Revolutionizing Benzoate Ester Production: A Deep Dive into Mild Catalytic Esterification for Industrial Scale-Up
Revolutionizing Benzoate Ester Production: A Deep Dive into Mild Catalytic Esterification for Industrial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, more efficient synthetic pathways that do not compromise on purity or yield. A pivotal advancement in this domain is detailed in patent CN109438270B, which discloses a robust preparation method for di(p-N,N-dialkylaminobenzoic acid)-N-alkyl diethanol amine esters. These compounds, particularly the dimethyl variant known as PC, serve as high-efficiency amine accelerators critical for the coatings, inks, and polymer industries, while also meeting stringent safety standards for food and cosmetic applications. The innovation lies in replacing hazardous acyl chloride intermediates with a direct esterification process mediated by specialized molecular sieve dehydrating agents. This shift not only mitigates the risks associated with handling corrosive acid chlorides but also resolves the persistent technical bottleneck of reactant salification, thereby enabling a smoother, more controllable industrial operation that aligns perfectly with modern environmental and safety regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex benzoate esters like the target amine accelerators has been plagued by significant operational challenges inherent to traditional acyl chloride chemistry. The conventional route typically involves reacting 4-N,N-dialkyl amino benzoyl chloride with N-alkyl diethanol amine in the presence of an organic base. However, a fundamental chemical incompatibility arises immediately upon mixing: the alkaline nature of the diethanol amine reacts instantly with the acyl chloride to form a salt. This salification phenomenon causes the reaction mixture to solidify or become highly viscous, drastically reducing the uniformity and fluidity of the system. Consequently, mass transfer is severely hindered, preventing the esterification from proceeding smoothly and leading to inconsistent batch quality and suboptimal yields. Alternatively, direct esterification using common strong acid dehydrating agents like sulfuric acid or p-toluenesulfonic acid often introduces severe side reactions and requires rigorous, costly neutralization and purification steps to remove acidic residues, making these methods less desirable for high-purity applications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes a sophisticated dehydration strategy driven by molecular sieves to facilitate direct esterification between 4-N,N-dialkylaminobenzoic acid and N-alkyl diethanol amine. By employing molecular sieves (Type A, X, or Y) as the dehydrating agent, the process effectively removes the water byproduct generated during esterification, shifting the chemical equilibrium towards the product side according to Le Chatelier's principle without the need for corrosive catalysts. This method operates under remarkably mild conditions, typically between 50-80°C, which significantly reduces energy consumption compared to high-temperature distillation methods. Furthermore, the process incorporates a refined post-treatment protocol involving specific solvent exchanges and controlled crystallization temperatures (25-40°C). This ensures that the final product precipitates with a qualified crystal form, effectively excluding impurities such as residual raw material acid and intermediate monoesters, thus achieving a purity level exceeding 99% with high conversion rates suitable for demanding industrial specifications.
Mechanistic Insights into Molecular Sieve-Catalyzed Esterification
The core mechanistic advantage of this synthesis lies in the physical-chemical interaction between the reaction byproducts and the porous structure of the molecular sieve catalyst. Unlike liquid acid catalysts that participate chemically and often degrade sensitive functional groups, molecular sieves act as selective adsorbents. As the carboxylic acid and amine react to form the ester bond, water is released. The molecular sieves, dispersed within the organic solvent matrix (such as dioxane or tetrahydrofuran), selectively trap these water molecules within their crystalline lattice pores. This continuous removal of water prevents the reverse hydrolysis reaction, driving the equilibrium forward to completion. The result is a high raw material conversion rate, consistently achieving between 90-95% efficiency. This mechanism is particularly crucial for bulky molecules like di(p-N,N-dialkylaminobenzoic acid) derivatives, where steric hindrance might otherwise slow down reaction kinetics. The mild thermal conditions preserve the integrity of the dialkylamino groups, preventing unwanted decomposition or quaternization that could occur under harsher acidic environments.
Controlling the impurity profile is another critical aspect of this mechanistic design, specifically addressing the challenge of monoester formation. In symmetric di-ester synthesis, there is always a risk of stopping at the mono-ester stage if the stoichiometry or reaction drive is insufficient. The patent describes a precise monitoring protocol using liquid chromatography to track the residual content of the starting benzoic acid, ensuring it drops below 0.2-1.0% before proceeding. Following the reaction, the strategic addition of a second organic solvent (like methanol or ethanol) during the cooling phase induces selective crystallization. This step is thermodynamically tuned to favor the precipitation of the di-ester while keeping mono-esters and unreacted amines in the solution phase. The subsequent washing of the filter cake with the same cold solvent further scrubs the surface of the crystals, removing adhered mother liquor and ensuring the final product meets the rigorous purity standards required for a reliable fine chemical intermediate supplier to deliver to top-tier pharmaceutical and coating manufacturers.
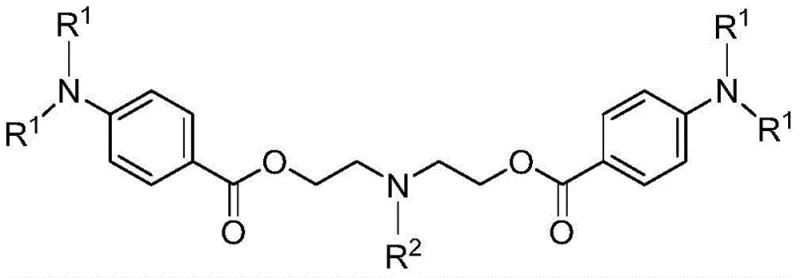
How to Synthesize Di(p-N,N-dialkylaminobenzoic acid)-N-alkyl diethanol amine ester Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature gradients to maximize yield and crystal quality. The process begins with the precise charging of 4-N,N-dialkylaminobenzoic acid and N-alkyl diethanol amine into a reactor containing a first organic solvent such as dioxane or toluene, followed by the addition of activated molecular sieves. The mixture is then heated to a moderate range of 50-80°C, where the esterification proceeds under stirring. Critical to success is the real-time monitoring of the reaction progress; operators must sample the system and analyze it via liquid chromatography to confirm that the residual acid content has fallen below the 1.0% threshold, indicating reaction completion. Once confirmed, the system is cooled, the molecular sieves are filtered off, and the filtrate undergoes desolventization. The resulting concentrate is then redissolved in a second solvent under nitrogen protection and slowly cooled to induce crystallization, followed by filtration and drying to obtain the final high-purity white powder. For the exact standardized operating procedures and specific parameter ranges, please refer to the detailed guide below.
- Mix 4-N,N-dialkylaminobenzoic acid, N-alkyl diethanol amine, a first organic solvent (e.g., dioxane), and a molecular sieve dehydrating agent (Type A, X, or Y).
- Heat the mixture to 50-80°C for esterification, monitoring residual acid content via liquid chromatography until it drops below 1.0%.
- Cool, filter out sieves, desolventize, add a second solvent (e.g., methanol) under nitrogen, and crystallize at 25-40°C to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this molecular sieve-mediated process represents a strategic opportunity to optimize both cost structures and supply reliability. Traditional acyl chloride routes are not only chemically problematic but also economically volatile due to the fluctuating costs and hazardous nature of acid chloride precursors. By switching to stable carboxylic acid raw materials, the supply chain becomes significantly more robust, as these acids are generally easier to source, store, and transport than their corrosive chloride counterparts. Furthermore, the elimination of strong mineral acids like sulfuric acid removes the need for extensive neutralization and wastewater treatment infrastructure, leading to substantial cost savings in waste management and environmental compliance. The mild reaction conditions also translate to lower energy expenditures, as the process does not require extreme heating or vacuum distillation, thereby reducing the overall carbon footprint and operational expenditure of the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this novel pathway are derived primarily from the simplification of the process flow and the utilization of cheaper, safer raw materials. By avoiding the use of acyl chlorides, manufacturers eliminate the costs associated with handling hazardous materials and the specialized equipment required for corrosion resistance. Additionally, the high conversion rate (90-95%) minimizes raw material waste, ensuring that a greater proportion of input mass is converted into saleable product. The absence of heavy metal catalysts or strong acids also means that downstream purification is less resource-intensive, avoiding the need for expensive chromatography columns or multiple recrystallization steps often required to remove metallic or acidic residues. This streamlined approach results in a leaner production cost model, allowing for more competitive pricing in the global market for polymer additives and fine chemical intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the stability of the starting materials offers a distinct advantage in maintaining continuous production schedules. Carboxylic acids and diethanol amines are stable solids or liquids with long shelf lives, unlike acyl chlorides which are moisture-sensitive and require strict anhydrous storage conditions. This stability reduces the risk of raw material degradation during transit or storage, ensuring consistent feedstock quality for every batch. Moreover, the process tolerance is higher; slight variations in temperature or mixing speeds do not lead to catastrophic failure or dangerous exotherms as seen in acyl chloride reactions. This operational robustness ensures that delivery timelines are met consistently, reducing the risk of stockouts for downstream customers who rely on these accelerators for their own paint and ink production lines.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is inherently safer and more straightforward due to the absence of aggressive reagents. The use of molecular sieves allows for a heterogeneous catalytic system that is easily separated by simple filtration, avoiding complex work-up procedures that often bottleneck scale-up efforts. Environmentally, the process generates significantly less hazardous waste; the spent molecular sieves can often be regenerated or disposed of more easily than acidic sludge. The solvent recovery systems can be optimized for the specific organic solvents used (e.g., methanol, dioxane), facilitating a closed-loop system that aligns with increasingly strict global environmental regulations. This makes the technology future-proof against tightening emission standards, securing the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this esterification technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on reaction mechanisms, purity profiles, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for large-scale production of amine accelerators and related benzoate derivatives. The answers reflect the specific advantages of the molecular sieve method over traditional acid-catalyzed or acyl chloride-based syntheses.
Q: Why are molecular sieves preferred over traditional acid catalysts for this synthesis?
A: Traditional strong acid catalysts like sulfuric acid often lead to side reactions and difficult purification. Molecular sieves act as a physical dehydrating agent, driving the equilibrium forward without introducing corrosive acidic impurities, resulting in higher purity (>99%) and milder reaction conditions.
Q: How does this method solve the salification issue found in acyl chloride routes?
A: Conventional acyl chloride routes suffer from immediate salification with alkaline amines, causing solidification and poor mixing. This direct esterification method bypasses the highly reactive acyl chloride intermediate entirely, maintaining a homogeneous liquid phase throughout the reaction for superior yield and operational smoothness.
Q: What represents the critical quality control point in this process?
A: The critical control point is monitoring the residual 4-N,N-dialkylaminobenzoic acid content via liquid chromatography. The reaction is only deemed complete when residual acid drops below 0.2-1.0%, ensuring minimal starting material carries over into the final crystallization step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Di(p-N,N-dialkylaminobenzoic acid)-N-alkyl diethanol amine ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced chemical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand the critical nature of purity in applications ranging from high-performance coatings to sensitive pharmaceutical formulations. Therefore, our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities at ppm levels. By leveraging the molecular sieve technology described in CN109438270B, we can offer a superior grade of benzoate esters that meet the highest international standards, providing our partners with a distinct competitive edge in their respective markets.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for these critical additives. Whether you are looking to secure a long-term supply of high-purity PC accelerators or need assistance in adapting this synthesis route for a specific derivative, our technical team is ready to support your goals. We encourage you to contact our Customized Cost-Saving Analysis team to discuss how switching to this greener, more efficient manufacturing process can benefit your bottom line. Reach out to our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements and quality specifications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
