl chloride group of p-acetamidobenzenesulfonyl chloride. In this step, pyridine serves a dual role as both a solvent and an acid scavenger, neutralizing the hydrochloric acid byproduct generated during the formation of the sulfonamide bond. This mechanistic detail is crucial for R&D teams as it explains the high conversion efficiency observed, with yields consistently exceeding ninety percent in the initial condensation phase. The precise control of temperature during the addition of the sulfonyl chloride is vital to prevent exothermic runaway and ensure the formation of the desired condensate A without degradation of the sensitive isoxazole moiety. Following filtration and washing to neutrality, the intermediate is ready for the next transformation.
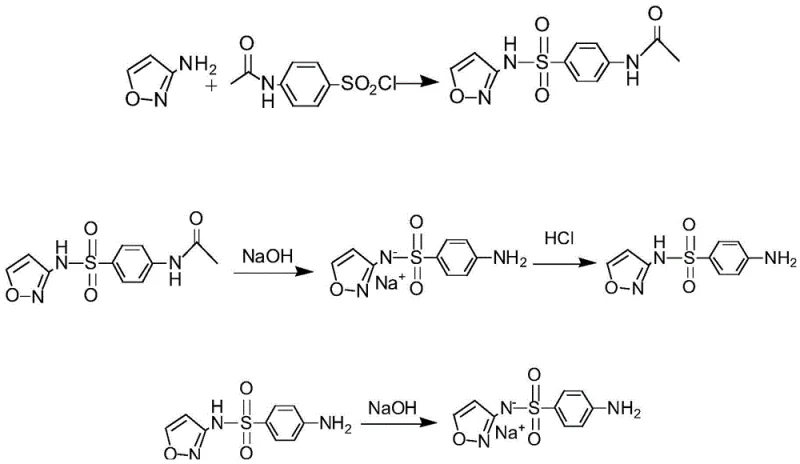
Following the condensation, the process moves to a base-catalyzed hydrolysis mechanism where the acetamide protecting group is removed to reveal the primary amine. The use of liquid caustic soda at elevated temperatures facilitates the cleavage of the amide bond, converting the acetylated intermediate into the free sulfisoxazole acid. This step is meticulously controlled by adjusting the pH to an acidic range post-reaction, which induces precipitation of the product, allowing for easy isolation via filtration. The final salt formation step involves a simple acid-base neutralization where the sulfisoxazole is treated with sodium hydroxide to form the water-soluble sodium salt. This final transformation is critical for the bioavailability of the drug in aquaculture applications, ensuring rapid dissolution and absorption in aquatic environments. The entire sequence demonstrates a logical progression of functional group manipulations designed for maximum efficiency.
How to Synthesize Sulfisoxazole Sodium Efficiently
Implementing this synthesis route requires strict adherence to the specified stoichiometric ratios and thermal profiles to replicate the high yields reported in the patent documentation. The process begins with the careful mixing of reactants in a non-aqueous medium to drive the condensation equilibrium forward, followed by a straightforward aqueous workup. Operators must pay close attention to the pH adjustments during the hydrolysis phase, as this determines the purity of the precipitated acid form. Detailed standard operating procedures regarding the addition rates of reagents and the specific drying temperatures are essential to maintain batch-to-batch consistency. For a comprehensive breakdown of the exact reagent quantities, reaction times, and equipment specifications required to execute this synthesis, please refer to the standardized guide below.
- Perform condensation of 3-aminoisoxazole with p-acetamidobenzenesulfonyl chloride in toluene and pyridine at controlled temperatures.
- Execute alkaline hydrolysis of the condensate using liquid caustic soda followed by acidification to isolate sulfisoxazole.
- Complete the process by reacting sulfisoxazole with sodium hydroxide and spray drying to obtain the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic benefits for procurement managers and supply chain leaders looking to optimize their sourcing of veterinary intermediates. The simplified process flow directly translates to reduced operational complexity, which lowers the barrier for contract manufacturing organizations to scale production without requiring specialized or exotic equipment. By minimizing the number of processing steps and utilizing common industrial solvents like toluene, the method significantly reduces the dependency on scarce or expensive reagents that often bottleneck supply chains. This robustness ensures a more reliable continuity of supply, shielding downstream pharmaceutical companies from market volatility associated with raw material shortages. Additionally, the high purity achieved through this method reduces the need for extensive downstream purification, further driving down the total manufacturing cost.
- Cost Reduction in Manufacturing: The elimination of complex extraction sequences and the use of high-yield reaction steps inherently lower the cost of goods sold. By achieving high conversion rates in the condensation step, raw material waste is minimized, leading to substantial savings on input costs. Furthermore, the ability to isolate intermediates through simple filtration rather than energy-intensive distillation or chromatography reduces utility consumption. These cumulative efficiencies allow for a more competitive pricing structure for the final active ingredient, providing a clear economic advantage over legacy manufacturing routes that suffer from lower throughput and higher waste generation.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 3-aminoisoxazole and p-acetamidobenzenesulfonyl chloride ensures that the supply chain is not vulnerable to single-source bottlenecks. These precursors are commodity chemicals produced by multiple global suppliers, which diversifies risk and stabilizes lead times. The robustness of the reaction conditions, which do not require cryogenic temperatures or high-pressure vessels, means that production can be easily transferred between different manufacturing sites globally. This flexibility is paramount for maintaining uninterrupted supply to the aquaculture industry, where seasonal demand spikes require agile and responsive manufacturing partners capable of ramping up volume quickly.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from laboratory benchtop to pilot plant reactors, indicating strong potential for multi-ton commercial production. The use of aqueous workups and the avoidance of heavy metal catalysts align with modern green chemistry principles, simplifying wastewater treatment and regulatory compliance. Reduced solvent usage and the ability to recover and recycle toluene further diminish the environmental footprint of the operation. For supply chain heads, this translates to fewer regulatory hurdles and a lower risk of production shutdowns due to environmental non-compliance, ensuring a sustainable and long-term manufacturing solution for this critical veterinary antibiotic.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Sulfisoxazole Sodium based on the disclosed patent technology. These insights are derived directly from the experimental data and process descriptions provided in the intellectual property documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing supply networks. The answers reflect the specific advantages of this synthesis method regarding purity, yield, and operational safety.
Q: What are the critical purity specifications for Sulfisoxazole Sodium in this patent?
A: The patent describes a process achieving final product content exceeding 99.2% with rigorous purification steps including activated carbon treatment and precise pH control during crystallization.
Q: How does this synthesis method improve upon traditional sulfonamide manufacturing?
A: This method utilizes a streamlined three-step sequence with mild reaction conditions (70-75°C) and avoids complex extraction processes, significantly simplifying operation and reducing environmental burden.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent provides experimental data scaling up to 50L reactors with consistent yields, demonstrating robust scalability for commercial manufacturing of veterinary antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfisoxazole Sodium Supplier
As the global demand for effective aquaculture treatments continues to rise, securing a partner with deep technical expertise in sulfonamide chemistry is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet the rigorous needs of international clients. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of Sulfisoxazole Sodium meets the highest pharmacopeial standards. We understand the complexities of veterinary drug manufacturing and are equipped to handle the specific challenges of scaling this patented process while maintaining cost-efficiency and supply stability.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can enhance your supply chain resilience. Contact us today to discuss how we can support your long-term strategic goals with reliable, high-quality Sulfisoxazole Sodium supplies.
