Advanced Synthesis of Novel Spiro-Expansion Monomers for High-Performance Polymer Additives
Advanced Synthesis of Novel Spiro-Expansion Monomers for High-Performance Polymer Additives
The development of advanced polymer additives capable of mitigating shrinkage during curing processes remains a critical frontier in materials science, particularly for high-performance coatings and composite materials. Patent CN112812124B introduces a groundbreaking class of novel spiro-expansion monomers that incorporate both unsaturated bonds and ester groups, addressing long-standing stability and reactivity issues found in traditional spiro orthocarbonates. These compounds are engineered to undergo volume expansion during polymerization, effectively counteracting the inherent shrinkage of resin systems and enhancing the mechanical integrity of the final product. As a reliable polymer additive supplier, understanding the nuanced chemistry behind these structures is essential for developing next-generation materials that meet rigorous industrial standards.
The core innovation lies in the molecular architecture, which features a robust spiro-bis-cyclic center flanked by polymerizable ester functionalities. This design not only facilitates free-radical polymerization but also ensures that the monomer remains stable under standard storage conditions, overcoming the thermal instability that has plagued earlier generations of expansion monomers. The versatility of the 'R' group allows for the customization of physical properties, enabling formulators to tailor the monomer for specific applications ranging from optical lenses to dental composites. By leveraging this patented technology, manufacturers can achieve superior dimensional stability without compromising on the processing window or shelf-life of their formulations.
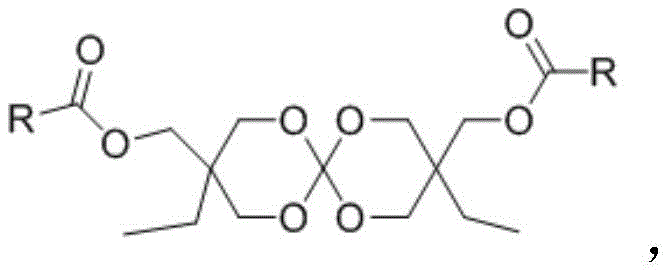
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the utilization of spiro orthocarbonates as expansion monomers has been hindered by significant practical drawbacks related to stability and reactivity. Many existing monomers in this category lack functional groups capable of participating in standard radical polymerization reactions, rendering them ineffective in common resin systems without complex modification. Furthermore, a critical deficiency in prior art is the thermal instability of these compounds; certain conventional spiro monomers require storage at temperatures below 10°C to prevent premature decomposition, imposing severe logistical burdens and increasing cold-chain transportation costs for supply chain managers. This instability often leads to inconsistent batch quality and limits the commercial viability of these additives in large-scale manufacturing environments where ambient temperature control is not always guaranteed.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by introducing ester-linked unsaturated groups directly onto the spiro framework, thereby creating a monomer that is both highly reactive and thermally robust. By incorporating groups such as methacrylate, acrylate, or fluorostyryl moieties, the new monomers can seamlessly integrate into existing radical polymerization workflows without the need for specialized initiators or conditions. Crucially, these novel compounds exhibit enhanced stability, remaining intact at temperatures up to 30°C, which drastically simplifies storage requirements to standard dry and ventilated conditions. This improvement not only reduces the risk of degradation during transit but also extends the usable shelf-life of the raw material, providing procurement teams with greater flexibility in inventory management and reducing waste associated with expired or compromised stock.
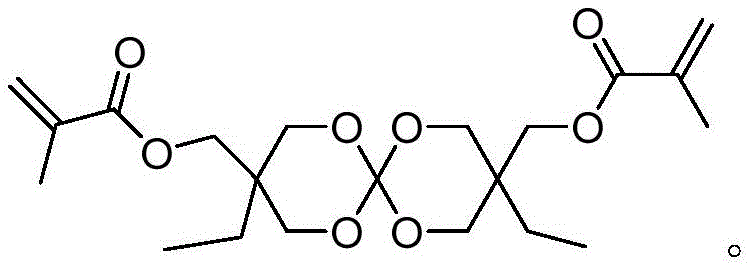
Mechanistic Insights into Spiro-Ring Formation and Esterification
The synthesis of these advanced monomers relies on a sophisticated yet operationally simple two-step mechanism that maximizes atom economy and yield. The first stage involves the acid-catalyzed condensation of trimethylolpropane (TMP) with tetraethyl orthocarbonate to form the key spiro-intermediate, 3,9-di(hydroxymethyl)-3,9-diethyl-1,5,7,11-tetraoxaspiro[5.5]undecane (DHOM). This cyclization reaction is driven by the removal of ethanol and requires precise control of acid catalysts, such as p-toluenesulfonic acid, to ensure the formation of the thermodynamically favored spiro-bicyclic structure over linear oligomers. The resulting intermediate possesses two primary hydroxyl groups positioned ideally for subsequent functionalization, serving as the foundational scaffold for the entire molecular architecture.
Following the isolation of the DHOM intermediate, the second stage employs a classic Schotten-Baumann type esterification to install the polymerizable unsaturated groups. By reacting the diol intermediate with unsaturated acyl chlorides, such as methacryloyl chloride or 4-fluorobenzeneacryloyl chloride, in the presence of an acid scavenger like triethylamine, the hydroxyl groups are efficiently converted into ester linkages. This step is critical for imparting the desired reactivity, as the resulting double bonds allow the monomer to copolymerize with standard acrylic or methacrylic resins. The reaction proceeds under mild conditions, typically between 0°C and 50°C, minimizing side reactions and ensuring high purity of the final product, which is essential for applications requiring optical clarity or high mechanical strength.
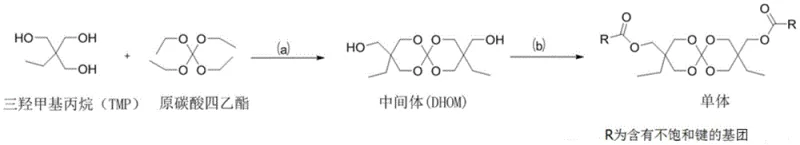
How to Synthesize Novel Spiro-Expansion Monomers Efficiently
The preparation of these high-value monomers follows a streamlined protocol designed for reproducibility and scalability, beginning with the careful selection of high-purity starting materials. The process initiates with the synthesis of the DHOM intermediate, where stoichiometric ratios of trimethylolpropane and tetraethyl orthocarbonate are reacted in a halogenated solvent under acidic catalysis, followed by a neutralization and crystallization step to isolate the solid intermediate. Subsequent esterification involves dissolving the purified intermediate in dichloromethane, cooling the solution to control exothermicity, and slowly adding the acyl chloride reagent while maintaining a basic environment to trap generated hydrogen chloride. Detailed standardized synthesis steps see the guide below.
- Synthesize the spiro intermediate (DHOM) by reacting trimethylolpropane with tetraethyl orthocarbonate using an acid catalyst.
- Purify the DHOM intermediate through washing, concentration, and crystallization using petroleum ether.
- Perform esterification by reacting the DHOM intermediate with unsaturated acyl chlorides (e.g., methacryloyl chloride) in the presence of triethylamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages for organizations seeking to optimize their supply chain resilience and reduce manufacturing overheads. The strategic shift towards using trimethylolpropane as a primary feedstock leverages a globally abundant and cost-effective commodity chemical, insulating production costs from the volatility associated with specialized fine chemical intermediates. By eliminating the need for expensive, pre-functionalized starting materials and reducing the number of synthetic steps, the overall cost of goods sold is significantly reduced, allowing for more competitive pricing strategies in the global polymer additives market without sacrificing margin.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by the substitution of costly precursors with readily available bulk chemicals like TMP and tetraethyl orthocarbonate. The elimination of transition metal catalysts and the use of simple acid catalysts remove the need for expensive heavy metal removal steps, which are often a bottleneck in pharmaceutical and electronic grade chemical production. Furthermore, the high yields reported in the patent examples indicate a minimization of raw material waste, directly translating to lower variable costs per kilogram of finished monomer and improved overall process economics.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the key raw materials are produced by multiple major chemical manufacturers worldwide, reducing dependency on single-source suppliers for niche intermediates. The stability of the final monomer at ambient temperatures (below 30°C) removes the necessity for energy-intensive refrigerated logistics, simplifying warehousing requirements and reducing the carbon footprint associated with cold-chain transportation. This robustness ensures consistent availability of the material for downstream customers, mitigating the risk of supply disruptions due to temperature excursions during shipping.
- Scalability and Environmental Compliance: The synthesis protocol is inherently scalable, utilizing standard unit operations such as liquid-liquid extraction, filtration, and crystallization that are easily adapted from laboratory to multi-ton reactor scales. The use of dichloromethane, while requiring proper handling, allows for efficient solvent recovery and recycling systems, aligning with modern environmental compliance standards. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a more sustainable manufacturing profile that appeals to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel spiro-expansion monomer technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear perspective on performance metrics and operational parameters. Understanding these details is crucial for R&D teams evaluating the material for new formulations and procurement officers assessing its integration into existing supply chains.
Q: What distinguishes this novel spiro monomer from existing spiro orthocarbonates?
A: Unlike traditional spiro orthocarbonates which may lack polymerizable groups or suffer from instability requiring deep-freeze storage, this novel monomer incorporates unsaturated ester groups. This modification enables effective radical polymerization while maintaining stability at temperatures below 30°C, significantly simplifying logistics and storage requirements.
Q: How does the new synthesis route impact production costs?
A: The process utilizes trimethylolpropane (TMP), a widely available and cost-effective commodity chemical, as the starting material rather than expensive pre-synthesized intermediates. By streamlining the synthesis into two efficient steps with standard solvents and mild conditions, the route eliminates complex purification stages, leading to substantial reductions in raw material and operational expenditures.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the patent explicitly highlights the method's suitability for industrial amplification. The reaction conditions are mild (0-50°C), utilize common solvents like dichloromethane, and involve straightforward workup procedures such as filtration and crystallization, making the transition from laboratory bench to multi-ton commercial production highly feasible and safe.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-Expansion Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in advancing the field of low-shrinkage polymer additives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of spiro-expansion monomer delivered meets the exacting standards required for high-performance applications in optics, electronics, and advanced composites.
We invite forward-thinking organizations to collaborate with us to leverage this technology for their specific product lines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current manufacturing setup, alongside specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of these critical monomers, driving innovation and efficiency in your polymer manufacturing processes while mitigating the risks associated with traditional expansion agents.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
