Advanced Biocatalytic Synthesis of Steroid Intermediates: Scaling High-Purity Production via Resting Cell Fermentation
The pharmaceutical industry is constantly seeking more efficient pathways for synthesizing complex steroid intermediates, and patent CN110713510A presents a groundbreaking methodology for producing 3 beta-hydroxyandrosta-5, 7-diene-17-one. This specific intermediate is a critical building block in the synthesis of various hormonal therapies and steroidal drugs, where stereochemical purity at the 3-position is paramount for biological activity. The disclosed invention leverages a sophisticated resting cell fermentation technique using a specifically mutagenized strain of Mycobacterium sp. B-NRRL 3683 to achieve side-chain cleavage of ergosterol derivatives. By integrating a preliminary chemical protection step with a highly optimized biological transformation, this process addresses longstanding challenges regarding isomer contamination and low yields that have plagued traditional synthetic routes. For R&D directors and procurement specialists, understanding this hybrid chemo-enzymatic approach is essential for evaluating next-generation supply chains that prioritize both environmental sustainability and cost-efficiency in high-value API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of 3 beta-hydroxyandrosta-5, 7-diene-17-one typically relies on the selective reduction of a 3-position ketone group, a process fraught with significant technical and economic inefficiencies. In conventional workflows, achieving the desired beta-configuration often results in the co-formation of the 3-alpha hydroxy isomer, necessitating complex and costly downstream purification steps to separate these stereoisomers. Furthermore, the chemical reagents required for such selective reductions are frequently expensive, hazardous, and generate substantial waste streams, complicating environmental compliance and increasing the overall cost of goods sold. The multi-step nature of these purely chemical routes also introduces cumulative yield losses at each stage, significantly impacting the final throughput and making large-scale production economically challenging for generic drug manufacturers. These inherent limitations create a bottleneck in the supply chain, leading to longer lead times and higher volatility in the pricing of critical steroid intermediates.
The Novel Approach
In stark contrast, the method described in patent CN110713510A introduces a streamlined pathway that begins with the strategic protection of the 3-hydroxyl group of ergosterol prior to fermentation. This initial etherification step fundamentally alters the physicochemical properties of the substrate, enhancing its solubility within the fermentation broth and thereby facilitating more efficient uptake by the microbial cells. The core innovation lies in the use of a resting cell system containing a mutant strain of Mycobacterium sp. B-NRRL 3683, which has been specifically engineered to cleave the C17 side chain while leaving the protected 3-position intact. This biological precision eliminates the formation of the unwanted 3-alpha isomer entirely, bypassing the need for difficult chromatographic separations. The subsequent hydrolysis step simply removes the protecting group to reveal the target molecule, resulting in a significantly shorter synthetic route that minimizes intermediate loss and maximizes overall process efficiency.
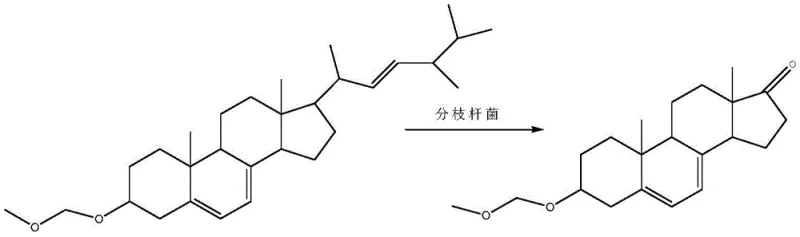
Mechanistic Insights into Resting Cell Fermentation and Strain Mutagenesis
The success of this biocatalytic process hinges on the precise genetic modification of the host organism and the careful control of the fermentation environment to optimize enzyme activity. The patent details a rigorous mutagenesis protocol using nitrosoguanidine (NTG) to evolve the parent Mycobacterium sp. B-NRRL 3683 strain, selecting for mutants that exhibit superior specificity for side-chain degradation without attacking the protected 3-position. This directed evolution is critical because wild-type strains often lack the necessary selectivity or tolerance for the modified substrate, leading to incomplete conversion or byproduct formation. The resting cell technique further refines this mechanism by separating the growth phase from the production phase, allowing the cells to focus their metabolic energy solely on the biotransformation rather than biomass accumulation. This decoupling of growth and production phases enables tighter control over reaction parameters such as pH, temperature, and oxygen transfer rates, which are vital for maintaining the stability of the delicate steroid skeleton during the oxidative cleavage process.
Impurity control is another mechanistic advantage provided by the inclusion of hydroxypropyl-beta-cyclodextrin in the conversion system. This solubilizing agent forms inclusion complexes with the hydrophobic ergosterol etherate, effectively increasing its apparent concentration in the aqueous fermentation medium and ensuring uniform exposure to the enzymatic machinery of the bacteria. By preventing substrate precipitation and aggregation, the cyclodextrin minimizes localized high-concentration zones that could otherwise lead to non-specific degradation or incomplete reactions. Furthermore, the protection of the 3-hydroxyl group as an ether serves as a robust chemical shield, preventing the microbial enzymes from oxidizing or epimerizing this sensitive center. This dual strategy of physical solubilization and chemical protection ensures that the final crude product contains minimal impurities, simplifying the subsequent refining steps and enabling the consistent production of material with purity levels exceeding 98 percent.
How to Synthesize 3 Beta-Hydroxyandrosta-5 7-Diene-17-One Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this technology from laboratory benchtop to industrial manufacturing volumes. The process is divided into five distinct operational units: 3-position protection, resting cell transformation, extraction, hydrolysis, and refining. Each step has been optimized to balance reaction kinetics with ease of handling, ensuring that the workflow remains practical for large-scale implementation. The initial protection step utilizes methylal and phosphorus pentoxide, followed by a filtration to remove catalyst residues, setting the stage for a clean fermentation feed. The biological transformation is conducted in a controlled bioreactor environment, after which the product is isolated via solid-liquid separation. Finally, acid hydrolysis removes the protecting group, and a series of recrystallizations yields the high-purity final API intermediate. For detailed standard operating procedures and specific parameter settings, please refer to the technical guide below.
- Protect the 3-hydroxyl group of ergosterol using methylal and phosphorus pentoxide to form ergosterol etherate.
- Perform biotransformation using a mutagenized Mycobacterium sp. B-NRRL 3683 strain in a resting cell system with hydroxypropyl-beta-cyclodextrin.
- Extract the solid fermentation product, hydrolyze the protecting group with hydrochloric acid, and refine via recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic route represents a strategic opportunity to de-risk the supply of critical steroid intermediates while simultaneously driving down manufacturing costs. The elimination of multiple chemical synthesis steps and the associated expensive reagents translates directly into a leaner cost structure, reducing the dependency on volatile petrochemical feedstocks. Moreover, the biological nature of the transformation operates under mild conditions, significantly lowering energy consumption compared to high-temperature and high-pressure chemical processes. This shift not only aligns with corporate sustainability goals but also reduces the regulatory burden associated with handling hazardous chemicals, thereby smoothing the path for facility approvals and inspections. The robustness of the fermentation process also implies a more stable and predictable production schedule, mitigating the risks of supply disruptions that are common in complex multi-step chemical syntheses.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic simplification of the synthetic route, which removes the need for costly chiral resolution steps and expensive reducing agents. By leveraging the inherent stereoselectivity of the mutagenized bacterial strain, manufacturers can avoid the significant material losses associated with separating alpha and beta isomers in traditional chemical methods. Additionally, the ability to recycle the bacterial liquid from the extraction phase for up to ten consecutive batches further amortizes the cost of seed culture preparation and reduces the overall consumption of fermentation media. This circular approach to biocatalyst usage creates a compounding effect on cost savings, making the final product highly competitive in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Sourcing ergosterol, the primary starting material, is inherently more stable than sourcing specialized synthetic precursors, as ergosterol is abundantly available from yeast and fungal sources globally. The fermentation process itself is highly scalable, utilizing standard bioreactor infrastructure that is widely available in the fine chemical industry, thus removing bottlenecks related to specialized equipment availability. The shortened reaction route also means fewer intermediate inventory points are required, reducing working capital tied up in work-in-progress materials and accelerating the overall cash-to-cash cycle time for production runs. This agility allows suppliers to respond more rapidly to fluctuations in market demand, ensuring continuous availability of the intermediate for downstream API synthesis.
- Scalability and Environmental Compliance: From an environmental perspective, this method significantly reduces the generation of organic solvent waste and heavy metal residues that are typical of traditional steroid synthesis. The use of aqueous fermentation media and the recycling of bacterial broth minimize the volume of effluent requiring treatment, lowering disposal costs and environmental impact fees. The process avoids the use of toxic catalysts and harsh oxidizing agents, creating a safer workplace environment and reducing the liability associated with chemical storage and handling. These green chemistry attributes not only facilitate easier regulatory approval in stringent markets like the EU and US but also enhance the brand reputation of manufacturers committed to sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resting cell fermentation technology. These answers are derived directly from the experimental data and claims presented in patent CN110713510A, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding the nuances of strain maintenance, substrate protection, and purification is crucial for successful technology transfer and scale-up operations.
Q: Why is 3-position protection necessary before fermentation?
A: Protecting the 3-hydroxyl group reduces product polarity and increases solubility in the fermentation medium, preventing unwanted side reactions and improving overall yield.
Q: What specific strain is used for this biotransformation?
A: The process utilizes a mutagenized strain of Mycobacterium sp. B-NRRL 3683, treated with nitrosoguanidine to optimize side-chain cleavage specificity.
Q: How does this method compare to traditional chemical synthesis?
A: Unlike chemical methods that often produce 3-alpha hydroxy isomers requiring difficult separation, this biological route offers high stereoselectivity for the 3-beta configuration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3 Beta-Hydroxyandrosta-5 7-Diene-17-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic routes like the one described in CN110713510A for the future of steroid manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with advanced fermentation suites and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to helping our partners navigate the complexities of process development, offering end-to-end support from strain optimization to final API intermediate delivery.
We invite you to engage with our technical procurement team to discuss how this advanced fermentation technology can be integrated into your supply chain to achieve a Customized Cost-Saving Analysis. By collaborating with us, you gain access to specific COA data and comprehensive route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable and cost-effective supply of high-purity steroid intermediates, positioning your organization for success in the competitive pharmaceutical landscape.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
