Advanced Microbial Hydroxylation for High-Purity Steroid Intermediates and Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for synthesizing complex steroid intermediates, which serve as the backbone for countless life-saving medications. Patent CN109680032B introduces a groundbreaking biotechnological advancement that addresses the longstanding challenges associated with introducing hydroxyl groups at the C7 position of the steroid nucleus. This innovation leverages the unique metabolic capabilities of specific fungal strains, including Mucor circinelloides and Aspergillus fumigatus, to convert 21-hydroxy-20-methylpregna-4-en-3-one (4-HBC) into valuable 7-hydroxy derivatives with exceptional precision. By shifting away from traditional chemical oxidation methods, this technology not only enhances the stereochemical control required for high-purity active pharmaceutical ingredients but also aligns with the global push towards green chemistry in fine chemical manufacturing. For R&D directors and procurement specialists, understanding the nuances of this microbial transformation is critical for optimizing supply chains and reducing the overall cost of goods sold for corticosteroid precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of oxygen functionality at the C7 position of steroid skeletons has been a formidable synthetic challenge, often relying on harsh chemical reagents that pose significant safety and environmental liabilities. Traditional chemical routes frequently employ toxic oxidants such as selenium dioxide or complex multi-step protection-deprotection sequences to achieve regioselectivity, which inevitably leads to lower overall yields and complicated purification processes. These conventional methods generate substantial amounts of hazardous waste, requiring expensive disposal protocols and rigorous safety monitoring that can drastically inflate production costs. Furthermore, chemical hydroxylation often lacks the precise stereoselectivity needed for pharmaceutical applications, resulting in difficult-to-separate mixtures of alpha and beta isomers that compromise the purity of the final API. The reliance on heavy metals and volatile organic solvents in these legacy processes creates a bottleneck for manufacturers aiming to meet increasingly stringent regulatory standards for residual impurities and environmental compliance.
The Novel Approach
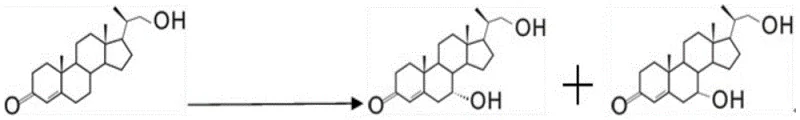
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated two-step fermentation strategy that harnesses the natural enzymatic machinery of mutated fungal strains to perform highly selective hydroxylation. By employing organisms such as Mucor circinelloides Lusitanicus and Acremonium erectum, the process achieves direct conversion of the substrate 4-HBC into both 7-alpha and 7-beta hydroxy isomers under mild aqueous conditions. This biological approach eliminates the need for toxic chemical oxidants and operates at moderate temperatures between 25°C and 35°C, significantly reducing energy consumption and operational hazards. The patent data highlights that under optimized substrate loading conditions of 10-20 g/L, the total yield of the desired products can reach impressive levels, demonstrating the robustness of the biocatalytic system. This shift to biocatalysis represents a paradigm change in steroid manufacturing, offering a cleaner, safer, and more economically viable route for producing high-value intermediates.
Mechanistic Insights into Microbial C7 Hydroxylation
The success of this synthesis relies on the specific activity of cytochrome P450 monooxygenases and other hydroxylase enzymes present within the fungal mycelium, which facilitate the insertion of an oxygen atom into the unactivated C-H bond at the 7-position. These enzymes exhibit remarkable regioselectivity, distinguishing between the chemically similar positions on the steroid ring system to target the C7 carbon exclusively while leaving other sensitive functional groups, such as the C3-ketone and C21-hydroxyl, intact. The mechanistic pathway likely involves the activation of molecular oxygen by the heme iron center of the enzyme, followed by hydrogen abstraction from the steroid substrate and subsequent radical rebound to form the hydroxyl group. This enzymatic precision ensures that the reaction proceeds with minimal formation of side products such as over-oxidized ketones or hydroxylations at unwanted positions like C11 or C14, which are common issues in less selective chemical processes. Understanding this biological mechanism allows process chemists to fine-tune fermentation parameters, such as pH and dissolved oxygen levels, to maximize the expression of these specific hydroxylases and drive the reaction equilibrium towards the desired products.
From an impurity control perspective, the microbial route offers a distinct advantage by producing a cleaner reaction profile that simplifies downstream processing. Unlike chemical methods that may generate a broad spectrum of structurally related byproducts requiring extensive chromatographic separation, the biotransformation primarily yields the two target isomers, 7-alpha-OH-4-HBC and 7-beta-OH-4-HBC. The patent details indicate that these isomers can be effectively separated using standard column chromatography techniques with common solvent systems like ethyl acetate and petroleum ether. The high specificity of the biological catalyst minimizes the formation of poly-hydroxylated impurities or degraded steroid fragments, thereby enhancing the overall purity of the crude product before final crystallization. This inherent selectivity reduces the burden on quality control laboratories and ensures that the final material meets the stringent specifications required for pharmaceutical grade intermediates, ultimately safeguarding the safety and efficacy of the downstream drug products.
How to Synthesize 7,21-Dihydroxy-20-Methylpregna-4-En-3-One Efficiently
The implementation of this microbial synthesis route requires careful attention to strain maintenance and fermentation kinetics to ensure consistent batch-to-batch reproducibility. The process begins with the preparation of a robust seed culture, where the selected fungal strain is grown in a nutrient-rich medium containing glucose and peptone to reach the logarithmic growth phase. Once the biomass is sufficient, the substrate 4-HBC is introduced into the fermentation broth at a controlled concentration, typically ranging from 12 to 19 g/L depending on the specific strain tolerance. Maintaining the correct temperature and agitation speed is crucial during the bioconversion phase to ensure adequate oxygen transfer, which is vital for the hydroxylation reaction to proceed efficiently. Detailed standardized synthetic steps see the guide below.
- Prepare seed cultures of specific strains such as Mucor circinelloides or Aspergillus fumigatus in a glucose-peptone medium at 28°C.
- Inoculate the fermentation medium with the seed culture and grow to the logarithmic phase before adding the substrate 4-HBC.
- Maintain bioconversion at 25-30°C for 60-80 hours, then extract products using ethyl acetate and separate isomers via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microbial fermentation technology translates into tangible strategic benefits that extend beyond simple technical feasibility. The elimination of hazardous chemical reagents and heavy metal catalysts fundamentally alters the cost structure of the manufacturing process by removing the need for expensive waste treatment and specialized containment infrastructure. This transition to a greener process not only mitigates regulatory risks associated with environmental compliance but also stabilizes the supply chain by reducing dependency on volatile raw material markets for toxic oxidants. Furthermore, the use of renewable biological catalysts ensures a more sustainable sourcing model, which is increasingly becoming a key criterion for multinational corporations when selecting long-term manufacturing partners. The ability to produce high-purity intermediates with reduced environmental impact enhances the brand value of the final pharmaceutical products and aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of expensive and toxic chemical oxidants with inexpensive fermentation media components like glucose and starch. By avoiding the use of selenium-based reagents or complex protecting group strategies, manufacturers can significantly reduce the cost of raw materials and the associated handling fees. Additionally, the simplified downstream processing required to isolate the products from the fermentation broth reduces solvent consumption and labor hours, leading to substantial operational savings. The high conversion rates reported in the patent data suggest that substrate utilization is efficient, minimizing the loss of valuable starting materials and further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: Biological processes are inherently scalable and can be adapted to various production volumes, from pilot plant studies to multi-ton commercial campaigns, ensuring a steady supply of critical intermediates. The robustness of the fungal strains described in the patent, which tolerate substrate concentrations up to 20 g/L, indicates a resilient process capable of withstanding minor fluctuations in operating conditions without catastrophic failure. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients. Moreover, the availability of multiple effective strains provides a backup strategy, allowing manufacturers to switch between different biological catalysts if necessary to maintain supply continuity in the face of unforeseen disruptions.
- Scalability and Environmental Compliance: The fermentation conditions outlined in the patent, such as moderate temperatures and near-neutral pH levels, are fully compatible with existing industrial bioreactor infrastructure, facilitating rapid scale-up without the need for capital-intensive equipment modifications. The aqueous nature of the reaction medium significantly reduces the generation of volatile organic compounds (VOCs) compared to traditional organic synthesis, simplifying exhaust gas treatment and lowering the carbon footprint of the facility. This alignment with green chemistry principles ensures long-term regulatory compliance and reduces the risk of production shutdowns due to environmental violations. The process generates biodegradable waste streams that are easier to treat than chemical sludge, further enhancing the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the microbial synthesis of 7-hydroxy steroid intermediates, based on the detailed experimental data provided in the patent documentation. These insights are designed to clarify the operational parameters and potential advantages for stakeholders evaluating this technology for integration into their supply chains. Understanding these specifics is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the primary advantages of using microbial fermentation over chemical synthesis for 7-position hydroxylation?
A: Microbial fermentation offers superior regioselectivity for the 7-alpha and 7-beta positions without the need for toxic oxidizing agents like selenium dioxide, resulting in a cleaner impurity profile and reduced environmental impact.
Q: Which specific microorganism strains are most effective for this conversion according to the patent data?
A: The patent identifies Mucor circinelloides, Acremonium erectum, Mucor racemosus, and Aspergillus fumigatus as highly effective strains, with Mucor circinelloides achieving total yields up to 85.32% under optimized conditions.
Q: Is this process scalable for industrial production of steroid intermediates?
A: Yes, the process utilizes standard fermentation parameters (pH 4.0-6.5, 25-35°C) and common substrates, making it highly adaptable for large-scale commercial manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7,21-Dihydroxy-20-Methylpregna-4-En-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of advanced steroid intermediates for the development of next-generation hormonal therapies. Our team of expert process chemists and fermentation engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical markets. We are committed to delivering materials that adhere to stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. By leveraging cutting-edge biocatalytic technologies like the one described in CN109680032B, we offer our partners a competitive edge through superior product quality and consistent performance.
We invite you to collaborate with us to optimize your steroid synthesis supply chain and achieve significant efficiency gains in your manufacturing operations. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our microbial routes can reduce your overall cost of goods. Please contact us today to request specific COA data and route feasibility assessments for 7,21-dihydroxy-20-methylpregna-4-en-3-one and related intermediates. Let us help you navigate the complexities of modern pharmaceutical manufacturing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
