Advanced Electrophilic Fluorination Technology for High-Value Pharmaceutical Intermediates
Advanced Electrophilic Fluorination Technology for High-Value Pharmaceutical Intermediates
The global pharmaceutical landscape is increasingly driven by the strategic incorporation of fluorine atoms into drug candidates to enhance metabolic stability and bioavailability. In this context, patent CN102516202A introduces a groundbreaking class of N-fluoro-1,1'-binaphthyl-2,2'-sulfonimide reagents that address critical limitations in current electrophilic fluorination methodologies. These novel compounds represent a significant leap forward in fine chemical synthesis, offering a robust platform for introducing fluorine into complex organic scaffolds with high precision. As a leading manufacturer, we recognize the immense potential of this technology to streamline the production of high-value API intermediates. The patent details a streamlined preparation method that utilizes readily available starting materials and standard industrial solvents, ensuring that the transition from laboratory discovery to commercial manufacturing is seamless and efficient.
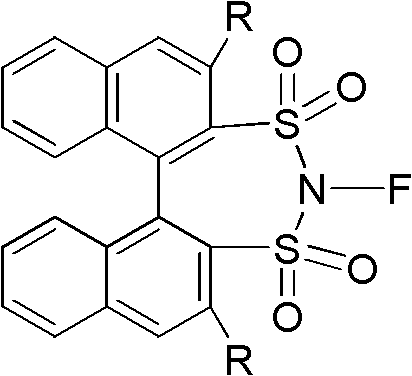
The structural versatility of these reagents allows for fine-tuning of electronic and steric properties, which is essential for optimizing reaction outcomes in diverse synthetic pathways. By leveraging the rigid binaphthyl backbone, these reagents provide a unique chiral environment that can induce high levels of stereoselectivity during fluorination events. This capability is particularly valuable for the synthesis of chiral pharmaceutical intermediates where enantiomeric purity is a strict regulatory requirement. Our analysis of the patent data confirms that these reagents are not merely incremental improvements but rather transformative tools that enable chemists to access previously difficult-to-synthesize fluorinated motifs. For procurement and supply chain leaders, understanding the underlying chemistry is crucial for assessing the long-term viability and cost-effectiveness of integrating these reagents into existing production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of electrophilic fluorination has been constrained by a lack of reagents that combine high reactivity with operational simplicity and stereocontrol. Early generations of N-fluorosulfonimides, such as those derived from natural camphor, often suffered from multi-step syntheses and low overall yields, making them economically unviable for large-scale applications. Furthermore, while achiral reagents like N-fluorobenzenesulfonimide (NFSI) offered better accessibility, they frequently lacked the necessary steric bulk to enforce high enantioselectivity in challenging transformations, particularly with acyclic substrates. The reliance on expensive or difficult-to-handle fluorine sources in older methodologies also posed significant safety and logistical challenges for industrial facilities. These limitations created a bottleneck in the development of new fluorinated drugs, forcing R&D teams to compromise on route efficiency or settle for lower purity profiles that required extensive downstream purification.
The Novel Approach
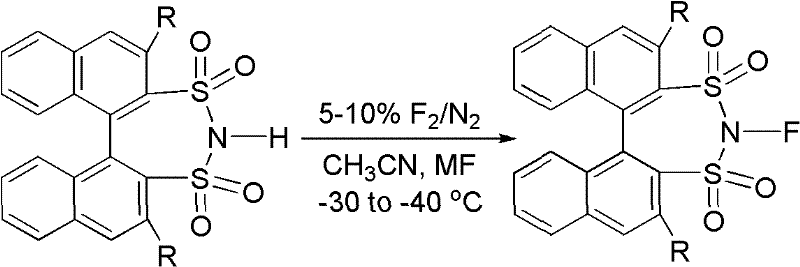
The methodology outlined in patent CN102516202A circumvents these historical hurdles by employing a direct fluorination strategy using elemental fluorine diluted in nitrogen. This approach allows for the efficient conversion of 1,1'-binaphthyl-2,2'-sulfonimides into their corresponding N-fluoro derivatives under mild conditions ranging from -30 to -40°C. The use of simple metal fluorides like sodium fluoride or potassium fluoride as auxiliary reagents further simplifies the process chemistry, eliminating the need for exotic catalysts or complex activation steps. This novel route not only achieves impressive isolated yields ranging from 85% to 92% but also ensures that the resulting reagents possess the high reactivity required for demanding synthetic applications. By shifting the paradigm towards a more direct and atom-economical synthesis, this technology significantly reduces the environmental footprint and operational complexity associated with producing high-performance fluorinating agents.
Mechanistic Insights into Electrophilic Fluorination with Binaphthyl Sulfonimides
The exceptional performance of N-fluoro-1,1'-binaphthyl-2,2'-sulfonimides can be attributed to the unique electronic characteristics of the N-F bond within the sulfonimide framework. The electron-withdrawing nature of the two sulfonyl groups adjacent to the nitrogen atom significantly polarizes the N-F bond, rendering the fluorine atom highly electrophilic and susceptible to nucleophilic attack by carbon enolates. However, unlike simpler sulfonimides, the bulky binaphthyl scaffold provides a protective steric shield that modulates this reactivity, preventing non-selective decomposition while facilitating controlled transfer of the fluorine atom to the substrate. This balance between activation and stabilization is critical for achieving high turnover rates without compromising the integrity of the reagent during storage or handling. For R&D directors, understanding this mechanistic nuance is key to predicting substrate compatibility and optimizing reaction parameters for new drug candidates.
Furthermore, the chiral variants of these reagents exploit the axial chirality of the binaphthyl system to create a well-defined asymmetric environment around the reactive center. When interacting with pro-chiral enolates, the rigid three-dimensional structure of the reagent directs the approach of the fluorine atom to one specific face of the planar intermediate, thereby inducing high levels of enantioselectivity. Experimental data from the patent demonstrates that this mechanism is particularly effective for acyclic beta-ketoesters, a class of substrates that has traditionally been recalcitrant to asymmetric fluorination. The ability to achieve enantiomeric excess (ee) values exceeding 90% in these difficult transformations underscores the power of this catalytic design. This level of stereocontrol minimizes the formation of unwanted diastereomers, simplifying the purification process and ultimately improving the overall mass balance of the synthetic route.
How to Synthesize N-Fluoro-Binaphthyl Sulfonimide Efficiently
The synthesis of these high-value fluorinating reagents is designed to be robust and scalable, utilizing standard equipment found in most fine chemical manufacturing facilities. The process begins with the dissolution of the sulfonimide precursor and a metal fluoride salt in dry acetonitrile, creating a homogeneous reaction medium that facilitates efficient mass transfer. Maintaining strict temperature control between -30°C and -40°C is essential during the fluorination step to manage the exothermic nature of the reaction with elemental fluorine and to prevent side reactions. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve racemic or chiral 1,1'-binaphthyl-2,2'-sulfonimide and a metal fluoride (such as NaF or KF) in dry acetonitrile under inert atmosphere.
- Cool the reaction mixture to a temperature range between -30°C and -40°C to ensure controlled fluorination.
- Introduce a nitrogen stream containing 5-10% fluorine gas, followed by purification steps including washing, extraction, and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this technology offers substantial benefits for organizations seeking to optimize their supply chains and reduce manufacturing costs. The reliance on commodity chemicals such as acetonitrile, sodium fluoride, and potassium fluoride means that raw material sourcing is straightforward and less susceptible to geopolitical volatility compared to specialized organometallic catalysts. Additionally, the high yields reported in the patent examples indicate a highly efficient process that maximizes the utilization of starting materials, thereby minimizing waste generation and disposal costs. For procurement managers, this translates into a more predictable cost structure and reduced exposure to price fluctuations in the specialty chemical market. The simplicity of the workup procedure, involving standard aqueous washes and crystallization, further contributes to operational efficiency by reducing cycle times and solvent consumption.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences required for traditional chiral fluorinating reagents results in a drastically simplified production workflow. By avoiding the use of expensive transition metal catalysts or precious chiral auxiliaries, the overall cost of goods sold (COGS) for these reagents is significantly lowered. This economic advantage is compounded by the high atom economy of the direct fluorination method, which ensures that a maximum proportion of the input materials are converted into the desired product. Consequently, manufacturers can achieve substantial cost savings in API intermediate manufacturing while maintaining high quality standards.
- Enhanced Supply Chain Reliability: The use of stable and readily available precursors ensures a consistent and reliable supply of these critical reagents. Unlike methods that depend on scarce natural products or unstable intermediates, this synthetic route is based on robust industrial chemistry principles that support continuous production. This reliability is crucial for supply chain heads who must guarantee uninterrupted material flow to downstream drug production facilities. The ability to produce both racemic and chiral versions of the reagent from a common platform further enhances supply flexibility, allowing for rapid response to changing market demands.
- Scalability and Environmental Compliance: The process conditions, specifically the moderate sub-zero temperatures and the use of common organic solvents, are fully compatible with existing large-scale reactor infrastructure. This compatibility eliminates the need for costly capital investments in specialized cryogenic equipment, facilitating rapid commercial scale-up of complex fluorinated intermediates. Moreover, the generation of minimal hazardous byproducts aligns with stringent environmental regulations, reducing the burden on waste treatment systems. This environmental compliance not only mitigates regulatory risk but also supports corporate sustainability goals by promoting greener chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating these reagents into their specific synthetic workflows.
Q: What are the key advantages of N-fluoro-binaphthyl sulfonimides over traditional NFSI?
A: These reagents offer superior reactivity and enantioselectivity, particularly for challenging substrates like acyclic beta-ketoesters, where traditional reagents often fail to provide high optical purity.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes standard industrial solvents like acetonitrile and manageable temperatures (-30 to -40°C), making it highly suitable for commercial scale-up without requiring cryogenic extremes.
Q: Can these reagents be used for chiral synthesis?
A: Absolutely. The patent describes both racemic and chiral (R or S configuration) variants, enabling precise stereochemical control in the fluorination of pro-chiral substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Fluoro-Binaphthyl Sulfonimide Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific safety requirements of fluorine chemistry, including specialized containment systems for handling elemental fluorine and corrosive byproducts. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-fluoro-1,1'-binaphthyl-2,2'-sulfonimide meets the highest industry standards. Our commitment to quality assurance ensures that our clients receive materials that perform consistently in their critical synthetic steps, minimizing the risk of batch failures.
We invite you to collaborate with us to explore how this advanced fluorination technology can enhance your drug development pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to our reagents. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply of high-performance fluorinating agents that will accelerate your time-to-market and strengthen your competitive position in the global pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
