Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Aryl Oxazoline Amide Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Aryl Oxazoline Amide Production
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign processes. A significant breakthrough in this domain is detailed in patent CN108586372B, which discloses a novel synthetic method for 2-aryl oxazoline amide compounds. These compounds are not merely end-products but serve as critical N,N-bidentate directing groups in inert C-H bond functionalization, a cornerstone strategy in modern medicinal chemistry for constructing complex molecular architectures. The patented methodology leverages a palladium-catalyzed carbonylation reaction, utilizing carbon monoxide (CO) gas as a readily available and inexpensive C1 building block. This approach represents a paradigm shift from traditional multi-step syntheses, offering a streamlined one-pot procedure that operates under relatively mild conditions. For research and development teams seeking reliable pharmaceutical intermediate suppliers, understanding the nuances of this technology is essential for optimizing supply chains and reducing the overall cost of goods sold in API manufacturing.
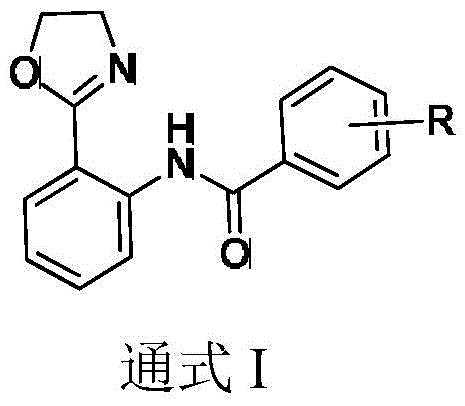
The versatility of this synthetic route is underscored by its broad substrate scope, accommodating various substituents on the aryl ring. As illustrated in the general formula, the R group can be hydrogen, alkyl, methoxy (-OMe), trifluoromethyl (-CF3), or halogens, allowing for the rapid generation of diverse chemical libraries. This flexibility is paramount for drug discovery programs where structure-activity relationship (SAR) studies require quick access to analogues. Furthermore, the use of CO gas as a carbonyl source eliminates the need for pre-functionalized carboxylic acid derivatives, which are often more expensive and generate more waste. By integrating this technology into their procurement strategies, supply chain heads can secure a more robust pipeline of high-purity intermediates, mitigating risks associated with raw material volatility and complex logistics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aryl oxazoline amides has been fraught with challenges that hinder scalability and economic efficiency. Traditional routes often involve multiple steps, starting from carboxylic acids that require activation prior to amidation, or coupling reactions that necessitate expensive coupling reagents and stringent anhydrous conditions. These multi-step sequences inevitably lead to cumulative yield losses, increased solvent consumption, and a larger environmental footprint due to higher E-factors. Moreover, the purification of intermediates at each stage adds significant time and operational costs, creating bottlenecks in the production schedule. For procurement managers focused on cost reduction in API manufacturing, these inefficiencies translate directly into higher unit prices and longer lead times. Additionally, the reliance on specialized reagents can introduce supply chain vulnerabilities, where disruptions in the availability of a single precursor can halt the entire production line, posing a significant risk to project timelines.
The Novel Approach
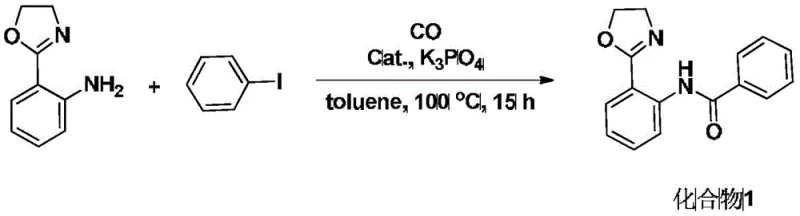
In stark contrast, the method described in CN108586372B offers a transformative solution by condensing the synthesis into a single catalytic step. This novel approach utilizes 2-oxazoline aniline and aryl iodides as starting materials, reacting them directly with CO gas in the presence of a palladium catalyst and a base. The reaction proceeds smoothly at temperatures between 80 and 100 degrees Celsius, which are easily achievable in standard industrial reactors without requiring cryogenic conditions or extreme pressures. The simplicity of the workup, involving merely rotary evaporation followed by chromatographic separation, drastically reduces processing time and labor costs. This streamlined process not only enhances the overall throughput but also aligns with green chemistry principles by minimizing waste generation. For organizations aiming for the commercial scale-up of complex pharmaceutical intermediates, this one-step carbonylation strategy provides a clear pathway to achieving both economic and operational excellence.
Mechanistic Insights into Pd-Catalyzed Carbonylative Coupling
The core of this innovation lies in the efficient palladium-catalyzed carbonylative coupling mechanism. The reaction initiates with the oxidative addition of the aryl iodide to the active palladium(0) species, forming an aryl-palladium(II) intermediate. Subsequently, the insertion of carbon monoxide into the palladium-carbon bond generates an acyl-palladium complex. This key intermediate then undergoes nucleophilic attack by the amino group of the 2-oxazoline aniline, followed by reductive elimination to release the desired 2-aryl oxazoline amide product and regenerate the palladium catalyst. The choice of ligand and catalyst precursor is critical; screening data within the patent reveals that PdCl2(CH3CN)2 exhibits superior activity compared to other palladium sources like Pd2(dba)3 or PdCl2, achieving yields as high as 99 percent. This high catalytic efficiency allows for lower catalyst loadings, typically in the range of 0.05 to 0.1 molar equivalents, which is a significant advantage for reducing metal contamination in the final product.
Impurity control is another critical aspect where this mechanistic understanding pays dividends. The high selectivity of the palladium catalyst ensures that side reactions, such as homocoupling of the aryl iodide or dehalogenation, are minimized. The use of a strong base like potassium phosphate facilitates the deprotonation of the amine nucleophile, driving the reaction forward while maintaining a clean reaction profile. The patent data indicates that yields consistently exceed 90 percent across a wide range of substrates, including those with electron-withdrawing groups like trifluoromethyl or electron-donating groups like methoxy. This robustness suggests that the catalytic cycle is tolerant to various electronic environments, making it a reliable method for producing high-purity intermediates. For R&D directors, this level of predictability and purity is essential for ensuring the quality of downstream processes and meeting stringent regulatory specifications for pharmaceutical ingredients.
How to Synthesize 2-Aryl Oxazoline Amide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and safety. The process begins with the sequential addition of 2-oxazoline aniline, the chosen aryl iodide, a base such as potassium phosphate, the palladium catalyst, and an organic solvent like anhydrous toluene into a dry reaction vessel. It is crucial to maintain an inert atmosphere initially to prevent catalyst deactivation. Once the reagents are mixed, CO gas is introduced into the system.
- Charge a dry reaction vessel with 2-oxazoline aniline, aryl iodide, base (e.g., K3PO4), palladium catalyst, and organic solvent like toluene.
- Bubble CO gas through the mixture at room temperature for 10 to 15 minutes to saturate the solution before sealing the system.
- Heat the sealed reaction system to 80 to 100 degrees Celsius for 10 to 15 hours, then concentrate and purify via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages that resonate deeply with procurement and supply chain objectives. The primary driver for cost optimization is the substitution of expensive activated carboxylic acid derivatives with carbon monoxide gas, which is an abundant and low-cost industrial commodity. This fundamental change in raw material sourcing significantly lowers the direct material costs associated with production. Furthermore, the reduction in synthetic steps from multiple stages to a single pot operation drastically cuts down on solvent usage, energy consumption, and labor hours. These operational efficiencies compound to deliver substantial cost savings, making the final intermediate more competitive in the global market. For procurement managers negotiating contracts, understanding these underlying process improvements provides leverage in discussions regarding pricing and long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of pre-activation steps and the use of low-loading palladium catalysts directly contribute to a leaner manufacturing cost structure. By avoiding the purchase of costly coupling reagents and reducing the volume of solvents required for multiple workups, the overall expense per kilogram of product is markedly decreased. Additionally, the high yields reported (>90 percent) mean that less raw material is wasted, further enhancing the economic viability of the process. This efficiency allows manufacturers to offer more attractive pricing models without compromising on quality, addressing the constant pressure to reduce costs in API manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically 2-oxazoline anilines and aryl iodides, are generally commercially available and stable, reducing the risk of supply disruptions. Unlike specialized reagents that may have limited suppliers or long lead times, these precursors can be sourced from multiple vendors, ensuring a continuous flow of materials. The robustness of the reaction conditions also means that production is less susceptible to minor variations in environmental factors, leading to more consistent output. This reliability is crucial for supply chain heads who need to guarantee delivery schedules to downstream clients and maintain inventory levels without excessive safety stock.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving standard concentration and chromatography, facilitates easy scale-up from gram to ton quantities. The process does not generate hazardous by-products in significant quantities, aligning with increasingly strict environmental regulations. The use of CO gas, while requiring safety precautions, is a well-established practice in the chemical industry, and the closed-system nature of the reaction minimizes emissions. This environmental compatibility reduces the burden of waste treatment and disposal, lowering overhead costs and enhancing the sustainability profile of the manufacturing operation, which is a growing priority for corporate social responsibility initiatives.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common questions based on the patent data. These insights are designed to address specific concerns regarding implementation, quality, and scalability. Understanding these details helps stakeholders make informed decisions about integrating this synthetic method into their existing portfolios.
Q: What is the typical yield for this palladium-catalyzed carbonylation process?
A: According to patent CN108586372B, the process demonstrates excellent substrate universality with yields typically exceeding 90 percent for most tested aryl iodides, including those with electron-donating or withdrawing groups.
Q: Which palladium catalysts are most effective for this transformation?
A: Screening data indicates that PdCl2(CH3CN)2 provides superior results with yields up to 99 percent, outperforming other common catalysts like Pd2(dba)3 or Pd(PPh3)4 in this specific carbonylative coupling.
Q: Is this method suitable for large-scale manufacturing of API intermediates?
A: Yes, the use of inexpensive CO gas as a carbonyl source and simple workup procedures involving rotary evaporation and chromatography makes this route highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Oxazoline Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN108586372B for the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific analogues or bulk supply of standard intermediates, our infrastructure is designed to support your growth and innovation goals with unwavering reliability and quality assurance.
We invite you to explore how our expertise in palladium-catalyzed carbonylation can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your requirements for high-purity intermediates. Let us be your partner in navigating the complexities of modern chemical synthesis and securing a competitive edge in the global marketplace.
