Revolutionizing Aliphatic Diisocyanate Production: A Deep Dive into Non-Phosgene PDI Manufacturing
Revolutionizing Aliphatic Diisocyanate Production: A Deep Dive into Non-Phosgene PDI Manufacturing
The global demand for high-performance aliphatic diisocyanates continues to surge, driven by the need for durable, weather-resistant polyurethane coatings and elastomers. At the forefront of this chemical evolution is the development of 1,5-pentamethylene diisocyanate (PDI), a versatile monomer that offers superior reactivity and material properties compared to traditional aromatic counterparts. A groundbreaking technical disclosure, identified as patent CN114105825A, outlines a sophisticated preparation method that addresses long-standing industry challenges regarding toxicity and process complexity. This patent details a robust, non-phosgene synthetic route that leverages advanced carbonylation chemistry to transform 1,5-pentanediamine into high-purity PDI. For R&D directors and technical leaders, this represents a pivotal shift away from hazardous phosgenation towards a greener, more controllable carbamate pyrolysis pathway. The methodology described not only promises exceptional yields exceeding 95% but also integrates seamlessly with emerging bio-based feedstock strategies, positioning it as a cornerstone technology for the next generation of sustainable polymer manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aliphatic diisocyanates has been dominated by the phosgenation of corresponding diamines, a process fraught with significant operational and environmental liabilities. The reliance on phosgene, a Schedule 1 chemical warfare precursor, necessitates extreme safety protocols, specialized containment infrastructure, and rigorous waste management systems to handle the copious amounts of hydrogen chloride byproduct generated during the reaction. Furthermore, conventional phosgene-free alternatives reported in prior art, such as the oxidation of formamides or the reaction of amine salts with phosgene in solid form, often suffer from multi-stage complexity and poor economic viability. These legacy methods frequently result in lower yields, typically hovering around 50% in some reported non-phosgene attempts, and generate difficult-to-remove impurities that compromise the optical clarity and mechanical performance of the final polyurethane products. The logistical burden of transporting and storing highly toxic reagents also creates substantial supply chain vulnerabilities, making these conventional routes increasingly untenable for modern, sustainability-focused chemical enterprises seeking reliable agrochemical intermediate supplier partnerships or advanced material solutions.
The Novel Approach
In stark contrast to these archaic methodologies, the novel approach detailed in the patent introduces a streamlined two-step sequence centered on the formation and subsequent thermal decomposition of a carbamate intermediate. This strategy bypasses the direct use of phosgene entirely, utilizing safer carbonylating agents such as alkyl carbamates or dialkyl carbonates which are significantly easier to handle and store. The process begins with a catalytic carbonylation reaction that converts 1,5-pentanediamine into 1,5-pentamethylene dicarbamate (PDC) under relatively mild thermal conditions. Crucially, this intermediate is subjected to a rigorous purification protocol involving deamination and desolvation before entering the pyrolysis stage, ensuring that the feedstock for the final conversion is of exceptional quality. By decoupling the carbonylation and isocyanate formation steps, the process allows for precise control over reaction parameters, minimizing side reactions and maximizing the overall atom economy. This innovative workflow not only enhances the safety profile of the manufacturing plant but also drastically simplifies the downstream purification requirements, leading to a final product with purity levels that meet the most stringent specifications for high-end coating applications.
Mechanistic Insights into Catalytic Carbonylation and Pyrolysis
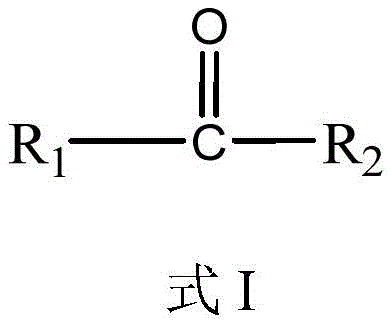
The core of this technological breakthrough lies in the precise selection of carbonylating agents and catalysts that facilitate the efficient transformation of the diamine backbone. The patent specifies a general structure for the carbonylating agent, represented as Formula I, where the substituents R1 and R2 can vary among amino and alkoxy groups such as methoxy, ethoxy, or butoxy. This structural flexibility allows chemists to tune the reactivity of the carbonyl source to match the specific nucleophilicity of the 1,5-pentanediamine, optimizing the formation of the urethane linkage without generating excessive byproducts. The reaction is mediated by a diverse array of metal-based catalysts, including oxides, chlorides, and acetates of metals like zinc, iron, cerium, and titanium, which activate the carbonyl group for nucleophilic attack by the amine. Following the initial synthesis, the intermediate undergoes a critical purification phase to remove unreacted amines and solvents, which is essential for preventing polymerization or degradation during the subsequent high-temperature treatment.
The second mechanistic phase involves the thermal cracking or pyrolysis of the purified 1,5-pentamethylene dicarbamate to release the isocyanate functionality. This endothermic reaction is conducted at elevated temperatures ranging from 150°C to 320°C, often in the presence of a high-boiling solvent such as xylene or chlorobenzene to manage the thermal load and dissolve the reactants effectively. The addition of specific metal catalysts in this stage, such as copper, nickel, or tin, serves to lower the activation energy required for the elimination of the alcohol moiety, thereby accelerating the reaction rate and suppressing the formation of cyclic impurities like N-carbamoylpiperidine. The mechanistic elegance of this route ensures that the cleavage of the carbamate bond occurs selectively, preserving the integrity of the pentamethylene chain while generating the highly reactive isocyanate groups necessary for polyurethane formation. This level of mechanistic control is what enables the process to achieve yields of PDI greater than 95%, a figure that significantly outperforms many existing non-phosgene technologies and rivals the efficiency of traditional phosgenation without the associated toxicological risks.
How to Synthesize 1,5-Pentamethylene Diisocyanate Efficiently
Implementing this synthesis route requires a disciplined approach to reaction engineering, particularly regarding the stoichiometry of the carbonylating agent and the management of thermal gradients during pyrolysis. The patent outlines a preferred embodiment where the molar ratio of the carbonylating agent to the diamine is maintained between 2:1 and 20:1, ensuring complete conversion of the amine while facilitating easy recovery of excess reagent. Operators must carefully monitor the carbonylation step, typically running between 100°C and 240°C for durations of 1 to 24 hours, to ensure the formation of the dicarbamate intermediate reaches equilibrium. Following solid-liquid separation to remove the catalyst, the crude reaction mixture undergoes a multi-tower purification process to strip away volatiles and unreacted species. The detailed standardized synthesis steps for replicating this high-yield process in a pilot or commercial setting are provided in the technical guide below.
- Mix 1,5-pentanediamine with a carbonylating agent (such as alkyl carbamates or carbonates), a solvent, and a metal catalyst, then heat to 100-240°C to form 1,5-pentamethylene dicarbamate.
- Purify the reaction mixture through deamination, desolvation, and removal of excess carbonylating agent to isolate the intermediate.
- Subject the purified intermediate to pyrolysis at 150-320°C in the presence of a second solvent and catalyst to generate the final 1,5-pentamethylene diisocyanate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this non-phosgene technology offers profound advantages that extend far beyond simple chemical yield metrics. The elimination of phosgene from the supply chain removes a major regulatory bottleneck and significantly reduces the insurance and compliance costs associated with handling extremely hazardous substances. By shifting to safer carbonylating agents and bio-compatible feedstocks, manufacturers can secure a more resilient supply chain that is less susceptible to geopolitical disruptions or strict transportation bans on toxic gases. Furthermore, the ability to utilize bio-based 1,5-pentanediamine derived from fermentation opens up new avenues for marketing "green" polyurethane products, appealing to end-consumers and regulatory bodies alike who are increasingly demanding sustainable material sourcing. This transition not only future-proofs the manufacturing asset against tightening environmental regulations but also aligns the production capability with the corporate social responsibility goals of major multinational clients.
- Cost Reduction in Manufacturing: The economic implications of this process are substantial, primarily driven by the simplification of the reaction workflow and the reduction in waste treatment expenses. By avoiding the generation of hydrochloric acid byproducts, the need for expensive corrosion-resistant equipment and neutralization chemicals is drastically reduced, leading to lower capital expenditure and operational overhead. Additionally, the high selectivity of the catalyst system minimizes the loss of valuable raw materials to side products, ensuring that a greater proportion of the input mass is converted into saleable high-purity 1,5-pentamethylene diisocyanate. The ability to recycle solvents and excess carbonylating agents back into the first reaction stage further enhances the overall process efficiency, creating a closed-loop system that maximizes resource utilization and drives down the unit cost of production significantly.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on stable, liquid carbonylating agents rather than gaseous phosgene greatly improves the reliability of raw material delivery and storage. Facilities no longer need to maintain complex onsite phosgene generation units or depend on precarious just-in-time deliveries of hazardous gases, which are often subject to strict transport windows and security escorts. This autonomy allows for larger inventory buffers of safe precursors, ensuring continuous production even during periods of supply disruption. Moreover, the compatibility of this method with bio-based diamines diversifies the sourcing options for the primary feedstock, reducing dependency on petrochemical crackers and insulating the supply chain from volatility in oil and gas markets.
- Scalability and Environmental Compliance: The modular nature of the carbonylation and pyrolysis steps makes this technology highly scalable, allowing for seamless expansion from pilot batches to multi-ton commercial production without fundamental changes to the chemistry. The process inherently generates fewer hazardous emissions and solid wastes, simplifying the permitting process for new facilities or the retrofitting of existing ones to meet stricter environmental standards. The absence of chlorinated byproducts in the main reaction pathway reduces the burden on wastewater treatment plants and lowers the risk of soil contamination, making it an environmentally compliant solution that facilitates smoother interactions with regulatory agencies and local communities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced PDI synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is critical for R&D teams planning scale-up activities and for procurement officers assessing the long-term viability of suppliers utilizing this methodology.
Q: What are the primary safety advantages of this non-phosgene PDI synthesis method?
A: This method completely eliminates the use of highly toxic phosgene gas, replacing it with safer carbonylating agents like alkyl carbamates or carbonates. This significantly reduces equipment corrosion risks, minimizes hazardous waste generation, and lowers the overall potential safety hazards associated with industrial isocyanate production.
Q: How does the purity of the intermediate PDC affect the final PDI yield?
A: The patent emphasizes a rigorous purification step for the 1,5-pentamethylene dicarbamate (PDC) intermediate, involving deamination and desolvation. Achieving high purity (>99.5%) in this stage is critical because impurities can catalyze side reactions during the subsequent high-temperature pyrolysis, thereby ensuring the final PDI yield remains consistently above 95%.
Q: Can this process utilize bio-based raw materials?
A: Yes, the process is designed to work efficiently with 1,5-pentanediamine (PDA), which can be sourced from bio-based fermentation of lysine. This aligns with global sustainability trends, allowing manufacturers to produce renewable aliphatic diisocyanates that compete with petrochemical-derived alternatives like HDI.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Pentamethylene Diisocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, non-phosgene chemistries requires a partner with deep technical expertise and a proven track record in process scale-up. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN114105825A are fully realized in practical, industrial settings. We are committed to delivering high-purity 1,5-pentamethylene diisocyanate that meets stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical techniques to verify every batch. Whether you are developing next-generation automotive coatings or high-performance elastomers, our capability to manufacture complex aliphatic diisocyanates reliably positions us as a strategic ally in your supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your material costs and enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and expert evaluation rather than speculation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
