Advanced Biocatalytic Synthesis of S-(+)-2,2-Dimethylcyclopropylcarboxamide for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing critical chiral intermediates, particularly for beta-lactam antibiotics and enzyme inhibitors. Patent CN1786179A introduces a groundbreaking microbiological preparation method for S-(+)-2,2-dimethylcyclopropylcarboxamide, a pivotal precursor in the synthesis of Cilastatin. This specific compound serves as a renal dehydropeptidase inhibitor, essential for enhancing the antibacterial activity of Imipenem by preventing its degradation in the kidney. The disclosed technology represents a significant leap forward from traditional chemical synthesis, leveraging the power of newly screened microbial strains to achieve high stereoselectivity under remarkably mild conditions. By utilizing a dual-enzyme system involving nitrile hydratase and optically selective amidase, this process effectively bypasses the harsh reagents and extreme temperatures associated with legacy methods. For R&D directors and process chemists, this patent offers a robust blueprint for producing high-purity pharmaceutical intermediates with superior control over impurity profiles. The ability to transform racemic 2,2-dimethylcyclopropanecarbonitrile directly into the desired S-enantiomer with an e.e. value exceeding 98% underscores the precision of modern biocatalysis. Furthermore, the integration of these biological steps simplifies the downstream processing, reducing the burden on purification units and aligning perfectly with green chemistry principles. As a reliable pharmaceutical intermediate supplier, understanding such patented innovations is crucial for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,2-dimethylcyclopropylcarboxamide derivatives relied heavily on aggressive chemical hydrolysis protocols that posed significant operational and environmental challenges. Traditional methods often necessitated the use of concentrated strong bases, such as 6mol/L potassium hydroxide solutions, coupled with prolonged reflux periods ranging from 8 to 16 hours. These harsh reaction conditions not only consumed substantial amounts of energy but also frequently led to the formation of undesirable by-products, most notably 2,2-dimethylcyclopropanecarboxylic acid, which complicates the isolation of the target amide. Moreover, chemical hydrolysis lacks inherent stereoselectivity, meaning that producing the optically active S-(+) isomer required additional, costly resolution steps involving chiral resolving agents or extensive chromatography. The generation of saline waste streams from neutralization processes further exacerbated the environmental footprint, creating disposal issues for manufacturing facilities striving to meet increasingly stringent regulatory standards. Consequently, the overall yield and purity of the final product were often compromised, leading to higher production costs and longer lead times for drug manufacturers. These limitations highlighted an urgent need for a more selective and sustainable synthetic strategy that could operate under milder parameters while delivering superior optical purity.
The Novel Approach
In stark contrast to these conventional chemical routes, the novel biocatalytic approach detailed in the patent utilizes specific microbial enzymes to drive the transformation with exceptional specificity and efficiency. By employing Rhodococcus equi CCTCC No.M 205114, the process first converts the readily available racemic nitrile into the corresponding racemic amide through a hydration reaction that occurs at ambient temperatures and neutral pH levels. This initial step eliminates the need for corrosive acids or bases, significantly reducing the risk of equipment corrosion and operator exposure to hazardous chemicals. Subsequently, the introduction of Delftia tsuruhatensis CCTCC No.M 205115 enables a kinetic resolution where the unwanted R-enantiomer is selectively hydrolyzed into a carboxylic acid, leaving the desired S-(+)-amide intact in the reaction mixture. This enzymatic discrimination is highly precise, achieving enantiomeric excess values greater than 98%, which drastically minimizes the need for complex downstream purification. The mild aqueous environment of the biotransformation ensures that sensitive functional groups remain intact, preserving the structural integrity of the molecule throughout the synthesis. This shift from brute-force chemistry to elegant biology not only enhances the quality of the pharmaceutical intermediate but also streamlines the entire manufacturing workflow, offering a compelling alternative for cost reduction in API manufacturing.
Mechanistic Insights into Dual-Enzyme Biocatalytic Kinetic Resolution
The core of this innovative synthesis lies in the synergistic action of two distinct enzymatic activities housed within different microbial hosts, creating a seamless cascade for chiral resolution. The first stage involves the expression of nitrile hydratase (E.C.4.2.1.84) in Rhodococcus equi, an inducible enzyme that facilitates the addition of water across the carbon-nitrogen triple bond of the nitrile group. This reaction is highly chemoselective, converting the nitrile functionality directly into a primary amide without over-hydrolyzing it to the carboxylic acid, a common side reaction in non-enzymatic systems. The enzyme's active site accommodates the bulky 2,2-dimethylcyclopropyl group efficiently, allowing for rapid conversion rates even at relatively high substrate concentrations up to 2% (wt/v). Following the formation of the racemic amide, the second stage employs an optically selective amidase (E.C.3.5.1.4) found in Delftia tsuruhatensis. This enzyme exhibits strict stereospecificity, recognizing and hydrolyzing only the R-(-)-enantiomer of the amide into its corresponding acid and ammonia. This kinetic resolution mechanism effectively acts as a molecular filter, removing the undesired isomer from the pool and enriching the solution with the target S-(+)-isomer. The separation of the resulting acid from the neutral amide is straightforward due to their differing physicochemical properties, allowing for easy extraction of the pure product. This dual-enzyme strategy exemplifies the power of biocatalysis in constructing complex chiral centers with minimal waste generation.
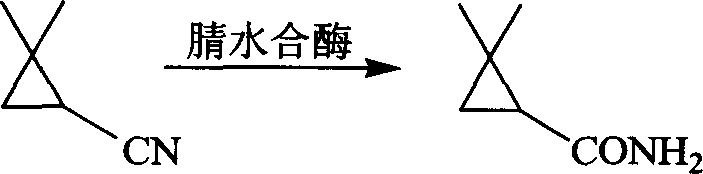
Controlling the impurity profile is paramount in pharmaceutical manufacturing, and this biocatalytic route offers inherent advantages in minimizing side reactions that typically plague chemical synthesis. In traditional alkaline hydrolysis, the high pH and temperature can promote ring-opening of the cyclopropane moiety or polymerization of the nitrile, leading to difficult-to-remove tarry by-products. However, the enzymatic process operates within a narrow physiological pH range of 6.5 to 9.0 and temperatures between 20°C and 40°C, conditions under which the cyclopropane ring remains stable and unreactive towards unintended cleavage. Furthermore, the high specificity of the amidase ensures that only the specific R-enantiomer is consumed, preventing the formation of racemic mixtures that would require further resolution. The use of whole-cell biocatalysts also provides a protective intracellular environment for the enzymes, enhancing their stability and longevity during the reaction. Any minor by-products formed, such as trace amounts of the carboxylic acid from the R-enantiomer, are easily separated during the ethyl acetate extraction step due to their acidity compared to the neutral amide product. This clean reaction profile translates directly into higher purity specifications for the final API intermediate, reducing the analytical burden on quality control laboratories and ensuring consistent batch-to-batch quality for downstream drug formulation.
How to Synthesize S-(+)-2,2-Dimethylcyclopropylcarboxamide Efficiently
Implementing this biocatalytic route requires careful optimization of fermentation and biotransformation parameters to maximize enzyme expression and catalytic efficiency. The process begins with the cultivation of the specific microbial strains in tailored enrichment media where the substrate or its analog serves as the sole nitrogen source, inducing the production of the necessary enzymes. Once the biomass is harvested and suspended in an appropriate buffer system, the substrate is introduced gradually to prevent inhibition effects, allowing the enzymes to convert the nitrile to the amide and subsequently resolve the enantiomers. The reaction progress is monitored using gas chromatography and chiral HPLC to ensure complete conversion of the nitrile and optimal enrichment of the S-enantiomer. Detailed standardized synthesis steps see the guide below.
- Cultivate Rhodococcus equi CCTCC No.M 205114 in induction medium to express nitrile hydratase, then react with racemic 2,2-dimethylcyclopropanecarbonitrile to form racemic amide.
- Separate the biomass and introduce Delftia tsuruhatensis CCTCC No.M 205115 to selectively hydrolyze the R-enantiomer of the amide into carboxylic acid.
- Extract the remaining S-(+)-2,2-dimethylcyclopropylcarboxamide using ethyl acetate and purify via recrystallization to achieve >98% e.e. value.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology presents a strategic opportunity to optimize costs and secure a more resilient supply of critical intermediates. The elimination of harsh chemical reagents such as concentrated potassium hydroxide removes the need for specialized corrosion-resistant reactors and extensive safety protocols associated with handling strong bases. This simplification of the infrastructure requirements leads to substantial cost savings in both capital expenditure for plant equipment and operational expenditure for maintenance and safety compliance. Additionally, the mild reaction conditions significantly reduce energy consumption, as there is no need for high-temperature reflux or cryogenic cooling, contributing to a lower carbon footprint and alignment with corporate sustainability goals. The high selectivity of the enzymes reduces the volume of organic solvents required for purification, as fewer by-products are generated that need to be washed away or separated. This efficiency gain allows for higher throughput in existing facilities without the need for major expansions, effectively increasing capacity to meet market demand. Overall, the transition to this biological pathway creates a leaner, more agile manufacturing process that is less susceptible to fluctuations in raw material prices for aggressive chemicals.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive and hazardous chemical reagents, which lowers both material costs and waste disposal fees significantly. By avoiding the generation of large volumes of saline wastewater typical of acid-base neutralization steps, the facility saves considerably on effluent treatment costs, which are often a hidden but major expense in chemical manufacturing. The high yield and purity achieved directly from the bioreactor minimize the loss of valuable starting materials during purification, ensuring that a greater proportion of the input mass is converted into saleable product. Furthermore, the enzymes act as reusable catalysts within the cells, providing a high turnover number that dilutes the cost of the biocatalyst over a large volume of product. These factors combine to create a highly economical production model that enhances profit margins while maintaining competitive pricing for customers.
- Enhanced Supply Chain Reliability: Relying on fermentation-based production diversifies the supply chain away from petrochemical-derived reagents, which can be subject to volatile market pricing and availability issues. The microbial strains used are robust and can be stored long-term, ensuring that production can be restarted quickly without the need for complex catalyst synthesis or sourcing of rare metal ligands. The aqueous nature of the reaction reduces dependence on large quantities of organic solvents, mitigating risks associated with solvent supply disruptions or regulatory restrictions on volatile organic compounds. This stability in raw material requirements allows for more accurate forecasting and inventory management, reducing the likelihood of stockouts or production delays. Consequently, partners can rely on a consistent and uninterrupted flow of high-quality intermediates, strengthening the overall resilience of the pharmaceutical supply network.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is inherently straightforward as it utilizes standard stirred-tank reactors common in the fermentation industry, allowing for seamless transition from pilot to commercial scale. The process generates minimal hazardous waste, primarily consisting of biodegradable biomass and aqueous buffers, which simplifies compliance with increasingly strict environmental regulations regarding industrial discharge. The absence of heavy metal catalysts eliminates the risk of metal contamination in the final product, a critical quality attribute for pharmaceutical ingredients that often requires costly removal steps. This environmental friendliness not only reduces regulatory hurdles but also enhances the brand reputation of the manufacturer as a sustainable partner in the healthcare value chain. The ability to scale up without compromising safety or environmental standards makes this technology a future-proof solution for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented biocatalytic method for producing chiral amides. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. We encourage further discussion with our technical team to explore specific customization options for your project needs.
Q: What are the advantages of this biocatalytic method over traditional chemical hydrolysis?
A: Unlike traditional methods requiring 6mol/L KOH and high-temperature reflux which generate significant by-products, this microbial method operates under mild conditions (20-40°C, neutral pH) with high stereoselectivity (>98% e.e.) and minimal environmental pollution.
Q: Which specific microbial strains are utilized in this patented process?
A: The process utilizes two novel strains: Rhodococcus equi CCTCC No.M 205114 for nitrile hydration and Delftia tsuruhatensis CCTCC No.M 205115 for the optical selective hydrolysis of the amide.
Q: Is this process suitable for large-scale commercial production of cilastatin intermediates?
A: Yes, the patent demonstrates high yields (>47% overall for the chiral product) and uses standard fermentation and biotransformation equipment, making it highly scalable for industrial API intermediate manufacturing without the need for expensive chiral columns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(+)-2,2-Dimethylcyclopropylcarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in modern pharmaceutical synthesis and are committed to delivering these advanced solutions to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral analysis tools to guarantee that every batch meets the highest standards of optical purity and chemical identity. Our dedication to quality assurance means that we can consistently supply S-(+)-2,2-dimethylcyclopropylcarboxamide that supports the efficient manufacture of Cilastatin and related therapeutics. By partnering with us, you gain access to a supply chain that prioritizes innovation, reliability, and regulatory compliance at every step of the value chain.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and production timelines. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how this biocatalytic method can optimize your manufacturing economics. Let us collaborate to bring high-quality, sustainably produced pharmaceutical intermediates to the market faster and more efficiently than ever before.
