Revolutionizing Fluorinated Intermediate Production: A Deep Dive into One-Pot Alpha-Fluoroacetophenone Synthesis
Revolutionizing Fluorinated Intermediate Production: A Deep Dive into One-Pot Alpha-Fluoroacetophenone Synthesis
The landscape of fluorinated chemical manufacturing is undergoing a significant transformation driven by the urgent need for safer, more cost-effective, and scalable synthetic routes. Patent CN101665394B introduces a groundbreaking methodology for the direct preparation of alpha-fluoroacetophenones from acetophenone derivatives using a novel one-pot strategy. This technology leverages the unique properties of low-melting eutectic mixtures to facilitate sequential alpha-halogenation and nucleophilic fluorination without the isolation of intermediates. For global pharmaceutical and agrochemical companies, this represents a pivotal shift away from hazardous electrophilic fluorination towards a robust, industrially viable process that utilizes readily available raw materials. The ability to synthesize these critical fluorinated building blocks efficiently addresses long-standing challenges in supply chain stability and production costs associated with fluorine chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-fluoroacetophenones has been plagued by significant technical and economic hurdles that hinder large-scale adoption. The first conventional pathway involves direct electrophilic fluorination, which traditionally relies on elemental fluorine gas, a substance that is highly toxic, corrosive, and extremely difficult to control safely in a standard manufacturing environment. Alternatively, modern laboratories utilize specialized N-F or O-F reagents like Selectfluor or trifluoroacetyl hypofluorite, but these chemicals are prohibitively expensive due to their complex synthesis from fluorine gas, making them unsuitable for ton-scale production. The second conventional pathway involves a two-step sequence where acetophenone is first halogenated to an alpha-halo ketone, isolated, and then subjected to nucleophilic fluorination. This multi-step approach not only increases operational complexity and solvent consumption but also exposes workers to lachrymatory and unstable alpha-halo intermediates during isolation and purification steps. Furthermore, the nucleophilic substitution step in traditional solvents often suffers from low reactivity, requiring harsh conditions or expensive phase transfer catalysts like crown ethers to achieve acceptable conversion rates.
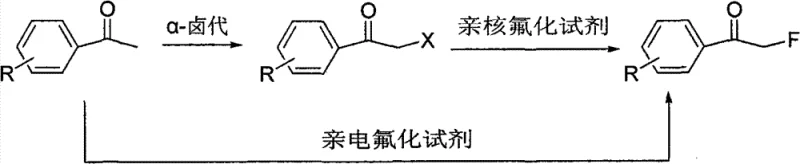
The Novel Approach
The innovative method disclosed in CN101665394B elegantly bypasses these limitations by integrating both reaction steps into a single pot using a tailored eutectic solvent system. By employing a mixture of a quaternary ammonium salt and an acidic substance, the process creates a liquid reaction medium with a melting point significantly lower than its individual components, allowing reactions to proceed under mild thermal conditions. This eutectic environment activates the acetophenone substrate for efficient alpha-halogenation using inexpensive reagents like 1,3-dichloro-5,5-dimethylhydantoin (DCDMH) or N-chlorosuccinimide (NCS). Crucially, without isolating the resulting alpha-halo intermediate, a nucleophilic fluoride source such as potassium fluoride (KF) or cesium fluoride (CsF) is added directly to the same vessel. The eutectic medium enhances the nucleophilicity of the fluoride ion, facilitating the displacement of the halogen atom to form the carbon-fluorine bond with high efficiency. This seamless integration eliminates the need for dangerous electrophilic fluorine sources and avoids the logistical burden of intermediate isolation, thereby streamlining the entire manufacturing workflow.
Mechanistic Insights into Eutectic-Mediated Sequential Fluorination
The core mechanism of this transformation relies on the dual functionality of the eutectic mixture, which acts simultaneously as a solvent and a catalytic promoter for both halogenation and fluorination steps. In the initial phase, the acidic component of the eutectic, such as p-toluenesulfonic acid or zinc chloride, likely assists in the enolization of the acetophenone carbonyl group, making the alpha-position more susceptible to electrophilic attack by the halogenating agent. This generates the alpha-haloacetophenone species in situ within the ionic liquid-like environment of the molten salt mixture. The unique solvation properties of the eutectic stabilize this reactive intermediate, preventing decomposition or side reactions that might occur in volatile organic solvents. Upon the addition of the metal fluoride, the quaternary ammonium cations in the eutectic may interact with the fluoride anions, effectively reducing the lattice energy of the salt and increasing the availability of naked fluoride ions for nucleophilic attack. This activation allows even less reactive fluorides like KF to participate effectively in the substitution reaction at moderate temperatures ranging from 50°C to 150°C, ensuring high conversion rates without the need for extreme thermal stress.

From an impurity control perspective, this one-pot strategy offers distinct advantages over traditional multi-step syntheses by minimizing the exposure of reactive intermediates to external contaminants. In conventional processes, the isolation of alpha-halo ketones often leads to degradation or polymerization, generating complex impurity profiles that are difficult to remove in subsequent steps. By keeping the intermediate dissolved in the eutectic matrix, the process limits its contact with moisture or air, thereby suppressing hydrolysis or oxidation side reactions. Furthermore, the use of specific halogenating agents like DCDMH provides a controlled release of halogen, reducing the risk of over-halogenation or poly-halogenated byproducts which are common when using molecular halogens. The final workup involves simple extraction and washing procedures, which effectively separate the organic product from the inorganic salts and eutectic components, resulting in a crude product that is amenable to straightforward purification. This inherent cleanliness of the reaction profile ensures that the final alpha-fluoroacetophenone meets stringent purity specifications required for pharmaceutical applications with minimal downstream processing.
How to Synthesize Alpha-Fluoroacetophenone Efficiently
Implementing this patented technology requires careful attention to the preparation of the eutectic solvent and the sequential addition of reagents to maximize yield and safety. The process begins with the formation of the eutectic mixture by heating a quaternary ammonium salt, such as choline chloride or tetrabutylammonium bromide, with an acidic substance like zinc chloride or p-toluenesulfonic acid until a clear, homogeneous liquid is obtained. Once the eutectic is prepared and cooled to the appropriate temperature range of 10°C to 100°C, the acetophenone substrate and the alpha-halogenating reagent are introduced to initiate the halogenation phase, which typically proceeds for 3 to 5 hours. Following the completion of halogenation, the nucleophilic fluorinating agent is added in batches to manage exotherms, and the temperature is raised to between 50°C and 150°C to drive the fluorination to completion over a period of 5 to 10 hours. Detailed standardized operating procedures regarding specific molar ratios, stirring speeds, and workup protocols are essential for reproducible results and are outlined in the comprehensive guide below.
- Prepare a low-melting eutectic mixture by heating a quaternary ammonium salt (e.g., choline chloride) with an acidic substance (e.g., p-toluenesulfonic acid or zinc chloride) until a homogeneous liquid forms.
- Add the acetophenone substrate and an alpha-halogenating reagent (such as DCDMH or NCS) to the eutectic at 10-100°C and react for 3-5 hours to generate the alpha-halo intermediate in situ.
- Introduce a nucleophilic fluorinating reagent (like KF or CsF) directly into the reaction mixture and heat to 50-150°C for 5-10 hours to complete the fluorination without isolating the intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this eutectic-based one-pot synthesis offers transformative benefits that directly impact the bottom line and operational resilience. The primary driver for cost reduction lies in the substitution of expensive, specialized fluorinating reagents with commodity chemicals that are available in bulk quantities globally. By eliminating the reliance on electrophilic fluorine sources like Selectfluor or elemental fluorine, manufacturers can drastically reduce raw material expenditures while simultaneously removing the need for specialized containment equipment required for handling hazardous gases. Additionally, the consolidation of two reaction steps into a single vessel reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours associated with intermediate isolation and purification. These efficiencies compound to create a significantly leaner manufacturing process that lowers the overall cost of goods sold for fluorinated intermediates, providing a competitive edge in price-sensitive markets.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior because it replaces high-cost reagents with low-cost alternatives like potassium fluoride and dichlorohydantoin, which are produced on a massive industrial scale. The elimination of intermediate isolation steps means that facilities save substantially on solvent purchase, recovery, and disposal costs, which are often a hidden but major expense in fine chemical synthesis. Furthermore, the reduced number of unit operations translates to lower utility consumption and decreased wear and tear on equipment, extending asset life and reducing maintenance overhead. This holistic reduction in operational expenditure allows for more aggressive pricing strategies or improved profit margins without compromising on product quality.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents required for this synthesis, such as choline chloride, acetophenones, and metal fluorides, are widely produced by multiple suppliers across different geographic regions. Unlike specialized fluorinating agents that may have limited supply chains or long lead times due to complex manufacturing requirements, these commodity chemicals are readily available in multi-ton quantities year-round. This diversification of the supply base mitigates the risk of production stoppages caused by raw material shortages or logistics disruptions, ensuring consistent delivery schedules to downstream customers. The robustness of the supply chain is further strengthened by the fact that the eutectic components are stable solids that can be stored for extended periods without degradation, simplifying inventory management.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of volatile organic solvents in the initial reaction phase make this process inherently safer and easier to scale from pilot plant to commercial production volumes. The use of a non-volatile eutectic medium minimizes emissions of volatile organic compounds (VOCs), aligning with increasingly stringent environmental regulations and sustainability goals of modern chemical enterprises. Waste generation is also minimized since the eutectic can potentially be recycled or treated more easily than complex organic solvent mixtures, reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only facilitates regulatory approval but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot fluorination technology, based on the detailed disclosures within the patent literature. Understanding these nuances is critical for R&D teams evaluating the feasibility of transferring this process to their own manufacturing lines. The answers provided reflect the specific capabilities and constraints identified in the experimental data, ensuring that expectations are managed accurately regarding yield, substrate scope, and operational parameters.
Q: Why is the eutectic-based one-pot method superior to traditional electrophilic fluorination?
A: Traditional electrophilic fluorination often requires hazardous elemental fluorine or expensive reagents like Selectfluor. The eutectic method utilizes cheap, stable nucleophilic fluorides (e.g., KF) and avoids the safety risks and high costs associated with handling elemental fluorine or specialized N-F reagents.
Q: Does this process require the isolation of the alpha-halo intermediate?
A: No, a key advantage of this patented process is that it is a true one-pot synthesis. The alpha-halogenation and subsequent nucleophilic fluorination occur sequentially in the same reactor within the eutectic medium, eliminating the need to isolate the potentially unstable or lachrymatory alpha-halo ketone intermediate.
Q: What types of substituents are tolerated on the acetophenone ring?
A: The method demonstrates broad substrate scope, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as nitro, cyano, trifluoromethyl, and halogens (chloro, bromo, fluoro) at various positions on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Fluoroacetophenone Supplier
As the demand for fluorinated building blocks continues to surge across the pharmaceutical and agrochemical sectors, partnering with a manufacturer that possesses deep technical expertise in advanced synthesis methods is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge technologies like the eutectic-mediated one-pot synthesis to deliver high-quality intermediates with unmatched consistency. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercial launch. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of alpha-fluoroacetophenone meets the exacting standards required for drug substance manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing method for your supply chain. We encourage you to contact us today to obtain specific COA data for our current inventory and to receive detailed route feasibility assessments that demonstrate our commitment to being your strategic partner in fluorine chemistry.
