Advanced Copper-Catalyzed Synthesis of Perfluoroalkyl Sulfenates for Scalable Pharmaceutical Intermediate Production
Introduction to Next-Generation Fluorinated Intermediate Synthesis
The strategic incorporation of fluorine atoms into organic molecules remains a cornerstone of modern medicinal chemistry and agrochemical design, fundamentally altering metabolic stability and lipophilicity profiles. Patent CN109020847B introduces a transformative methodology for the preparation of perfluoroalkyl sulfenates, a critical class of electrophilic and nucleophilic reagents often utilized as versatile building blocks in complex molecule assembly. This innovation leverages a copper-catalyzed oxidative coupling strategy that directly utilizes alpha-carbonyl diazo compounds and commercially available sodium perfluoroalkyl sulfinates as reaction substrates. By employing inexpensive anhydrous copper acetate as the optimal catalyst and tert-butyl hydroperoxide (TBHP) as a green oxidant, this process achieves high reaction yields under remarkably mild conditions. The significance of this technical breakthrough lies in its ability to bypass the cumbersome pre-activation steps typically required for sulfinate salts, thereby streamlining the synthetic workflow for reliable pharmaceutical intermediate supplier operations globally.
Furthermore, the environmental footprint of this synthesis is drastically minimized compared to legacy protocols, as the oxidation byproducts are merely tert-butanol and water, avoiding the generation of toxic halide salts. This aligns perfectly with contemporary green chemistry mandates while ensuring that the resulting high-purity OLED material or API precursors meet stringent regulatory standards. The reaction operates efficiently in common organic solvents such as ethyl acetate, facilitating straightforward downstream processing and solvent recovery. For procurement teams evaluating cost reduction in fine chemical manufacturing, this route offers a compelling alternative to precious metal-catalyzed processes, reducing both raw material expenditure and waste disposal costs associated with heavy metal removal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluoroalkyl sulfinates has been plagued by significant operational inefficiencies and environmental hazards that hinder scalable production. Traditional literature methods often rely on the activation of trifluoromethanesulfinate using aggressive reagents such as 2,4,6-trimethylbenzenesulfonyl chloride or phosphorus oxychloride to generate reactive sulfinyl chlorides in situ. These processes inevitably produce stoichiometric amounts of sulfonate or chloride by-products that contaminate the environment and complicate purification workflows. Additionally, alternative routes utilizing silver triflate or palladium catalysts suffer from narrow substrate scopes, excessively long reaction times extending up to 18 hours, and the formation of equivalent amounts of silver bromide or iodobenzene waste. Such reliance on expensive noble metals and harsh activating agents creates substantial bottlenecks in the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN109020847B revolutionizes this chemical transformation by enabling the direct coupling of unactivated sodium perfluoroalkyl sulfinate with diazo precursors. As illustrated in the representative reaction scheme below, the process utilizes a catalytic amount of copper acetate to facilitate the bond formation without requiring any pre-functionalization of the sulfinate salt. 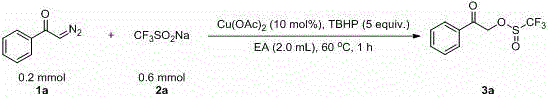 The reaction proceeds rapidly, typically completing within 1 hour at a moderate temperature of 60 °C, which represents a drastic improvement over the multi-day reflux conditions of prior art. This novel approach not only expands the accessible chemical space to include longer perfluoroalkyl chains such as perfluorobutyl and perfluorooctyl but also ensures that the reaction byproducts are environmentally benign. The elimination of halogenated activating agents means that the final product streams are free from corrosive halide salts, significantly reducing the burden on wastewater treatment systems and enhancing the overall sustainability profile of the manufacturing process.
The reaction proceeds rapidly, typically completing within 1 hour at a moderate temperature of 60 °C, which represents a drastic improvement over the multi-day reflux conditions of prior art. This novel approach not only expands the accessible chemical space to include longer perfluoroalkyl chains such as perfluorobutyl and perfluorooctyl but also ensures that the reaction byproducts are environmentally benign. The elimination of halogenated activating agents means that the final product streams are free from corrosive halide salts, significantly reducing the burden on wastewater treatment systems and enhancing the overall sustainability profile of the manufacturing process.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The mechanistic pathway of this transformation involves a sophisticated interplay between the transition metal catalyst and the peroxide oxidant to generate reactive radical species. The alpha-carbonyl diazo compound, characterized by the general structure shown below, serves as the carbon-centered radical precursor upon interaction with the copper catalyst. 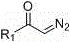 The copper(II) species likely facilitates the decomposition of the diazo functionality or activates the peroxide to generate a radical intermediate that subsequently attacks the sulfur center of the perfluoroalkyl sulfinate. This radical-mediated insertion mechanism allows for the construction of the carbon-sulfur bond under neutral conditions, preserving sensitive functional groups that might otherwise degrade under acidic or basic activation protocols. The use of TBHP as the terminal oxidant is crucial, as it regenerates the active copper species while being reduced to tert-butanol, a volatile alcohol that is easily removed during workup. This catalytic cycle ensures high turnover numbers and minimizes the loading of the transition metal, which is vital for meeting residual metal specifications in active pharmaceutical ingredients.
The copper(II) species likely facilitates the decomposition of the diazo functionality or activates the peroxide to generate a radical intermediate that subsequently attacks the sulfur center of the perfluoroalkyl sulfinate. This radical-mediated insertion mechanism allows for the construction of the carbon-sulfur bond under neutral conditions, preserving sensitive functional groups that might otherwise degrade under acidic or basic activation protocols. The use of TBHP as the terminal oxidant is crucial, as it regenerates the active copper species while being reduced to tert-butanol, a volatile alcohol that is easily removed during workup. This catalytic cycle ensures high turnover numbers and minimizes the loading of the transition metal, which is vital for meeting residual metal specifications in active pharmaceutical ingredients.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the formation of inorganic salts that are difficult to separate from organic products. In traditional methods involving silver or palladium, the removal of trace metal residues often requires specialized scavenging resins or multiple recrystallization steps, which erode overall yield. Here, the primary byproduct is nitrogen gas, which evolves from the reaction mixture, driving the equilibrium forward and leaving a clean organic phase. The broad tolerance for various substituents on the diazo component, including electron-donating methoxy groups and electron-withdrawing halogens, suggests a robust radical pathway that is insensitive to electronic effects. This mechanistic resilience ensures consistent product quality across a wide range of substrates, providing supply chain heads with the confidence needed for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Perfluoroalkyl Sulfenate Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, making it highly suitable for technology transfer from laboratory to pilot plant scales. The standard protocol involves charging the reaction vessel with the alpha-carbonyl diazo compound and three equivalents of sodium perfluoroalkyl sulfinate, followed by the addition of 10 mol% anhydrous copper acetate. Ethyl acetate is employed as the solvent of choice due to its favorable safety profile and ease of removal, while five equivalents of TBHP are added to drive the oxidative coupling to completion. The detailed standardized synthesis steps, including precise addition rates and quenching procedures, are outlined in the guide below to ensure operators can replicate the high yields reported in the patent data.
- Combine alpha-carbonyl diazo compound (0.2 mmol), sodium perfluoroalkyl sulfinate (0.6 mmol), and anhydrous copper acetate (10 mol%) in a reaction vessel.
- Add ethyl acetate (2.0 mL) as solvent and tert-butyl hydroperoxide (TBHP, 5 equivalents) as the oxidant to the mixture.
- Heat the reaction system to 60 °C under air atmosphere for 1 hour, then purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The shift from precious metal catalysts like palladium or silver to abundant and inexpensive copper acetate results in a substantial reduction in raw material costs, eliminating the volatility associated with noble metal pricing. Furthermore, the avoidance of pre-activation reagents such as sulfonyl chlorides removes an entire unit operation from the manufacturing process, thereby reducing labor hours, energy consumption, and reactor occupancy time. This streamlined workflow translates into faster batch cycles and increased throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on product quality or purity specifications.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the replacement of expensive catalysts and the elimination of stoichiometric activating agents. By utilizing cheap copper salts and avoiding the purchase of specialized chlorinating or phosphorylating reagents, the overall cost of goods sold is significantly lowered. Additionally, the simplified workup procedure, which involves direct dilution and column chromatography without complex aqueous extractions to remove inorganic salts, reduces solvent usage and waste treatment expenses. These cumulative efficiencies create a leaner manufacturing model that enhances competitiveness in the global fine chemical market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a robust supply chain that is less susceptible to disruptions. Sodium perfluoroalkyl sulfinates and alpha-carbonyl diazo compounds are readily sourced from multiple vendors, mitigating the risk of single-source dependency often associated with exotic catalysts. The ability to run the reaction under air atmosphere further simplifies facility requirements, removing the need for complex inert gas manifolds or glovebox operations. This operational simplicity facilitates easier technology transfer to contract manufacturing organizations, ensuring consistent supply continuity for critical downstream applications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a superior profile by generating benign byproducts like tert-butanol and water instead of toxic halide wastes. The absence of heavy metal contamination in the final product simplifies regulatory compliance for pharmaceutical applications, where strict limits on residual metals are enforced. The mild reaction conditions of 60 °C reduce energy demands and thermal risks, making the process inherently safer for large-scale production. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the corporate sustainability image of the producing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and disclosure sections of the patent to provide accurate guidance for R&D teams evaluating this technology for their specific projects. Understanding these nuances is essential for optimizing reaction parameters and anticipating potential challenges during scale-up activities.
Q: What are the primary advantages of this copper-catalyzed method over traditional sulfinate synthesis?
A: This method eliminates the need for pre-activation of sodium perfluoroalkyl sulfinate, avoids harsh conditions and expensive noble metal catalysts like palladium or silver, and significantly reduces halide waste generation.
Q: Is the reaction sensitive to air or moisture during the oxidative coupling process?
A: The reaction is robust and can be carried out directly under air conditions without the need for inert gas protection, simplifying the operational requirements for large-scale manufacturing.
Q: What is the substrate scope for the perfluoroalkyl chain length in this synthesis?
A: The methodology demonstrates broad universality, successfully accommodating perfluoroalkyl chains ranging from trifluoromethyl (n=1) to perfluorooctyl (n=8), allowing for diverse fluorinated intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoroalkyl Sulfenate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fluorination technologies play in accelerating drug discovery and development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the copper-catalyzed sulfinate synthesis can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of perfluoroalkyl sulfenate meets the exacting standards required by top-tier pharmaceutical and agrochemical companies. Our commitment to quality assurance ensures that clients receive materials that are ready for immediate use in sensitive downstream transformations without the need for additional purification.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective synthesis route can be integrated into your existing supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a collaborative approach to achieving your manufacturing goals with efficiency and precision.
