Revolutionizing Pharmaceutical Intermediate Production via Iron-Catalyzed C-H Activation Technology
Introduction to Advanced Heterocyclic Synthesis
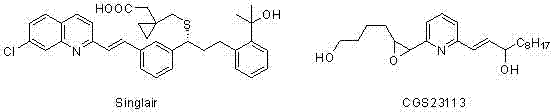
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable synthetic routes. A pivotal development in this sector is documented in patent CN102060639A, which discloses a highly efficient preparation method for nitrogen-containing heterocyclic olefin derivatives. These compounds serve as critical pharmacophores in a wide array of bioactive molecules, including well-known drugs such as Singulair (Montelukast) for asthma treatment and CGS23113. The structural complexity of these molecules, particularly the 2-alkenyl substitution pattern on quinoline, pyridine, or quinoxaline rings, has historically posed significant challenges for process chemists seeking reliable production pathways.
This patent introduces a transformative approach that leverages sp3 C—H activation and C—N bond cleavage to construct new C=C double bonds in a single step. By utilizing 2-alkyl substituted heterocycles as nucleophiles and imines as electrophiles under metal catalysis, this method bypasses many of the tedious multi-step sequences traditionally required. The implications for the supply chain of high-purity pharmaceutical intermediates are profound, offering a streamlined alternative that enhances both economic viability and environmental compliance. As we delve deeper into the technical specifics, it becomes clear why this technology represents a benchmark for modern fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
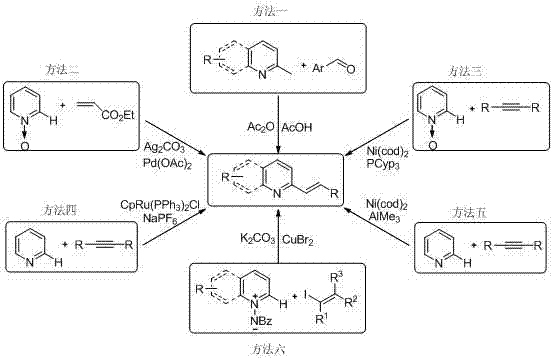
Prior to this innovation, the synthesis of 2-alkenyl quinoline and pyridine derivatives relied heavily on methodologies that were fraught with inefficiencies and economic burdens. As illustrated in the comparative analysis of existing techniques, early methods often utilized large excesses of acetic anhydride and acetic acid as both solvents and reactants. These processes typically required prolonged heating at elevated temperatures ranging from 120 to 160 degrees Celsius for up to five days, yet still resulted in disappointing yields of merely 10% to 30%. Such conditions are not only energy-intensive but also generate substantial chemical waste, complicating downstream purification and waste management protocols.
Furthermore, subsequent developments involving transition metal catalysis, while improving yields, introduced new bottlenecks related to cost and complexity. Methods employing palladium, ruthenium, or nickel catalysts often necessitated the use of expensive ligands such as PCyp3 or additives like NaPF6. Additionally, these routes frequently depended on costly starting materials like alkynes or iodoalkenes, which are less atom-economical and harder to source at scale compared to simple imines. The reliance on precious metals also raises concerns regarding residual metal contamination in the final API, requiring rigorous and expensive purification steps to meet stringent regulatory standards for pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN102060639A offers a streamlined, one-pot solution that dramatically simplifies the synthetic workflow. The core of this innovation lies in the direct coupling of readily available 2-alkyl heterocycles with imine derivatives using inexpensive iron-based catalysts. This approach eliminates the need for pre-functionalized substrates like alkynes or halides, thereby reducing the number of synthetic steps and the overall material cost. The reaction proceeds through a mechanism of sp3 C—H activation followed by C—N bond cleavage, efficiently constructing the desired carbon-carbon double bond with high selectivity.
The versatility of this method is evidenced by its broad substrate scope, accommodating various substituents on both the heterocyclic ring and the imine component. Whether utilizing electron-rich or electron-deficient groups, the system maintains robust performance, delivering high-purity products suitable for direct use in subsequent drug synthesis stages. By shifting from precious metal catalysts to abundant iron salts, this process not only lowers the direct cost of goods but also aligns with green chemistry principles by reducing the environmental footprint associated with heavy metal mining and disposal. This makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.

Mechanistic Insights into Iron-Catalyzed C-H Activation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific target molecules. The reaction initiates with the coordination of the iron catalyst to the nitrogen atom of the heterocyclic substrate, which activates the adjacent methyl or methylene group at the 2-position. This activation facilitates the abstraction of a proton, generating a nucleophilic carbon species capable of attacking the electrophilic imine. The subsequent C—N bond cleavage of the imine moiety is the key step that drives the formation of the new C=C double bond, releasing the amine byproduct and regenerating the catalytic species.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal reactions. The controlled nature of the metal-mediated activation minimizes side reactions such as polymerization or over-oxidation, which are common pitfalls in olefin synthesis. The use of mild Lewis acidic iron salts ensures that sensitive functional groups on the aromatic rings remain intact, preserving the integrity of the molecular scaffold. This high level of chemoselectivity is paramount for producing pharmaceutical intermediates where even trace impurities can impact the safety and efficacy of the final drug product, thereby reducing the burden on quality control laboratories.
How to Synthesize Quinoline Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The protocol generally involves mixing the 2-substituted pyridine or quinoline derivative with the imine electrophile in a suitable solvent such as toluene, nitromethane, or dichloromethane. An iron catalyst, typically ferrous acetate or ferric chloride, is added in catalytic amounts ranging from 1 to 10 mol%. The mixture is then heated under an inert argon atmosphere to temperatures between 25 and 200 degrees Celsius, depending on the reactivity of the specific substrates involved.
- Prepare the reaction mixture by combining 2-alkyl substituted quinoline or pyridine nucleophile with an imine electrophile under argon protection.
- Add an inexpensive iron-based metal catalyst, such as Fe(OAc)2 or FeCl3, to the solvent system like toluene or nitromethane.
- Heat the mixture to temperatures between 25-200°C for 2-72 hours, then purify the resulting olefin derivative via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology translates into tangible strategic benefits that extend beyond simple chemistry. The most immediate impact is seen in the cost structure of the manufacturing process. By replacing expensive palladium or ruthenium catalysts with commodity iron salts, the direct material costs are significantly reduced. Furthermore, the elimination of costly ligands and specialized additives simplifies the bill of materials, making the supply chain more resilient to fluctuations in the prices of rare earth metals and specialty chemicals.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzing the total cost of ownership. Traditional methods often incur hidden costs related to the removal of trace heavy metals from the final product, a requirement for FDA compliance. Since iron is biologically benign and easier to remove than precious metals, the downstream purification costs are drastically simplified. Additionally, the high atom economy of using imines instead of alkynes means less waste is generated per kilogram of product, leading to substantial cost savings in waste disposal and raw material consumption.
- Enhanced Supply Chain Reliability: Sourcing reliability is a critical metric for any procurement strategy. The starting materials for this reaction, specifically 2-methyl quinolines and various benzaldehyde-derived imines, are commodity chemicals available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks that often plague processes relying on proprietary ligands or exotic reagents. Consequently, lead times for high-purity pharmaceutical intermediates can be stabilized, ensuring consistent production schedules for downstream API manufacturing.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the ton-scale often reveals thermal and safety hazards that were not apparent in small batches. This iron-catalyzed method operates under relatively mild thermal conditions and does not require high-pressure equipment, making it inherently safer and easier to scale. From an environmental standpoint, the reduction in hazardous waste and the use of non-toxic iron catalysts facilitate easier compliance with increasingly stringent environmental regulations, thereby mitigating regulatory risk and enhancing the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled answers to common questions regarding its implementation. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios.
Q: What are the primary advantages of this iron-catalyzed method over traditional palladium-catalyzed routes?
A: The primary advantage is the drastic reduction in catalyst cost. Traditional methods often require expensive precious metals like Palladium, Ruthenium, or Nickel, along with costly ligands. This patent utilizes abundant and inexpensive iron salts, significantly lowering the raw material cost profile while maintaining high yields and selectivity.
Q: Can this methodology be applied to the synthesis of Montelukast (Singulair) intermediates?
A: Yes, the patent explicitly demonstrates the successful application of this technology to synthesize the key olefinic intermediate for Singulair (Montelukast). Example 82 in the patent data confirms the formation of the specific quinoline derivative required for this asthma medication with high efficiency.
Q: Does this process require harsh reaction conditions or specialized equipment?
A: No, the process is designed for operational simplicity. It operates under relatively mild temperatures (often around 120°C) and uses common organic solvents like toluene. The reaction can be performed in standard glassware under inert atmosphere without the need for high-pressure reactors or cryogenic conditions often associated with organometallic chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
The technological potential demonstrated in patent CN102060639A underscores the importance of having a manufacturing partner who understands both the chemistry and the complexities of industrial scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of heterocyclic olefin derivatives meets the exacting standards required by the global pharmaceutical industry.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in iron-catalyzed C-H activation can become a competitive advantage for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
