Scalable Synthesis of 2-Methyl-5-Bromopyrimidine: A Safe Alternative for Pharmaceutical Manufacturing
Introduction to Advanced Pyrimidine Synthesis
The pharmaceutical and agrochemical industries rely heavily on functionalized heterocycles as critical building blocks for drug discovery and development. Among these, 2-methyl-5-bromopyrimidine (CAS: 7752-78-5) stands out as a versatile intermediate, particularly noted for its utility in synthesizing complement factor D inhibitors for treating autoimmune and neurodegenerative diseases. However, the historical manufacturing landscape for this compound has been fraught with significant safety hazards and economic inefficiencies. Recent intellectual property, specifically patent CN113683571B, discloses a groundbreaking preparation method that fundamentally shifts the paradigm from hazardous organometallic chemistry to a robust, nucleophilic substitution strategy. This technical breakthrough addresses the urgent need for a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the baggage of explosive reagents. By leveraging diazotization followed by ester substitution and decarboxylation, this new methodology offers a pathway that is not only chemically elegant but also commercially viable for multi-ton production.
The significance of this patent extends beyond mere academic interest; it represents a tangible solution to supply chain vulnerabilities associated with pyrophoric reagents. For R&D directors and procurement managers alike, the transition away from dimethyl zinc and trimethyl aluminum towards stable beta-keto esters marks a pivotal moment in process safety and cost optimization. The following analysis dissects the mechanistic advantages, operational parameters, and commercial implications of this novel synthetic route, providing a comprehensive roadmap for stakeholders seeking to secure a sustainable supply of this vital pyrimidine derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-5-bromopyrimidine has been dominated by methodologies that pose severe risks to personnel and infrastructure. As detailed in the background art of the patent, prior literature describes the reaction of 5-bromo-2-iodopyrimidine with dimethyl zinc or trimethyl aluminum. These organometallic reagents are notoriously unstable; dimethyl zinc is pyrophoric, meaning it can spontaneously ignite upon contact with air, while trimethyl aluminum presents similar explosive hazards. The handling of such materials requires specialized inert atmosphere equipment, rigorous safety protocols, and often results in compromised yields due to side reactions or reagent decomposition. Furthermore, the starting material, 5-bromo-2-iodopyrimidine, is significantly more expensive than its chloro or fluoro counterparts due to the cost of iodine and the complexity of iodination processes.
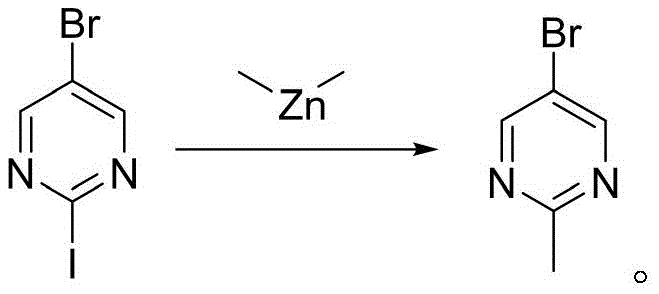
In addition to safety concerns, the economic viability of these traditional routes is questionable. Alternative methods involving the decarboxylation of 2-methyl-5-bromopyrimidine-4-carboxylic acid have been reported, yet they suffer from the high cost and limited availability of the carboxylic acid precursor. The cumulative effect of expensive raw materials, low yields (often reported around 36% for organometallic routes), and the高昂 costs associated with hazard mitigation renders these conventional methods unsuitable for modern, large-scale pharmaceutical intermediate manufacturing. The industry demands a process that eliminates these bottlenecks, ensuring both operator safety and consistent product quality.
The Novel Approach
The inventive method disclosed in CN113683571B circumvents these challenges by employing a three-step sequence that utilizes readily available and stable starting materials. The core innovation lies in the conversion of 2-amino-5-bromopyrimidine or 2-hydroxy-5-bromopyrimidine into a reactive 2-halo intermediate (fluoro, chloro, or bromo), followed by a nucleophilic substitution with beta-keto esters such as ethyl acetoacetate or diethyl malonate. This approach completely avoids the use of pyrophoric organometallics, replacing them with common organic esters that are inexpensive and easy to handle. The final step involves a hydrolysis and decarboxylation sequence that cleanly installs the methyl group at the 2-position of the pyrimidine ring.
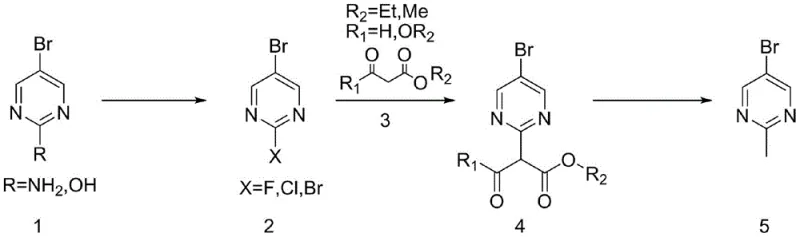
This novel route offers substantial improvements in process robustness. By selecting 2-fluoro-5-bromopyrimidine or 2-chloro-5-bromopyrimidine as the electrophile, the process leverages the high reactivity of these halides towards nucleophilic aromatic substitution (SNAr) without the extreme sensitivity associated with iodides. The subsequent decarboxylation can be achieved under either acidic or basic conditions, providing flexibility in process design. Crucially, the patent reports yields ranging from 71.5% to 85.6%, a dramatic improvement over the 36% yield of the dimethyl zinc method. This increase in efficiency, combined with the use of commodity chemicals, positions this method as the preferred choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nucleophilic Substitution and Decarboxylation
The success of this synthetic strategy hinges on the precise control of two key chemical transformations: the nucleophilic aromatic substitution (SNAr) on the pyrimidine ring and the subsequent thermal decarboxylation. In the first stage, the electron-deficient nature of the pyrimidine ring, activated by the nitrogen atoms and the 5-bromo substituent, facilitates the attack of the enolate derived from the beta-keto ester. When using a strong base like potassium tert-butoxide (t-BuOK) or sodium tert-amylate, the alpha-proton of the ester is abstracted to form a resonance-stabilized enolate. This nucleophile attacks the C2 position of the 2-halo-5-bromopyrimidine, displacing the halide leaving group (fluorine or chlorine). Fluorine, despite being a poor leaving group in aliphatic systems, is an excellent leaving group in SNAr reactions due to its high electronegativity which stabilizes the Meisenheimer complex intermediate.
Following the substitution, the resulting intermediate contains a bulky ester side chain that must be converted to a methyl group. This is achieved through hydrolysis of the ester functionalities to their corresponding carboxylic acids, followed by thermal decarboxylation. Under acidic conditions (e.g., refluxing with HCl), the ester groups are hydrolyzed, and the resulting beta-keto acid undergoes spontaneous loss of carbon dioxide upon heating. Alternatively, under basic conditions (e.g., K2CO3 in DMSO at 110-120°C), the decarboxylation proceeds via a different mechanistic pathway but achieves the same result. This dual-pathway capability allows manufacturers to optimize the process based on their existing equipment and waste treatment capabilities, ensuring high purity specifications are met with minimal impurity carryover.
How to Synthesize 2-Methyl-5-Bromopyrimidine Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly temperature control during the diazotization and substitution steps to maximize yield and minimize byproduct formation. The patent outlines specific protocols for generating the 2-halo intermediates, such as maintaining temperatures between -4°C and 4°C during diazotization to prevent premature decomposition of the diazonium salt. For the substitution step, the choice of solvent (THF or 2-Me-THF) and base concentration is critical to ensure complete conversion of the starting material. The detailed standardized synthesis steps below provide a clear guide for laboratory and pilot-scale execution.
- Convert 2-amino-5-bromopyrimidine or 2-hydroxy-5-bromopyrimidine into a 2-halo intermediate (fluoro, chloro, or bromo) using diazotization or phosphorus oxychloride.
- Perform nucleophilic substitution using ethyl acetoacetate or diethyl malonate with a strong base like potassium tert-butoxide in THF.
- Execute hydrolysis and decarboxylation under acidic reflux or basic high-temperature conditions to yield the final methyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this new synthetic route translates directly into enhanced operational stability and reduced total cost of ownership. The elimination of hazardous organometallic reagents removes the need for specialized storage facilities and expensive safety containment systems, thereby lowering capital expenditure and ongoing operational costs. Furthermore, the reliance on commodity chemicals like ethyl acetoacetate and 2-amino-5-bromopyrimidine ensures a stable supply chain that is less susceptible to the volatility often seen with specialized organometallic suppliers. This shift significantly de-risks the procurement process, allowing for more accurate long-term planning and budget forecasting.
- Cost Reduction in Manufacturing: The replacement of expensive 5-bromo-2-iodopyrimidine and pyrophoric dimethyl zinc with low-cost esters and stable halo-pyrimidines drives a substantial decrease in raw material costs. Additionally, the higher yields achieved (over 70% compared to 36%) mean less waste generation and lower cost per kilogram of finished product. The simplified workup procedures, which avoid complex quenching of reactive metals, further reduce labor and utility expenses, contributing to overall margin improvement.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials, the risk of supply disruption is minimized. Unlike specialized organometallics which may have limited global suppliers and long lead times, the reagents used in this process are produced by multiple chemical manufacturers worldwide. This diversification of the supply base ensures continuity of supply, a critical factor for pharmaceutical companies aiming to maintain uninterrupted production of their final drug substances.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent's emphasis on industrial amplification. The absence of heavy metal catalysts or explosive reagents simplifies environmental compliance and waste disposal. The solvents used, such as toluene and THF, are well-understood and can be efficiently recovered and recycled, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-methyl-5-bromopyrimidine using this patented methodology. These insights are derived directly from the experimental data and advantageous effects described in the patent documentation, providing clarity on the feasibility and benefits of the new route.
Q: Why is the new synthesis route safer than traditional organometallic methods?
A: Traditional methods utilize dimethyl zinc or trimethyl aluminum, which are pyrophoric and explosive. The new patent employs stable beta-keto esters and standard halogenated intermediates, eliminating fire hazards and simplifying storage requirements.
Q: What represents the primary cost driver reduction in this manufacturing process?
A: The process replaces expensive 5-bromo-2-iodopyrimidine and costly organometallic reagents with commodity chemicals like ethyl acetoacetate and 2-amino-5-bromopyrimidine, significantly lowering raw material expenditure.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights industrial amplification prospects due to mild reaction conditions, high yields exceeding 70%, and the avoidance of hazardous reagents that complicate scale-up safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-5-Bromopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our technical team has thoroughly analyzed the pathways described in CN113683571B and possesses the expertise to implement this advanced synthesis at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-methyl-5-bromopyrimidine delivered meets the highest industry standards.
We invite you to collaborate with us to leverage this safer, more efficient manufacturing technology. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized processes can enhance your supply chain resilience and drive down your overall production costs.
