Advanced Manufacturing of n-Octyl Gallate: A Solvent-Free Esterification Breakthrough for Global Supply Chains
The global demand for high-performance antioxidants in the food and pharmaceutical sectors has necessitated a re-evaluation of traditional synthesis routes for key intermediates like n-octyl gallate. Patent CN101781214A introduces a transformative approach to the esterification of gallic acid, specifically addressing the longstanding inefficiencies associated with solvent management and product isolation. This technology leverages an overdosed esterification strategy where n-octanol acts simultaneously as the reactant and the reaction medium, fundamentally altering the economic and operational landscape of production. By shifting away from complex multi-solvent systems, this method offers a robust pathway for manufacturers seeking to optimize their cost reduction in fine chemical manufacturing while maintaining exceptional product quality. The implications for supply chain resilience are profound, as the simplification of unit operations directly correlates with reduced downtime and enhanced throughput capabilities for large-scale facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gallic acid esters has been plagued by the reliance on aromatic solvents such as benzene or toluene, often requiring the addition of co-solvents like dioxane to facilitate the interaction between the polar gallic acid and the non-polar alcohol. These conventional methods suffer from significant thermodynamic and kinetic barriers; the poor miscibility of reactants in standard aromatic systems leads to sluggish reaction rates and incomplete conversion. Furthermore, the presence of dioxane introduces a severe downstream processing bottleneck, as it forms difficult-to-break azeotropes with the water generated during esterification, making effective dehydration nearly impossible without energy-intensive distillation. The removal of these residual solvents from the final product is not only costly but also raises critical safety and regulatory concerns regarding solvent residues in food-grade additives. Consequently, traditional processes often result in lower yields, higher impurity profiles, and substantial environmental burdens due to solvent waste, creating a fragile supply chain vulnerable to raw material volatility.
The Novel Approach
The innovative methodology disclosed in the patent data circumvents these historical constraints by employing a massive excess of n-octanol, typically ranging from 2.5 to 7.5 times the weight of the gallic acid substrate. This strategic excess drives the esterification equilibrium strongly towards the product side according to Le Chatelier's principle, effectively eliminating the need for external dehydrating agents or entrainers. In this system, n-octanol functions as a self-solvating medium, ensuring homogeneous reaction conditions that drastically accelerate the kinetics, reducing reaction times to merely two hours at a moderate temperature of 80°C. The post-reaction workup is elegantly simplified; the excess alcohol is recovered directly through filtration and vacuum concentration without the need for complex fractional distillation columns typically required to separate dioxane. This streamlined workflow not only preserves the thermal stability of the sensitive gallate ester by avoiding high-temperature pyrogenic distillation but also establishes a closed-loop system where the valuable alcohol reactant is recycled for subsequent batches, delivering substantial operational efficiencies.
Mechanistic Insights into Organic Sulfonic Acid Catalyzed Esterification
The core of this technological advancement lies in the precise selection of the catalytic system and the manipulation of reaction stoichiometry to favor product formation. The process utilizes an organic sulfonic acid, specifically methylsulfonic acid, which offers superior proton-donating capabilities compared to traditional mineral acids like sulfuric acid, yet without the aggressive oxidative properties that lead to charring or degradation of the phenolic hydroxyl groups on the gallic acid ring. This mild yet effective catalysis ensures that the three hydroxyl groups on the aromatic ring remain intact while the carboxylic acid group undergoes efficient nucleophilic attack by the n-octanol. The reaction mechanism proceeds through the protonation of the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon, which is then attacked by the oxygen of the n-octanol molecule. The use of excess alcohol shifts the equilibrium constant effectively, ensuring that the forward reaction dominates until near-complete consumption of the limiting reagent, gallic acid, is achieved.
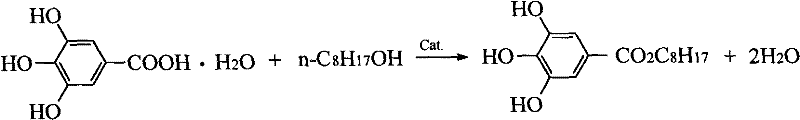
Following the reaction, the control of impurities is managed through a sophisticated crystallization protocol that exploits the differential solubility of the product and unreacted starting materials in cyclohexane. The crude product, obtained after the recovery of excess octanol, is subjected to a double recrystallization process using cyclohexane, a solvent in which the n-octyl gallate has low solubility at ambient temperatures but high solubility when heated. This thermal gradient purification effectively excludes polar impurities and residual catalysts from the crystal lattice. Furthermore, the final washing step with purified water is critical for removing any trace acidic residues, adjusting the pH of the filter cake to a neutral range of 6.0 to 7.0. This rigorous purification sequence guarantees a final product with a purity profile exceeding 99%, satisfying the stringent specifications required for a reliable food additive supplier serving the global nutraceutical and preservative markets.
How to Synthesize n-Octyl Gallate Efficiently
The implementation of this synthesis route requires careful attention to the stoichiometric ratios and temperature controls to maximize the benefits of the excess alcohol strategy. The process begins with the quantitative charging of industrial-grade gallic acid into a reactor, followed by the addition of n-octanol at a ratio optimized around 5 times the weight of the acid, alongside a catalytic amount of methylsulfonic acid. Maintaining the reaction temperature at 80°C for a duration of two hours is sufficient to drive the conversion to completion, after which the mixture is cooled to induce precipitation of the product. Detailed standardized operating procedures regarding filtration rates, vacuum levels for solvent recovery, and specific washing volumes are essential for reproducibility. For the complete technical breakdown and step-by-step laboratory instructions, please refer to the synthesis guide below.
- React gallic acid with 2.5 to 7.5 times its weight in n-octanol using methylsulfonic acid as a catalyst at 80°C for 2 hours.
- Cool the reaction mixture to 20°C, filter, and concentrate the filtrate under reduced pressure to recover excess n-octanol.
- Recrystallize the crude product twice using cyclohexane, wash with purified water to neutral pH, and vacuum dry at 60°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this overdosed esterification technology presents a compelling value proposition for procurement managers and supply chain directors focused on margin improvement and risk mitigation. The elimination of dioxane, a solvent with significant handling costs and regulatory scrutiny, immediately reduces the raw material bill of materials and simplifies waste disposal logistics. Moreover, the ability to recycle the excess n-octanol directly back into the process creates a circular economy within the manufacturing plant, drastically reducing the net consumption of this key reagent. This efficiency translates into a more stable cost structure, shielding the buyer from fluctuations in alcohol market prices. The simplified process flow also means fewer unit operations, which reduces the potential for mechanical failure and maintenance downtime, thereby enhancing the overall reliability of the supply chain.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dual role of n-octanol as both reactant and solvent, which removes the capital and operational expenditure associated with purchasing, storing, and recovering separate solvent systems like dioxane. By avoiding high-temperature distillation steps that are energy-intensive and prone to causing product degradation, the facility saves significantly on utility costs while preserving yield. The use of a highly efficient organic sulfonic acid catalyst further contributes to cost savings by reducing reaction times and minimizing catalyst loading, which lowers the burden on downstream neutralization and washing steps. These cumulative efficiencies result in a leaner manufacturing process with a substantially lower cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and predictable output, which is critical for maintaining inventory levels in a just-in-time manufacturing environment. Because the process does not rely on complex solvent separations that are sensitive to feedstock variations, the batch-to-batch consistency is markedly improved, reducing the rate of off-spec material that could disrupt supply commitments. The moderate reaction conditions (80°C) place less stress on reactor vessels and ancillary equipment compared to high-temperature alternatives, extending asset life and reducing the frequency of unplanned shutdowns. This operational stability allows suppliers to offer more reliable lead times and commit to larger volume contracts with confidence.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the absence of hazardous azeotropic distillations and the use of relatively benign reagents. The reduction in solvent variety simplifies the environmental permitting process and lowers the complexity of wastewater treatment, as the effluent primarily consists of recyclable alcohol and aqueous washings rather than toxic organic mixtures. The high atom economy achieved by recycling the excess alcohol aligns with modern green chemistry principles, making the facility more attractive to environmentally conscious stakeholders. This scalability ensures that the production capacity can be ramped up to meet surging market demand for high-purity n-octyl gallate without requiring prohibitive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of n-octyl gallate using this advanced esterification technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this material into your existing supply chain or product formulations.
Q: Why is excess n-octanol used instead of traditional solvents like dioxane?
A: Using excess n-octanol serves a dual purpose as both reactant and solvent, eliminating the need for difficult-to-separate co-solvents like dioxane. This simplifies the purification process, reduces environmental hazards, and allows for the direct recycling of the unreacted alcohol, significantly lowering production costs.
Q: What are the purity specifications achievable with this esterification method?
A: The patented process utilizes a double recrystallization step with cyclohexane followed by precise pH-controlled washing. This rigorous purification protocol consistently yields n-octyl gallate with a content exceeding 99%, meeting stringent requirements for food additives and pharmaceutical intermediates.
Q: How does this process improve supply chain stability compared to conventional methods?
A: By removing the dependency on specialized solvents like dioxane and reducing reaction times to just 2 hours at moderate temperatures (80°C), the process minimizes equipment corrosion and energy consumption. This operational simplicity enhances batch consistency and ensures a more reliable, continuous supply of the final product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable n-Octyl Gallate Supplier
The technical superiority of the overdosed esterification process underscores the potential for n-octyl gallate to serve as a cornerstone ingredient in next-generation antioxidant formulations. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent are fully realized in our manufacturing operations. Our commitment to quality is backed by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of n-octyl gallate meets the exacting standards required for food and pharmaceutical applications. We understand that consistency is key, and our process controls are designed to deliver uniform particle size and purity profiles batch after batch.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis route, we can offer competitive pricing structures that reflect the inherent efficiencies of the process. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate the quality and supply security of our n-octyl gallate offerings against your internal benchmarks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
