Advanced One-Step Acetylation Strategy for Commercial Deflazacort Production and Scale-Up
Advanced One-Step Acetylation Strategy for Commercial Deflazacort Production and Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the manufacture of high-value corticosteroids. Patent CN100358914C presents a transformative methodology for the preparation of Deflazacort, a potent glucocorticoid widely utilized for its anti-inflammatory properties and favorable calcium-sparing profile compared to prednisone. This intellectual property discloses a highly efficient, one-step acetylation process that converts the dihydroxy precursor directly into the active pharmaceutical ingredient (API). By leveraging a specific catalytic system involving 4-(N,N-dimethylamino)pyridine (DMAP) in an aqueous-organic biphasic environment, this technology addresses longstanding challenges in regioselectivity and process safety. For R&D directors and procurement strategists, understanding the nuances of this patent is critical for securing a reliable Deflazacort supplier capable of delivering high-purity materials at a competitive cost structure.
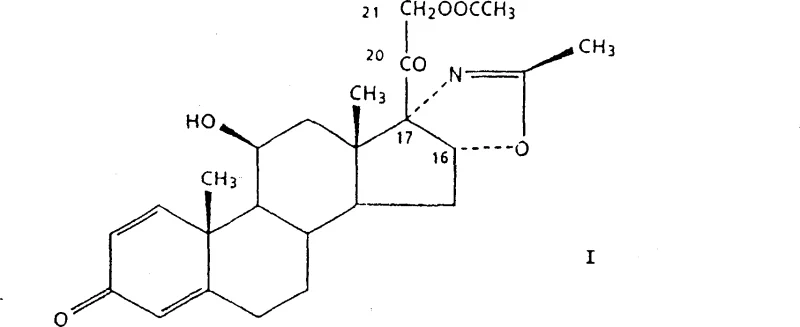
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Deflazacort, such as those detailed in earlier patents like US 3413286, are characterized by excessive complexity and significant environmental liabilities. These traditional methodologies typically initiate from 5-pregnane-3β-ol-11,20-dione derivatives and necessitate a multi-step sequence involving halogenation with elemental bromine in dioxane, followed by thermal dehydrohalogenation using lithium salts. Subsequent conversion to the 21-iodo compound and finally to the 21-acetoxy derivative introduces multiple unit operations, each carrying risks of yield loss and impurity generation. Furthermore, the handling of volatile halogens and the generation of stoichiometric amounts of inorganic salt waste pose severe safety hazards and disposal costs. The cumulative effect of these steps results in a prolonged manufacturing cycle, increased capital expenditure for specialized corrosion-resistant equipment, and a broader impurity profile that complicates downstream purification efforts.
The Novel Approach
In stark contrast, the process disclosed in CN100358914C streamlines the synthesis into a single, elegant transformation. The innovation lies in the direct acetylation of the C21-hydroxyl group of the precursor, known as Formula II or (11β,16β)-11,21-dihydroxy-2'-methyl-5'H-pregna-1,4-dieno[17,16-d]oxazole-3,20-dione. 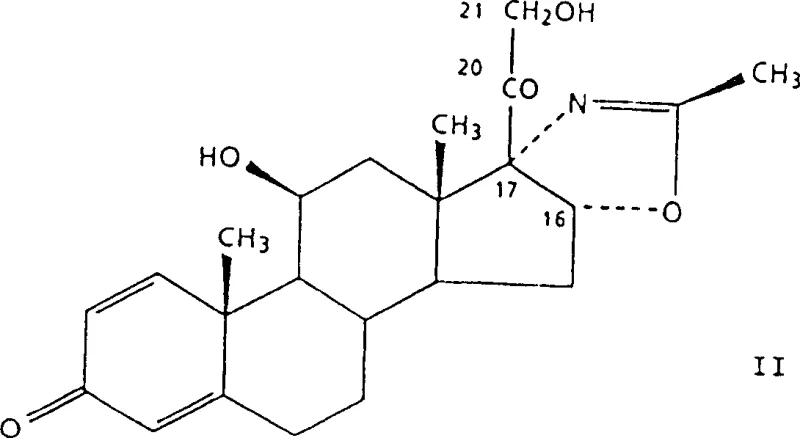 This precursor is typically obtained via fermentation, aligning the chemical step with biotechnological upstream processing. The novel method bypasses the need for halogenation entirely, reacting the dihydroxy compound directly with acetic anhydride in the presence of a basic catalyst and water. This simplification not only reduces the number of isolation steps but also dramatically improves the atom economy of the process. By eliminating the iodination and bromination stages, the manufacturing footprint is significantly reduced, allowing for faster batch turnover and a more agile response to market demand fluctuations.
This precursor is typically obtained via fermentation, aligning the chemical step with biotechnological upstream processing. The novel method bypasses the need for halogenation entirely, reacting the dihydroxy compound directly with acetic anhydride in the presence of a basic catalyst and water. This simplification not only reduces the number of isolation steps but also dramatically improves the atom economy of the process. By eliminating the iodination and bromination stages, the manufacturing footprint is significantly reduced, allowing for faster batch turnover and a more agile response to market demand fluctuations.
Mechanistic Insights into DMAP-Catalyzed Selective Acetylation
The core of this technological advancement rests on the precise control of reaction kinetics through nucleophilic catalysis. The use of 4-(N,N-dimethylamino)pyridine (DMAP) is pivotal, as it acts as a superior acyl transfer agent compared to traditional tertiary amines like pyridine or triethylamine. In the reaction medium, DMAP attacks the acetic anhydride to form a highly reactive N-acetylpyridinium intermediate. This activated species then selectively transfers the acetyl group to the primary hydroxyl group at the C21 position of the steroid backbone. The selectivity is crucial because the molecule also possesses a secondary hydroxyl group at the C11 position; however, steric hindrance and electronic factors favor the acylation of the less hindered primary alcohol. The presence of water in the reaction mixture, seemingly counterintuitive for an acetylation, plays a vital role in solubilizing the catalyst and potentially moderating the exotherm, ensuring that the reaction proceeds smoothly without degrading the sensitive oxazoline ring fused to the D-ring of the steroid.
Impurity control is inherently built into this mechanistic design. In conventional acid-catalyzed acetylations, there is a risk of hydrolyzing the oxazoline ring or causing migration of the double bonds in the A-ring. The mild basic conditions provided by the DMAP catalyst, coupled with the controlled temperature range of -5°C to 10°C (optimally 5°C), preserve the stereochemical integrity of the 11β-hydroxyl and the 1,4-diene system. Furthermore, the specific molar ratio of acetic anhydride (1.2 to 2.0 equivalents, preferably 1.4) ensures complete conversion of the starting material while minimizing the formation of di-acetylated byproducts. The ability to monitor the reaction progress via HPLC allows operators to quench the reaction precisely at the point of maximum yield, typically within 4 to 15 hours, preventing over-reaction or degradation. This tight control over the reaction trajectory results in a crude product profile that is exceptionally clean, facilitating the high recovery rates observed in the examples.
How to Synthesize Deflazacort Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and workup procedures to maximize yield and purity. The patent highlights ethyl acetate as the preferred solvent, which serves a dual purpose: it acts as the reaction medium and subsequently as the crystallization solvent. This "telescoped" approach eliminates the need for solvent swaps, which are often major sources of yield loss and operational delay in pharmaceutical manufacturing. The process begins by dissolving the Formula II precursor in ethyl acetate containing a specific amount of water, followed by the addition of acetic anhydride and the DMAP catalyst under cooling. Following the reaction, the mixture is washed with a mild buffer solution to remove the catalyst and acidic byproducts, and the product is induced to crystallize directly from the organic layer by concentration and cooling. This streamlined workflow exemplifies modern process chemistry principles, focusing on efficiency and waste minimization.
- Dissolve the Formula II precursor (11β,16β)-11,21-dihydroxy-2'-methyl-5'H-pregna-1,4-dieno[17,16-d]oxazole-3,20-dione in ethyl acetate containing controlled amounts of water.
- Add acetic anhydride (1.4 molar equivalents) and 4-(N,N-dimethylamino)pyridine (DMAP) catalyst (0.06 molar equivalents) at a controlled temperature of 5°C.
- Stir the reaction mixture for 4 to 15 hours, monitor via HPLC, then quench with phosphate buffer and crystallize the product directly from the organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN100358914C offers compelling strategic advantages beyond mere technical elegance. The shift from a multi-step halogenation route to a direct acetylation fundamentally alters the cost structure and risk profile of Deflazacort manufacturing. By removing the requirement for elemental bromine, iodine, and lithium salts, the raw material costs are drastically simplified, and the dependency on volatile commodity chemicals subject to market price fluctuations is reduced. Moreover, the elimination of hazardous halogenation steps significantly lowers the regulatory burden associated with waste disposal and worker safety, translating into lower operational overheads and insurance costs for the manufacturing facility.
- Cost Reduction in Manufacturing: The most significant economic driver is the reduction in processing time and unit operations. Traditional routes require multiple isolation and purification steps between bromination, elimination, and iodination, each consuming energy, solvents, and labor. The novel one-step acetylation consolidates these into a single reactor operation. Additionally, the ability to crystallize the product directly from the reaction solvent (ethyl acetate) avoids the costly and time-consuming process of solvent exchange or extensive chromatography. This efficiency gain leads to substantial cost savings in utilities and consumables, allowing for a more competitive pricing strategy for the final API without compromising margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes; the more steps involved, the higher the probability of a bottleneck or failure at any given stage. This streamlined process enhances reliability by reducing the number of critical control points. The starting material, Formula II, is produced via fermentation, a scalable and robust technology that decouples the supply of the precursor from petrochemical feedstock volatility. By partnering with a supplier utilizing this fermentation-plus-acetylation strategy, buyers can secure a more stable supply of high-purity Deflazacort, mitigating the risks associated with complex chemical synthesis bottlenecks.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a process becomes a key selection criterion. This method generates significantly less hazardous waste compared to halogen-based routes, aligning with Green Chemistry principles. The use of ethyl acetate, a relatively benign solvent, further supports environmental compliance. From a scalability perspective, the reaction conditions (near ambient temperature, atmospheric pressure) are easily transferable from pilot plant to commercial scale (100 MT level) without requiring exotic high-pressure or cryogenic equipment. This ease of scale-up ensures that suppliers can rapidly ramp up production to meet surges in global demand for corticosteroid therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Deflazacort using this patented methodology. Understanding these details helps stakeholders evaluate the feasibility and quality implications of this supply source. These insights underscore the robustness of the process and its suitability for GMP manufacturing environments.
Q: What are the critical reaction parameters for Deflazacort acetylation?
A: The process requires precise control of temperature (preferably 5°C), a molar ratio of precursor to acetic anhydride of approximately 1:1.4, and the presence of water (approx. 10 molar equivalents) to facilitate the reaction kinetics without hydrolyzing the product.
Q: How does this method improve upon historical synthesis routes?
A: Unlike prior art methods involving hazardous bromination and iodination steps, this novel approach utilizes a direct, one-step enzymatic-friendly acetylation that eliminates heavy metal waste and significantly reduces processing time.
Q: What is the expected purity profile of the final API intermediate?
A: By employing direct crystallization from the reaction solvent (ethyl acetate) after a mild aqueous workup, the process consistently yields Deflazacort with purity exceeding 98%, minimizing the need for extensive chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deflazacort Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous quality assurance. Our team specializes in the process development and scale-up of complex corticosteroid intermediates, including the implementation of advanced acetylation technologies like those described in CN100358914C. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to perform comprehensive impurity profiling, guaranteeing that every batch of Deflazacort adheres to the highest international pharmacopoeial standards.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By leveraging our capabilities, you can achieve significant efficiencies in your API sourcing strategy. We encourage you to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our technical prowess can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
