Advanced Synthesis of 2-Methyl-5-Aminophenol: Technical Upgrades for Commercial Mass Production
The chemical industry is constantly evolving, driven by the need for more efficient, environmentally friendly, and cost-effective synthetic pathways for critical intermediates. A recent technological breakthrough documented in patent CN114436864A introduces a superior preparation method for 2-methyl-5-aminophenol, a versatile compound widely utilized in the synthesis of resins, pigments, dyes, and increasingly in pharmaceutical and agrochemical applications. This patent details a novel three-step process that fundamentally shifts the paradigm from traditional, waste-intensive methods to a streamlined nucleophilic substitution and hydrogenation strategy. By leveraging benzyl alcohol as a key reagent in a controlled low-temperature environment, this innovation addresses long-standing challenges regarding yield, purity, and environmental impact. For global procurement and R&D teams, understanding this shift is crucial, as it represents a tangible opportunity for cost reduction in fine chemical manufacturing while ensuring a stable supply of high-purity 2-methyl-5-aminophenol. The following analysis dissects the technical merits and commercial implications of this patented technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-5-aminophenol has been plagued by inefficient routes that impose heavy burdens on both production costs and environmental compliance. One of the most common traditional methods involves using 2-methyl-5-nitroaniline as a starting material, which undergoes salification, diazotization, and subsequent hydrolysis to form 2-methyl-5-nitrophenol before final reduction. As illustrated in the reaction scheme below, this multi-step pathway is inherently flawed due to the diazo hydrolysis step, which generates a significant quantity of oxidation byproducts and waste acid.
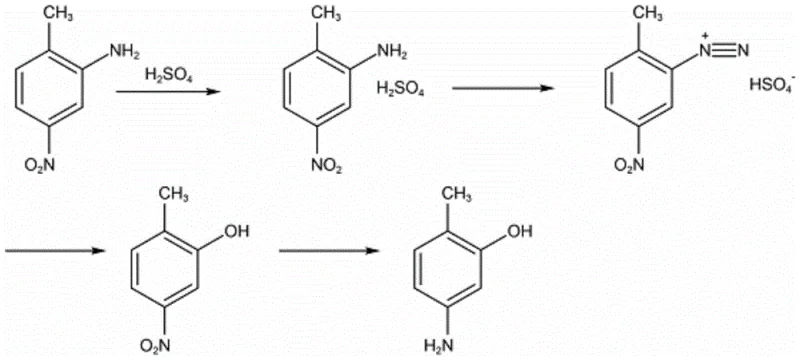
These byproducts not only depress the total reaction yield to approximately 70% but also create severe pollution issues that require expensive treatment protocols. Other historical routes, such as those involving fluorination or high-temperature hydrolysis of chloro-anilines, suffer from similar drawbacks, including the use of prohibitively expensive fluorinating reagents, the generation of fluorine-containing wastewater, or the need for extreme pressure and temperature conditions that strain equipment integrity. These conventional limitations result in a fragmented supply chain where reliable pharmaceutical intermediate supplier status is hard to maintain due to inconsistent batch quality and regulatory hurdles associated with waste disposal.
The Novel Approach
In stark contrast, the method disclosed in patent CN114436864A offers a transformative solution by employing a nucleophilic substitution reaction between benzyl alcohol and 3-chloro-4-methylnitrobenzene. This innovative route bypasses the problematic diazotization and harsh hydrolysis steps entirely. The core of this novelty lies in the strategic use of benzyl alcohol, which acts as a nucleophile to displace the chlorine atom under mild alkaline conditions, forming a stable intermediate that is subsequently converted to the target phenol via hydrogenation.
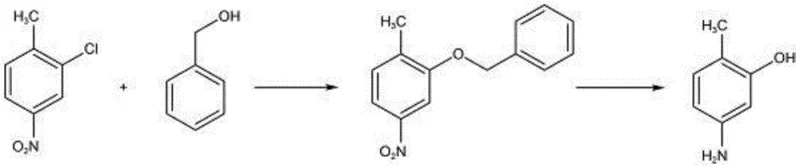
This approach drastically simplifies the operational workflow, reducing the number of unit operations and eliminating the need for hazardous reagents like strong mineral acids in large quantities or expensive fluorinating agents. The reaction conditions are remarkably mild, typically proceeding at temperatures between 10-20 ℃ for the substitution step, which minimizes thermal degradation and side reactions. Furthermore, the subsequent hydrogenation step effectively reduces the nitro group and cleaves the benzyl protecting group in a single operation, delivering the final product with exceptional efficiency. This streamlined methodology not only enhances the economic viability of the process but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nucleophilic Substitution and Catalytic Hydrogenation
The success of this synthesis hinges on a precise understanding of the reaction mechanisms involved, particularly the nucleophilic aromatic substitution facilitated by the strong base. In the first stage, a strong base such as sodium hydroxide or potassium hydroxide reacts with benzyl alcohol to generate the corresponding alkoxide species in situ within a toluene solvent system. This alkoxide then attacks the electron-deficient aromatic ring of 3-chloro-4-methylnitrobenzene, specifically targeting the carbon bearing the chlorine atom, which is activated by the adjacent nitro group. The reaction is meticulously controlled at a low temperature range of 10-20 ℃ to prevent competing side reactions and ensure the selective formation of the benzyl ether intermediate. The use of a water separator during the initial mixing allows for the continuous removal of water generated during the alkoxide formation, driving the equilibrium forward and maximizing the conversion of the starting materials.
Following the isolation of the intermediate, the process transitions to a catalytic hydrogenation phase, which serves a dual purpose: reducing the nitro group to an amino group and simultaneously cleaving the benzyl-oxygen bond to reveal the phenolic hydroxyl group. This tandem transformation is achieved using robust catalysts such as palladium on carbon (Pd/C) or Raney nickel under a hydrogen pressure of 0.7-1.0 MPa and temperatures between 50-75 ℃. The choice of catalyst and the careful control of hydrogen pressure are critical for minimizing impurities; for instance, maintaining the pressure within the specified range ensures complete reduction without over-hydrogenating the aromatic ring. The purification step further refines the product through decolorization and recrystallization from a 45% ethanol-water mixture, effectively removing trace organic impurities and residual catalysts. This rigorous control over the reaction parameters and purification logic ensures that the final high-purity 2-methyl-5-aminophenol meets stringent specifications, with purity levels consistently exceeding 99.89%.
How to Synthesize 2-Methyl-5-Aminophenol Efficiently
Implementing this patented technology requires adherence to specific operational protocols to maximize yield and safety. The process begins with the preparation of the nucleophilic species in toluene, followed by the controlled addition of the chloronitrobenzene substrate. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and workup strategies are essential for reproducibility. For a comprehensive guide on the exact molar ratios, solvent recovery techniques, and crystallization parameters required to achieve the reported 95.3% yield, please refer to the standardized synthesis steps outlined below.
- Perform nucleophilic substitution between benzyl alcohol and 3-chloro-4-methylnitrobenzene using a strong base in toluene at 10-20°C.
- Execute catalytic hydrogenation reduction on the intermediate using Pd/C or Raney Nickel at 50-75°C and 0.7-1.0 MPa pressure.
- Purify the crude product through decolorization and recrystallization in a 45% ethanol-water solution to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into immediate and substantial strategic benefits. The shift away from complex, multi-step traditional methods towards this streamlined two-reaction sequence significantly de-risks the supply chain. By eliminating the reliance on hazardous diazotization reagents and high-pressure hydrolysis equipment, manufacturers can operate with greater flexibility and lower capital expenditure. The simplicity of the process means that production bottlenecks are minimized, leading to more consistent lead times and a more reliable flow of materials to downstream customers. This reliability is paramount for industries such as pharmaceuticals and agrochemicals, where supply continuity is often as critical as price.
- Cost Reduction in Manufacturing: The economic advantages of this method are derived from several key factors inherent to the process design. Firstly, the raw materials, specifically 3-chloro-4-methylnitrobenzene and benzyl alcohol, are commodity chemicals that are readily available and cost-effective compared to the specialized reagents required for fluorination or Mannich reaction routes. Secondly, the elimination of waste acid treatment and the reduction in three-waste generation drastically lower the operational costs associated with environmental compliance and waste disposal. Furthermore, the ability to recover and reuse solvents like toluene and ethanol, along with the catalyst, contributes to a leaner manufacturing model. These cumulative efficiencies result in a significantly reduced cost of goods sold (COGS), allowing for more competitive pricing structures without compromising margin.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route directly enhances supply chain resilience. Because the reaction conditions are mild (10-20 ℃ for substitution and moderate pressure for hydrogenation), the process is less susceptible to equipment failures or safety incidents that often plague high-temperature, high-pressure operations. This operational stability ensures that production schedules can be met with high predictability. Additionally, the use of common, non-proprietary catalysts like Raney Nickel or Pd/C means that supply disruptions for specialized catalytic materials are unlikely to impact production. This reliability makes the manufacturer a preferred partner for long-term contracts, ensuring that clients receive their pharmaceutical intermediates on time, every time.
- Scalability and Environmental Compliance: From a scalability perspective, this method is exceptionally well-suited for mass production. The absence of exothermic runaway risks associated with diazotization and the use of standard hydrogenation equipment allow for seamless scaling from pilot plants to multi-ton commercial reactors. Moreover, the environmental profile of the process is vastly superior to legacy methods. By generating mainly chloride salts as byproducts instead of high-COD wastewater or fluorine-laden effluents, the facility can maintain a smaller environmental footprint. This alignment with green chemistry standards not only simplifies regulatory permitting but also appeals to end-users who are increasingly prioritizing sustainable sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-methyl-5-aminophenol using this advanced methodology. These insights are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity on how this process outperforms traditional alternatives in terms of purity, yield, and operational safety.
Q: What are the primary advantages of this new synthesis route over traditional diazotization methods?
A: The novel route eliminates the generation of large amounts of waste acid and oxidation byproducts associated with diazo hydrolysis, resulting in a cleaner process with yields exceeding 95% and purity above 99.89%.
Q: How does the process ensure environmental compliance and waste reduction?
A: By utilizing mild reaction conditions and generating mainly chloride salts as byproducts instead of high-COD wastewater or fluorine-containing effluents, the method significantly lowers the environmental burden and simplifies three-waste treatment.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process operates at low temperatures (10-20°C) and moderate hydrogen pressures (0.7-1.0 MPa), reducing equipment requirements and safety risks, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-5-Aminophenol Supplier
The technical potential of patent CN114436864A represents a significant leap forward in the manufacturing of 2-methyl-5-aminophenol, offering a pathway to higher purity and lower environmental impact. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this innovation to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of 2-methyl-5-aminophenol we deliver meets the exacting standards required by the pharmaceutical and fine chemical industries.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the partner of choice for high-quality chemical intermediates.
