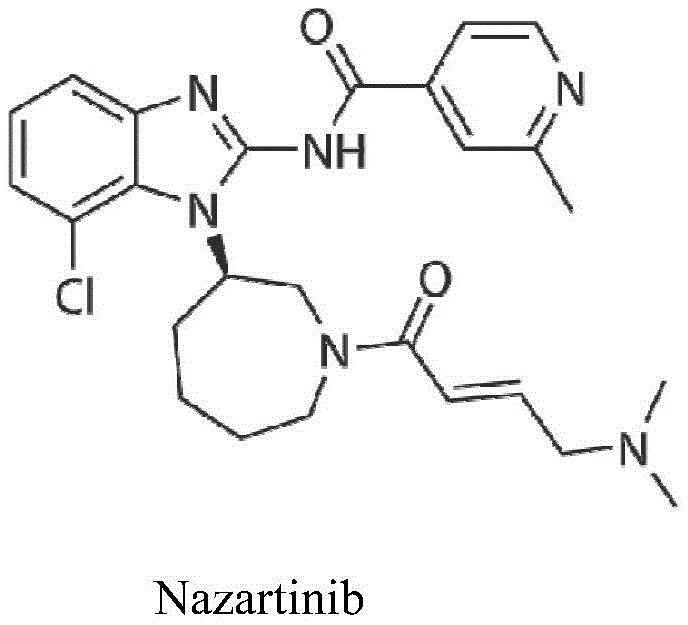
Optimizing 2-Methylpyridine-4-Formic Acid Production for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously demands more efficient, sustainable, and cost-effective pathways for synthesizing critical active pharmaceutical ingredient (API) intermediates. A significant breakthrough in this domain is detailed in patent CN110452165B, which outlines a novel preparation method for 2-methylpyridine-4-formic acid. This specific pyridine derivative serves as a pivotal building block for the synthesis of Nazartinib (EGF816), a third-generation EGFR inhibitor currently in advanced clinical stages for treating non-small cell lung cancer. The traditional manufacturing of such heterocyclic acids often suffers from harsh conditions, low selectivity, and difficult purification processes. However, the methodology disclosed in this patent introduces a streamlined approach that leverages mild reaction conditions and high-selectivity cyclization to achieve superior yields. By optimizing the interaction between dialkoxy precursors and ketone functionalities, this technology addresses the critical pain points of impurity control and environmental impact. For global procurement teams and R&D directors, understanding this synthetic evolution is paramount for securing a stable supply of high-quality intermediates.
The limitations of conventional pyridine synthesis methods often revolve around the use of aggressive reagents and multi-step isolation procedures that degrade overall efficiency. Traditional routes may involve high-temperature condensations that promote polymerization or require expensive transition metal catalysts that necessitate rigorous removal protocols to meet residual metal specifications. These factors invariably drive up the cost of goods sold (COGS) and extend the lead time for batch release. Furthermore, older methodologies frequently struggle with regioselectivity, leading to complex impurity profiles that complicate downstream crystallization. In contrast, the novel approach presented in the patent data utilizes a strategic construction of the pyridine ring through a carefully orchestrated sequence starting from accessible acetal-protected precursors. This new pathway avoids the pitfalls of uncontrolled exothermic reactions and instead employs a controlled, step-wise assembly that maximizes atom economy. The shift from chaotic high-energy environments to moderated, catalytic, or base-mediated conditions represents a fundamental upgrade in process safety and operational simplicity.
Mechanistic Insights into the Cyclization and Oxidation Sequence
The core of this synthetic innovation lies in the precise mechanistic execution of the ring-closing step and the subsequent oxidative aromatization. The process begins with the formation of a 1,5-dicarbonyl equivalent (Compound III) through either a nucleophilic substitution with chloroacetone or a ring-opening addition with propylene oxide followed by oxidation. This intermediate possesses a unique reactivity profile where the acetal moiety acts as a masked aldehyde. Upon exposure to an ammonia and ammonium salt system, the acetal undergoes hydrolysis in situ to reveal the reactive aldehyde group, which then immediately participates in a Hantzsch-like condensation with the adjacent ketone and the nitrogen source. This cascade reaction forms the 1,4-dihydropyridine scaffold (Compound IV) with remarkable specificity. The patent highlights that maintaining a low concentration of the ketone intermediate through dropwise addition is critical; this kinetic control prevents intermolecular self-condensation or polymerization, which are common failure modes in pyridine synthesis. Following cyclization, the dihydropyridine ring is aromatized using mild oxidants such as hydrogen peroxide or tert-butyl peroxide. This oxidation step is seamlessly integrated into the same reaction vessel, eliminating the need for intermediate isolation and significantly reducing solvent consumption and waste generation.
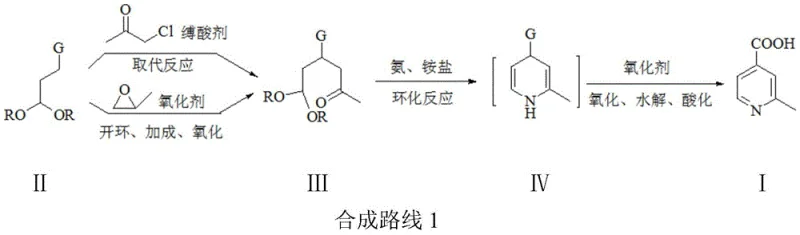
Impurity control is inherently built into the chemical design of this route, offering distinct advantages for quality assurance teams. The use of acetal protection ensures that the aldehyde functionality is only revealed when the reaction environment is perfectly tuned for cyclization, thereby preventing premature side reactions such as aldol condensations that could occur if free aldehydes were present from the start. Furthermore, the selection of ammonium salts alongside ammonia buffers the reaction pH, facilitating a smoother cyclization trajectory that minimizes the formation of tar-like byproducts. The final acidification step using hydrochloric acid not only liberates the free carboxylic acid from its salt form but also precipitates the product in a highly pure state, as evidenced by liquid phase purity readings consistently above 99.5% in the provided examples. This level of intrinsic purity reduces the burden on downstream processing, allowing manufacturers to bypass extensive chromatographic purifications that are often cost-prohibitive at scale. The result is a robust process capable of delivering material that meets stringent regulatory standards for pharmaceutical intermediates with minimal post-reaction workup.
How to Synthesize 2-Methylpyridine-4-Formic Acid Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and addition rates to ensure reproducibility and safety. The process is designed to be operationally simple, utilizing common industrial solvents like water, alcohols, and toluene, which facilitates easy technology transfer from the laboratory to the pilot plant. The initial formation of the hexyl-ketone precursor can be achieved via two distinct pathways, offering flexibility depending on raw material availability and cost fluctuations in the supply market. Once the precursor is secured, the critical cyclization and oxidation sequence proceeds in a 'one-pot' fashion, drastically simplifying the equipment requirements and operator intervention. For detailed standard operating procedures and specific reaction conditions tailored to your facility's capabilities, please refer to the technical guide below.
- Prepare compound III by reacting 4,4-dialkoxy n-butyronitrile or ester (II) with chloroacetone via substitution, or with propylene oxide via ring-opening and oxidation.
- Perform cyclization of compound III with ammonia and ammonium salt in solvent B to form the dihydropyridine intermediate (IV).
- Execute one-pot oxidation, hydrolysis, and acidification to yield high-purity 2-methylpyridine-4-formic acid (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of complex isolation steps between the cyclization and oxidation phases translates into significant reductions in processing time and solvent usage. By consolidating multiple chemical transformations into a single reactor train, facilities can increase their throughput capacity without requiring additional capital investment in hardware. This intensification of the process flow is a key driver for cost reduction in API manufacturing, as it lowers both variable costs (solvents, energy, labor) and fixed costs associated with equipment occupancy. Moreover, the high selectivity of the reaction means that raw materials are converted into the desired product with minimal waste, enhancing the overall mass balance and sustainability profile of the operation. These efficiencies make the production of 2-methylpyridine-4-formic acid more economically viable, allowing for competitive pricing strategies in the global market.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the avoidance of expensive transition metal catalysts and the reduction of unit operations. By utilizing inexpensive oxidants like hydrogen peroxide and common bases, the direct material costs are kept low. Additionally, the high yield reported in the patent examples implies that less raw material is required to produce a kilogram of finished product, effectively lowering the cost per unit. The simplified workup procedure further reduces the consumption of auxiliary chemicals and the energy load associated with distillation and drying, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as 4,4-dialkoxy n-butyronitrile and chloroacetone, ensures a stable and diversified supply base. These commodities are produced by multiple chemical suppliers globally, mitigating the risk of single-source bottlenecks that can plague specialized reagent markets. The robustness of the synthesis against minor variations in conditions also means that production schedules are less likely to be disrupted by batch failures or off-spec material. This reliability is crucial for maintaining continuous supply to downstream drug manufacturers who operate on tight just-in-time inventory models.
- Scalability and Environmental Compliance: The mild reaction temperatures, typically ranging from 60°C to 80°C, reduce the thermal load on reactor cooling systems and minimize safety risks associated with high-pressure or high-temperature operations. This makes the process inherently safer and easier to scale from pilot batches to multi-ton commercial production. Furthermore, the generation of low amounts of waste acid and wastewater aligns with increasingly stringent environmental regulations. The ability to operate a 'greener' process not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing site, which is becoming a key criterion for supplier selection by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when adopting this technology. Understanding these nuances helps in making informed decisions about process validation and long-term sourcing strategies.
Q: What is the primary advantage of this synthesis route regarding purity?
A: The process utilizes controlled dropwise addition during cyclization to minimize intermolecular side reactions and polymerization, achieving liquid phase purity exceeding 99.5% without complex recrystallization steps.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method features mild reaction conditions (60-80°C), uses commercially available raw materials, and generates low amounts of waste acid and water, making it highly suitable for green industrial manufacturing.
Q: What are the key starting materials for this process?
A: The synthesis begins with 4,4-dialkoxy n-butyronitrile or 4,4-dialkoxy n-butyric ester, which can be sourced commercially or prepared from acrylonitrile/acrylic esters, ensuring a robust supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylpyridine-4-Formic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medicines like Nazartinib. Our technical team has thoroughly analyzed the pathway described in CN110452165B and is fully equipped to execute this synthesis with precision and scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are outfitted with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, minimizing the risk of delays in your own drug substance manufacturing. We are committed to being a transparent and responsive partner in your supply chain.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume requirements and quality standards. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that identifies further opportunities for efficiency within your specific supply chain context. We encourage potential partners to request specific COA data and route feasibility assessments to validate our capabilities against your internal benchmarks. Let us collaborate to optimize the production of this vital intermediate and support the broader goal of bringing effective therapies to patients faster.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
