Scalable Exemestane Manufacturing via Recyclable IBX Oxidation Technology
Introduction to Advanced Steroid Dehydrogenation Technology
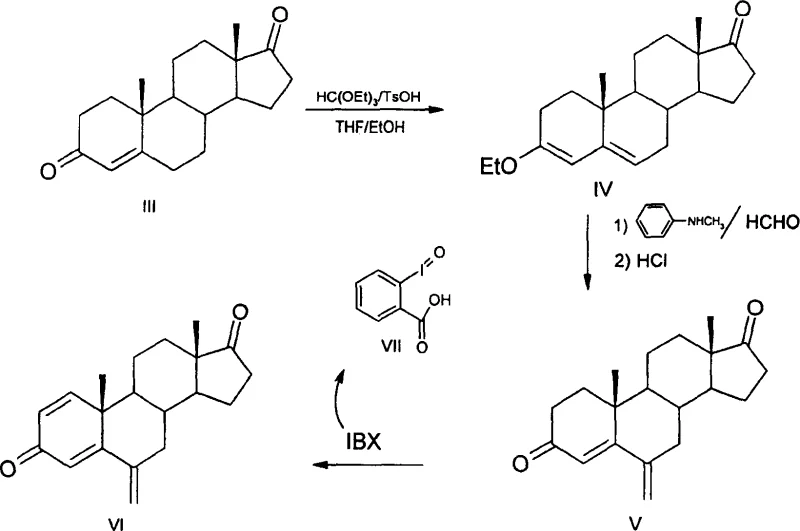
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology therapeutics, and the production of Exemestane, a potent third-generation aromatase inhibitor, remains a focal point for process optimization. Patent CN1304413C introduces a transformative methodology that addresses the longstanding economic and environmental inefficiencies associated with traditional steroid dehydrogenation. By replacing hazardous and costly reagents with a recyclable hypervalent iodine species, this technology offers a streamlined route from Androst-4-ene-3,17-dione to the final active pharmaceutical ingredient. The strategic implementation of 2-iodoxybenzoic acid (IBX) not only enhances reaction selectivity but also fundamentally alters the downstream processing requirements, eliminating the need for resource-intensive chromatographic separations. For global supply chain stakeholders, this represents a significant opportunity to secure a more stable and cost-efficient source of high-purity steroid intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Exemestane has relied heavily on 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) as the primary dehydrogenation agent, a protocol established in earlier patents such as US 4,904,650. While chemically effective, the DDQ-mediated pathway suffers from severe drawbacks that hinder large-scale manufacturing efficiency and economic viability. The reaction typically yields a complex mixture of byproducts, necessitating rigorous purification via column chromatography to achieve pharmaceutical-grade purity, a step that is notoriously difficult to scale and results in substantial material loss. Furthermore, DDQ is an expensive reagent with significant toxicity profiles, imposing strict handling protocols and waste disposal burdens that inflate operational expenditures. In many reported instances, the final step yield using DDQ hovers around 40% without chromatography, dropping drastically if purification is attempted on a massive scale, rendering the process economically fragile for high-volume production demands.
The Novel Approach
The innovative process detailed in CN1304413C circumvents these bottlenecks by utilizing IBX as a superior oxidizing agent, facilitating a cleaner conversion of the 6-methylene-androst-4-ene-3,17-dione intermediate to Exemestane. This novel approach leverages the unique reactivity of hypervalent iodine to effect dehydrogenation under mild conditions, typically in mixed solvent systems like DMSO and toluene, without generating the complex impurity profiles associated with quinone-based oxidants. Crucially, the workup procedure is dramatically simplified; the reduced reagent precipitates as a solid that can be easily filtered off, allowing the product to be isolated through straightforward recrystallization rather than chromatography. This shift from chromatographic purification to crystallization is a pivotal advancement for process chemistry, as it directly translates to higher throughput, reduced solvent consumption, and a significantly improved overall yield profile suitable for commercial manufacturing environments.
Mechanistic Insights into IBX-Mediated Dehydrogenation
The core of this technological advancement lies in the mechanistic behavior of 2-iodoxybenzoic acid (IBX), a hypervalent iodine(V) reagent that acts as a two-electron oxidant. In the context of steroid synthesis, IBX facilitates the abstraction of hydrogen atoms from the saturated positions adjacent to the carbonyl groups, promoting the formation of the conjugated diene system essential for Exemestane's biological activity. The reaction proceeds through a cyclic transition state where the iodine center coordinates with the substrate, enabling a concerted elimination process that preserves the sensitive exocyclic methylene group at the C6 position. Unlike radical-based oxidations that might lead to indiscriminate bond cleavage, the IBX mechanism offers high chemoselectivity, ensuring that the ketone functionalities at C3 and C17 remain intact while the A-ring is aromatized. This precision is vital for maintaining the structural integrity of the steroid backbone, minimizing the formation of over-oxidized side products that complicate purification.
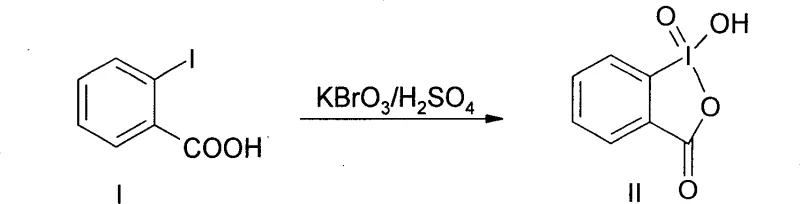
Furthermore, the sustainability of this mechanism is underpinned by the recyclability of the iodine species. Upon completion of the oxidation, IBX is reduced to 2-iodobenzoic acid (IBA), which, as shown in the reagent preparation scheme, can be quantitatively recovered from the reaction mixture. The patent data indicates that this reduced species can be re-oxidized back to the active IBX form using simple inorganic oxidants like potassium bromate in acidic media. This closed-loop cycle for the catalytic metal-free oxidant means that the stoichiometric consumption of iodine is effectively negated over multiple batches. From a mechanistic standpoint, this ensures that the reaction environment remains consistent batch-to-batch, reducing the variability often seen when fresh, potentially unstable reagents are introduced repeatedly. The ability to regenerate the active oxidant in situ or ex situ adds a layer of process robustness that is highly attractive for continuous manufacturing strategies.
How to Synthesize Exemestane Efficiently
The execution of this synthetic route requires precise control over reaction parameters to maximize the benefits of the IBX system. The process begins with the conversion of the starting diketone into an enol ether intermediate, which serves as a protected species to direct the subsequent methylenation reaction regioselectively. Following the introduction of the methylene group, the critical dehydrogenation step is performed in a biphasic or mixed solvent system to balance the solubility of the polar IBX reagent with the lipophilic steroid substrate. Maintaining an inert atmosphere is crucial during the heating phase to prevent side reactions, and the temperature must be carefully regulated to ensure complete conversion without degrading the sensitive exocyclic double bond. The detailed standardized synthesis steps for implementing this protocol in a pilot or production setting are outlined below.
- React Androst-4-ene-3,17-dione with triethyl orthoformate and TsOH catalyst to form the enol ether intermediate without isolation.
- Treat the crude enol ether with N-methylaniline and formaldehyde, followed by acid hydrolysis to yield 6-methylene-androst-4-ene-3,17-dione.
- Perform dehydrogenation using 2-iodoxybenzoic acid (IBX) in a mixed solvent system (e.g., DMSO/Toluene) under nitrogen protection to obtain Exemestane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this IBX-based synthesis route offers compelling economic arguments rooted in tangible process improvements rather than speculative projections. The elimination of column chromatography is perhaps the most significant cost driver, as chromatographic purification is one of the most expensive and time-consuming unit operations in fine chemical manufacturing. By replacing this with recrystallization, the process drastically reduces solvent usage, silica gel consumption, and labor hours, leading to a leaner cost structure. Additionally, the ability to recover and recycle the iodine reagent mitigates the volatility of raw material pricing, providing a hedge against market fluctuations for specialty oxidants. This creates a more predictable cost of goods sold (COGS), allowing for more stable long-term pricing agreements with downstream pharmaceutical partners.
- Cost Reduction in Manufacturing: The economic impact of switching from DDQ to recyclable IBX is profound, primarily driven by the reusability of the iodine backbone. Since the reduced byproduct can be recovered with high efficiency and re-oxidized, the effective consumption of the expensive iodine component is minimized over the lifecycle of the production campaign. Moreover, the avoidance of chromatographic purification removes a major bottleneck that typically limits batch size and increases facility occupancy time. This streamlining of the downstream process allows for larger batch sizes and faster turnover, effectively increasing the capacity of existing manufacturing assets without requiring capital investment in new equipment. The cumulative effect is a substantial reduction in variable manufacturing costs per kilogram of Exemestane produced.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the reagent system. DDQ is a specialized reagent with a limited supplier base and potential supply constraints, whereas the precursors for IBX, such as o-iodobenzoic acid and potassium bromate, are commodity chemicals available from multiple global sources. This diversification of the supply base reduces the risk of single-source dependency and ensures continuity of supply even during market disruptions. Furthermore, the simplified workup procedure reduces the complexity of waste management, as the process generates less hazardous waste compared to the cyanide-containing byproducts of DDQ degradation. This ease of waste handling accelerates the release of batches and reduces the administrative burden on logistics and compliance teams.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles and regulatory expectations. The replacement of toxic DDQ with the less hazardous IBX reduces the occupational exposure risks for plant personnel and lowers the toxicity profile of the effluent streams. The high atom economy achieved through reagent recycling minimizes the generation of solid waste, supporting corporate sustainability goals and reducing disposal costs. The scalability of the recrystallization purification method is inherently superior to chromatography, as crystallization kinetics are well-understood and easily modeled for large-scale reactors. This ensures that the quality and yield observed in the laboratory can be faithfully reproduced in multi-ton commercial production, de-risking the technology transfer process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this IBX-mediated synthesis for Exemestane. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical implications of adopting this technology. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using IBX over DDQ for Exemestane synthesis?
A: Unlike DDQ, which is expensive, toxic, and often requires cumbersome column chromatography, IBX is non-toxic, cost-effective, and allows for product purification via simple recrystallization. Furthermore, the reduced byproduct (o-iodobenzoic acid) can be recovered and recycled with high efficiency.
Q: Is column chromatography required for the final purification of Exemestane in this process?
A: No, column chromatography is not required. The patent specifies that the final product can be purified effectively through recrystallization methods, such as using a methylene dichloride-normal hexane solvent system, which significantly simplifies industrial scale-up.
Q: Can the oxidizing reagent be recovered in this synthetic route?
A: Yes, the reduced form of the reagent, o-iodobenzoic acid, precipitates out during the workup and can be filtered off. The patent reports a recovery rate of over 85%, allowing the material to be re-oxidized and reused, thereby lowering raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Exemestane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for high-value oncology intermediates like Exemestane. Our technical team has extensively analyzed the IBX oxidation pathway and possesses the expertise to optimize this chemistry for industrial application, ensuring that the theoretical benefits of reagent recycling and simplified purification are fully realized in practice. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to maintain stringent purity specifications and rigorous QC labs. Our commitment to process excellence ensures that every batch meets the exacting standards required for global pharmaceutical registration, providing our partners with a secure and compliant supply chain.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible economic advantages of this advanced synthesis method. Let us help you optimize your supply chain for Exemestane, ensuring cost-efficiency and reliability for your critical drug development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
