Advanced Biocatalytic Route for High-Purity (S)-(+)-DMCPA: Scalable Manufacturing Insights
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for synthesizing critical chiral intermediates. A significant advancement in this domain is detailed in patent CN102757924A, which discloses a novel strain of Rhodococcus sp., designated as ECU 1013 (CGMCC No.5911), specifically engineered for the preparation of (S)-(+)-2,2-dimethylcyclopropanecarboxylic acid, commonly abbreviated as (S)-(+)-DMCPA. This compound serves as a pivotal chiral building block in the synthesis of Cilastatin, a renal dehydrodipeptidase inhibitor that is co-formulated with Imipenem to prevent antibiotic degradation in the kidney. The patent outlines a comprehensive biocatalytic process that leverages the unique esterase activity of this specific bacterial strain to perform enantioselective hydrolysis with exceptional precision. By utilizing whole cells or crude enzyme preparations derived from ECU 1013, manufacturers can achieve optical purities exceeding 98% ee, addressing the rigorous quality demands of modern API production while mitigating the environmental hazards associated with traditional synthetic routes.
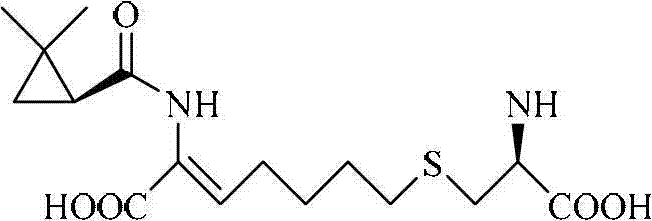
The structural complexity of Cilastatin necessitates high-fidelity intermediates to ensure therapeutic efficacy and safety. As illustrated in the molecular architecture, the (S)-(+)-2,2-dimethylcyclopropane moiety is integral to the drug's function as a specific enzyme inhibitor. Conventional supply chains for such intermediates often struggle with consistency and cost-efficiency, making the discovery of highly selective biocatalysts like ECU 1013 a transformative development for reliable pharmaceutical intermediate suppliers. The technology described offers a direct route to bypass the limitations of racemic synthesis, providing a streamlined pathway that aligns with green chemistry principles and reduces the reliance on hazardous reagents typically found in older methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure (S)-(+)-DMCPA has relied heavily on chemical resolution techniques or asymmetric synthesis strategies that present significant operational and economic challenges. Traditional chemical resolution often involves the use of optically active chiral reagents to form diastereomeric salts or esters, which are then separated via fractional crystallization. This approach is inherently inefficient, typically resulting in single-step yields ranging merely between 20% and 30%, thereby generating substantial material waste and increasing the cost of goods sold. Furthermore, many established protocols require the use of highly toxic cyanide compounds, particularly when starting from nitrile precursors, posing severe environmental risks and necessitating complex waste treatment infrastructure. The reliance on expensive chiral auxiliaries that are difficult to recover and recycle further exacerbates the economic burden, rendering these methods less attractive for large-scale commercial manufacturing where margin compression is a constant concern for procurement teams.
The Novel Approach
In stark contrast, the biocatalytic methodology presented in the patent utilizes the intrinsic stereoselectivity of the Rhodococcus sp. ECU 1013 esterase to discriminate between enantiomers of racemic 2,2-dimethylcyclopropane carboxylates. This biological resolution operates under mild reaction conditions, typically around 30°C to 35°C and near-neutral pH, eliminating the need for extreme temperatures or corrosive acids and bases. The process achieves a single-resolution yield exceeding 40%, which is theoretically close to the maximum 50% yield possible in a kinetic resolution of a racemate, indicating highly efficient substrate utilization. By employing whole cells or lyophilized enzyme powder, the technology simplifies the catalyst handling process and enhances stability, allowing for potential reuse or continuous processing setups. This shift from harsh chemical environments to benign aqueous or biphasic systems represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, offering a cleaner, safer, and more economically viable alternative for industrial adoption.
Mechanistic Insights into Rhodococcus-Mediated Enantioselective Hydrolysis
The core of this technological breakthrough lies in the specific catalytic mechanism of the esterase produced by the Rhodococcus sp. ECU 1013 strain. The enzyme functions through a kinetic resolution mechanism wherein it preferentially recognizes and hydrolyzes one enantiomer of the racemic ester substrate—specifically the (S)-enantiomer in this context—while leaving the (R)-enantiomer largely untouched or reacting with it at a negligible rate. This discrimination is governed by the precise three-dimensional arrangement of amino acid residues within the enzyme's active site, which creates a chiral environment complementary only to the transition state of the desired (S)-isomer. The patent data indicates an enantioselectivity factor (E-value) greater than 100, signifying an extremely high preference for the target stereoisomer. Such high selectivity is crucial for minimizing the formation of unwanted byproducts and simplifying downstream purification, as the unreacted (R)-ester can often be easily separated from the acidic (S)-product through simple pH adjustments and extraction.
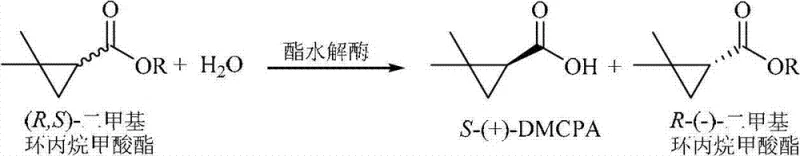
Furthermore, the impurity profile of the final product is tightly controlled by the specificity of the biocatalyst. Unlike chemical catalysts that might promote side reactions such as ring-opening of the cyclopropane moiety or non-specific hydrolysis of other functional groups, the biological catalyst exhibits remarkable chemoselectivity. The process tolerates various ester groups, including methyl, ethyl, and chloroethyl esters, allowing flexibility in raw material sourcing without compromising product quality. The reaction proceeds efficiently in both single aqueous phases and water-organic solvent biphasic systems, with the latter being particularly advantageous for substrates with low water solubility. The use of organic solvents like isooctane in a biphasic system helps to solubilize the hydrophobic ester substrate while maintaining the enzyme's activity in the aqueous phase, thereby enhancing the overall reaction rate and conversion efficiency without denaturing the biocatalyst.
How to Synthesize (S)-(+)-2,2-Dimethylcyclopropanecarboxylic Acid Efficiently
The implementation of this biocatalytic route involves a streamlined sequence of fermentation, catalyst preparation, and bioconversion steps that are amenable to standard pharmaceutical manufacturing equipment. The process begins with the cultivation of the Rhodococcus sp. ECU 1013 strain in a nutrient-rich fermentation medium, followed by the harvest of biomass which serves directly as the biocatalyst or is processed into a stable enzyme powder. The subsequent hydrolysis reaction is conducted under controlled pH and temperature conditions to maximize enantioselectivity and yield. For a detailed breakdown of the standardized operating procedures, including specific media compositions and reaction parameters, please refer to the technical guide below.
- Ferment Rhodococcus sp. ECU 1013 (CGMCC No.5911) in a rich medium containing glucose and yeast extract at 30°C to generate whole-cell biomass or crude enzyme powder.
- Prepare the biocatalyst by harvesting wet cells via centrifugation or disrupting cells to obtain crude esterase solution, which is then freeze-dried for stability.
- Conduct enantioselective hydrolysis of 2,2-dimethylcyclopropane carboxylate esters in a buffered aqueous or water-isooctane biphasic system at pH 7.0-7.5 and 30-35°C.
- Isolate the target (S)-(+)-DMCPA by adjusting pH to alkaline for extraction, followed by acidification and organic solvent extraction to achieve >98% ee purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology offers compelling strategic advantages that extend beyond mere technical performance. The elimination of toxic cyanide reagents and expensive chiral resolving agents translates directly into significant cost reduction in manufacturing operations by lowering raw material expenses and reducing the regulatory burden associated with hazardous waste disposal. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and aligning with corporate sustainability goals. Moreover, the robustness of the Rhodococcus strain ensures consistent supply continuity, as the fermentation process can be scaled up reliably to meet fluctuating market demands without the volatility associated with sourcing rare chemical catalysts.
- Cost Reduction in Manufacturing: The transition from chemical resolution to enzymatic hydrolysis removes the necessity for costly chiral auxiliaries and reduces the number of synthetic steps required to achieve high optical purity. By avoiding the use of heavy metals or toxic cyanides, the process eliminates the need for expensive metal scavenging units and complex effluent treatment plants, leading to substantial operational expenditure savings. The high single-pass yield of over 40% maximizes raw material efficiency, ensuring that a greater proportion of the input substrate is converted into valuable product rather than waste, thereby optimizing the overall cost structure of the supply chain.
- Enhanced Supply Chain Reliability: The reliance on a fermentable microbial strain provides a secure and renewable source of catalytic activity, insulating the supply chain from the geopolitical and logistical risks often associated with the sourcing of specialized chemical reagents. The ability to produce the catalyst in-house or through trusted fermentation partners ensures a steady availability of high-performance biocatalysts, reducing lead time for high-purity pharmaceutical intermediates. Additionally, the stability of the freeze-dried enzyme powder allows for convenient storage and transportation, facilitating just-in-time manufacturing strategies and minimizing inventory holding costs.
- Scalability and Environmental Compliance: The process is designed for seamless commercial scale-up of complex pharmaceutical intermediates, having been validated from shake-flask levels to fermenter scales with consistent performance metrics. The use of aqueous buffers and benign organic solvents like isooctane ensures compliance with stringent environmental regulations, simplifying the permitting process for new production facilities. The reduced generation of hazardous byproducts minimizes the environmental impact, making this technology an ideal choice for companies aiming to enhance their green chemistry credentials while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of the Rhodococcus sp. ECU 1013 technology for (S)-(+)-DMCPA production. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs.
Q: What is the optical purity achievable with the Rhodococcus sp. ECU 1013 catalyst?
A: According to patent CN102757924A, the process consistently achieves an optical purity (ee value) greater than 98% for the target (S)-(+)-DMCPA product, meeting stringent pharmaceutical standards.
Q: Which substrates are compatible with this enzymatic hydrolysis method?
A: The Rhodococcus sp. ECU 1013 esterase demonstrates high activity and selectivity towards various 2,2-dimethylcyclopropane carboxylates, including methyl, ethyl, and chloroethyl esters, with methyl ester showing particularly high enantioselectivity.
Q: How does this biocatalytic route compare to traditional chemical resolution?
A: Unlike chemical resolution which often requires toxic cyanides or expensive chiral reagents and yields only 20-30%, this biological method operates under mild conditions, avoids toxic reagents, and achieves single-resolution yields exceeding 40% with superior environmental safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-(+)-DMCPA Supplier
The biocatalytic synthesis of (S)-(+)-2,2-dimethylcyclopropanecarboxylic acid represents a mature and highly efficient technology that is ready for industrial deployment. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the >98% ee benchmark required for high-quality Cilastatin intermediates. We understand the critical nature of chiral purity in antibiotic formulations and are committed to delivering products that meet the highest global regulatory standards.
We invite you to engage with our technical procurement team to discuss how this advanced biocatalytic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the compatibility of this technology with your existing production frameworks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
