Scalable Production of High-Purity Bile Acid Intermediates Using Mutagenic Strains
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for synthesizing complex steroid intermediates, particularly bile acid derivatives which are critical for metabolic disease treatments. Patent CN112899191B introduces a groundbreaking methodology that seamlessly integrates advanced microbial fermentation with precise chemical synthesis to produce high-value cholic acid compounds. This technical disclosure details the use of a specific mutagenic strain, Mycobacterium sp. NRRL B-3805 (preservation number CCTCC M2020987), to transform abundant plant-derived phytosterols into 4-cholenic acid-3-ketone, a pivotal intermediate. By shifting the raw material base from limited animal sources to renewable plant sterols, this innovation addresses critical supply chain vulnerabilities while adhering to green chemistry principles. The process not only simplifies the operational workflow but also significantly enhances the purity profile of the final products, making it an attractive solution for global API manufacturers seeking reliable partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the sourcing of bile acids and their derivatives has relied heavily on extraction from animal organs, a practice fraught with significant logistical and safety challenges. The availability of animal-derived raw materials is inherently limited by agricultural cycles and slaughter rates, creating volatile supply dynamics that can disrupt production schedules for downstream pharmaceutical applications. Moreover, biological extracts from animal sources carry the risk of containing unknown pollutants, viruses, or immunogenic proteins, necessitating rigorous and costly purification steps to ensure patient safety. The environmental footprint of animal extraction is also substantial, involving complex waste management protocols that do not align with modern sustainability goals. Consequently, the industry has long sought alternative synthetic routes, yet many existing chemical-only syntheses suffer from harsh reaction conditions, low stereoselectivity, and the generation of difficult-to-remove isomer impurities.
The Novel Approach
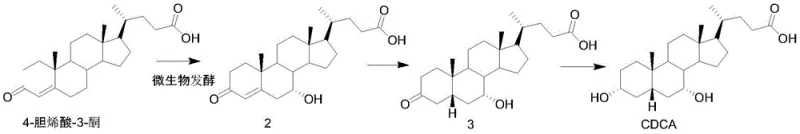
The methodology outlined in the patent represents a paradigm shift by leveraging the catalytic power of a specialized mutagenic microorganism to perform the initial structural modification of the steroid skeleton. By utilizing phytosterol, which is abundantly available in plant waste streams such as soybean oil deodorizer distillates, the process secures a stable and cost-effective raw material supply that is independent of animal agriculture. The biological fermentation step effectively constructs the necessary carbonyl side chain on the phytosterol backbone in a single, highly selective transformation, drastically reducing the number of synthetic steps required compared to total synthesis. Following fermentation, the subsequent chemical steps involve mild hydrogenation and reduction reactions that are easily controlled and scalable, ensuring high yields without the need for extreme temperatures or pressures. This hybrid bio-chemical strategy effectively bypasses the impurity profiles associated with animal extraction while avoiding the complexity of fully synthetic routes.
Mechanistic Insights into Microbial Side-Chain Cleavage and Chemical Reduction
The core of this technological advancement lies in the specific metabolic capabilities of the mutagenic strain CCTCC M2020987, which has been optimized through nitrosoguanidine treatment to enhance its biotransformation efficiency. This strain possesses the enzymatic machinery to selectively oxidize and cleave the side chain of various phytosterol analogues, converting them into 4-cholenic acid-3-ketone with high specificity. The fermentation process operates under controlled aerobic conditions, where parameters such as dissolved oxygen, pH, and agitation speed are meticulously managed to maximize the conversion rate of the substrate. The biological system effectively distinguishes between the steroid nucleus and the varying side chains of different phytosterols, funneling them into a common intermediate structure that serves as the foundation for further derivatization. This biological precision minimizes the formation of unwanted isomers, thereby simplifying the downstream purification burden and improving the overall mass balance of the process.
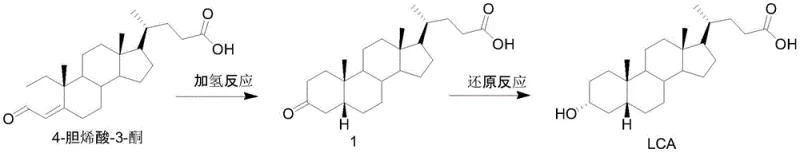
Following the fermentation stage, the chemical synthesis phase employs well-established catalytic hydrogenation and hydride reduction mechanisms to finalize the steroid structure. The hydrogenation step utilizes a palladium-carbon catalyst in the presence of a base to selectively reduce the double bond in the A-ring of the steroid nucleus without affecting other sensitive functional groups. Subsequently, a reduction reaction using sodium borohydride in a tetrahydrofuran and water solvent system converts the ketone functionality into the desired hydroxyl group with excellent stereocontrol. These chemical transformations are conducted under mild conditions, typically at ambient or slightly elevated temperatures, which preserves the integrity of the complex steroid skeleton. The combination of biological specificity and chemical reliability ensures that the final products, such as lithocholic acid and chenodeoxycholic acid, meet stringent purity specifications required for pharmaceutical applications.
How to Synthesize 4-Cholenic Acid-3-Ketone Efficiently
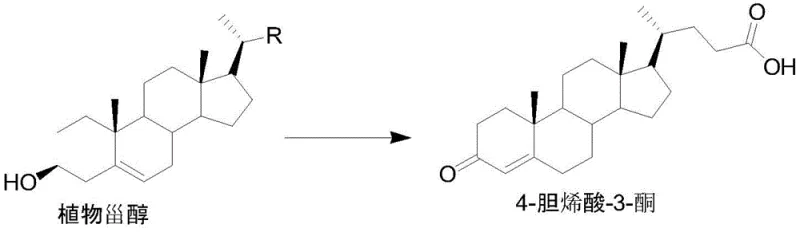
The synthesis protocol described in the patent provides a clear roadmap for producing 4-cholenic acid-3-ketone, starting from the preparation of the seed culture to the final isolation of the product. The process begins with the activation of the mutagenic strain in a nutrient-rich medium, followed by inoculation into a transformation medium containing the phytosterol substrate and a carbon source like soybean oil. The fermentation is allowed to proceed for a defined period, typically ranging from 96 to 144 hours, during which the microorganism metabolizes the sterol into the target ketone. Post-fermentation, the broth undergoes a series of extraction and purification steps involving pH adjustment, solvent extraction with methanol or ethyl acetate, and crystallization to isolate the pure intermediate. For a detailed, step-by-step guide including specific reagent quantities and equipment settings, please refer to the standardized synthesis instructions below.
- Ferment phytosterol using the mutagenic strain Mycobacterium sp. CCTCC M2020987 to obtain 4-cholenic acid-3-ketone.
- Perform a hydrogenation reaction on 4-cholenic acid-3-ketone using a palladium-carbon catalyst to generate Compound 1.
- Conduct a reduction reaction on Compound 1 using sodium borohydride in tetrahydrofuran and water to yield Lithocholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phytosterol-based fermentation route offers compelling strategic advantages over traditional sourcing models. The primary benefit stems from the decoupling of raw material supply from the volatile livestock market, as phytosterols can be sourced from stable, high-volume agricultural by-products that are consistently available globally. This shift not only mitigates the risk of supply interruptions due to animal disease outbreaks or seasonal fluctuations but also aligns corporate sourcing strategies with increasingly strict environmental, social, and governance (ESG) mandates. Furthermore, the simplified process flow reduces the dependency on exotic reagents and complex multi-step synthetic sequences, leading to a more resilient and transparent supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive animal-derived starting materials and the utilization of industrial waste residues as feedstocks fundamentally lowers the baseline cost of goods sold. By streamlining the synthesis into a efficient fermentation step followed by straightforward chemical conversions, the process reduces energy consumption and labor hours associated with complex purification protocols. The high selectivity of the biological transformation minimizes the loss of valuable material to side products, thereby improving overall yield and reducing the cost per kilogram of the active intermediate. Additionally, the mild reaction conditions decrease the wear and tear on reactor equipment and lower utility costs related to heating and cooling.
- Enhanced Supply Chain Reliability: Sourcing phytosterols from diverse agricultural streams ensures a continuous and predictable supply of raw materials, shielding production schedules from the unpredictability of biological extraction. The robustness of the mutagenic strain allows for consistent batch-to-batch performance, reducing the variability that often plagues biological processes and ensuring reliable delivery timelines for customers. The scalability of the fermentation process means that production capacity can be ramped up quickly to meet surges in demand without the long lead times associated with securing additional animal sources. This reliability is crucial for maintaining uninterrupted manufacturing of downstream pharmaceutical products.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard fermentation tanks and chemical reactors that are readily available in most cGMP facilities. The green nature of the synthesis, characterized by reduced organic solvent usage and the valorization of waste biomass, simplifies regulatory compliance regarding waste discharge and environmental impact. By avoiding the use of heavy metal catalysts in the biological step and minimizing hazardous waste generation in the chemical steps, the facility can operate with a lower environmental footprint. This sustainability profile not only reduces disposal costs but also enhances the brand reputation of the manufacturer among eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in an industrial setting. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation to ensure accuracy and relevance. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using phytosterol over animal extraction for bile acids?
A: Using phytosterol eliminates the risk of unknown pollutants often found in animal sources and overcomes the limited availability of biological raw materials. Furthermore, phytosterol can be sourced from industrial waste residues, making the process more sustainable and environmentally friendly.
Q: Which specific strain is used for the bioconversion in this patent?
A: The process utilizes a mutagenic strain of Mycobacterium sp. NRRL B-3805, specifically deposited under the preservation number CCTCC M2020987, which has been optimized for high-yield transformation of phytosterol.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial mass production, featuring mild reaction conditions, simple operation, and the use of easily obtained reagents, ensuring high reaction yields and scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Cholenic Acid-3-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of combining biocatalysis with traditional organic synthesis to create superior pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 4-cholenic acid-3-ketone and its derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch supplied adheres to the highest international standards, providing our partners with the confidence they need to advance their drug development programs.
We invite you to collaborate with us to optimize your supply chain and achieve significant cost efficiencies in your bile acid manufacturing processes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and comprehensive route feasibility assessments that demonstrate how our advanced fermentation technology can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
