Revolutionizing 16 Beta Alkyl Steroid Production: High Selectivity and Commercial Scalability
Revolutionizing 16 Beta Alkyl Steroid Production: High Selectivity and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways for critical steroid intermediates, particularly those required for high-potency corticosteroids like betamethasone. Patent CN107746420B introduces a groundbreaking preparation method for 16 beta alkyl steroid compounds that addresses long-standing challenges in stereoselectivity and process efficiency. This technology leverages a unique three-step sequence involving carbonate-mediated activation, mild alkylation, and subsequent decarboxylation to achieve yields exceeding 85% with purity levels reaching 95%. For R&D directors and procurement strategists, this represents a significant leap forward, offering a reliable alternative to cumbersome traditional syntheses that often suffer from poor isomer control and excessive step counts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
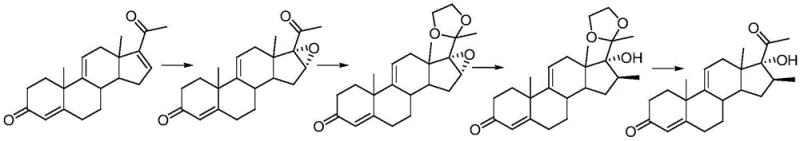
Historically, the synthesis of 16 beta alkyl steroids has been plagued by inefficiencies inherent in multi-step protection strategies. Traditional routes typically require the construction of a side chain at the 17-position followed by the introduction of an epoxy group at the 16,17-positions to facilitate methylation. This approach necessitates rigorous protection of the 21-position carbonyl, followed by difficult deprotection steps, resulting in a lengthy and costly operational timeline. Furthermore, these legacy methods frequently yield a problematic mixture of 16-alpha and 16-beta methyl isomers, necessitating complex and yield-loss-inducing purification processes to isolate the desired beta-configuration. The reliance on harsh conditions and multiple isolation steps not only inflates production costs but also creates significant supply chain bottlenecks due to extended cycle times.
The Novel Approach
In stark contrast, the methodology disclosed in CN107746420B streamlines the synthesis by eliminating the need for epoxy intermediates and extensive protecting group manipulations. By utilizing a first steroid compound and a carbonic ester, the process introduces an active 16-site alpha-formate group through a direct substitution reaction. This strategic activation allows the subsequent alkylation to proceed under remarkably mild conditions, avoiding the thermal stress that typically degrades sensitive steroid skeletons. The final hydrolysis decarboxylation step cleanly removes the activating formate group, delivering the target 16 beta alkyl steroid with exceptional stereochemical purity. This simplified workflow not only reduces the total number of unit operations but also significantly enhances the overall mass balance of the manufacturing process.
Mechanistic Insights into Carbonate-Mediated Activation and Alkylation
The core innovation of this technology lies in the electronic modulation of the steroid nucleus at the 16-position. The initial reaction employs a carbonate, such as diethyl carbonate, in the presence of a base like sodium hydride to substitute the 16-position hydrogen with an alpha-formate group. This formate moiety exerts a potent electron-withdrawing effect on the adjacent 17-position carbonyl, which in turn increases the acidity of the remaining 16-position proton. This activation is crucial as it enables the subsequent nucleophilic attack by halogenated hydrocarbons to occur at significantly lower temperatures, typically between -20°C and 30°C. Such mild conditions are instrumental in suppressing side reactions, such as double alkylation or elimination, which are common pitfalls in high-temperature steroid functionalization.
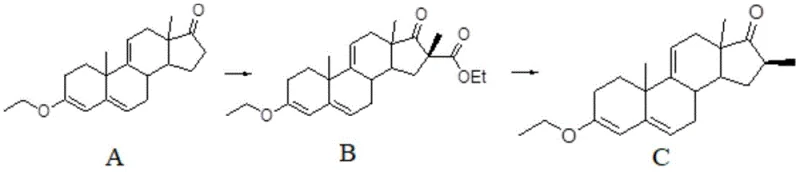
Furthermore, the stereochemical outcome is rigorously controlled by the spatial orientation of the intermediate species. The alpha-formate group serves as a temporary directing group that favors the approach of the alkylating agent from the beta-face, ensuring the formation of the thermodynamically and pharmacologically preferred 16-beta configuration. Following alkylation, the formate group is no longer needed and is efficiently removed via hydrolysis under either acidic or alkaline conditions. This decarboxylation step restores the 17-ketone functionality while retaining the newly installed beta-alkyl group. The mechanistic elegance of this route ensures that impurities related to 16-alpha isomers are minimized at the source, rather than being removed downstream, thereby simplifying the purification burden and enhancing the final product quality.
How to Synthesize 16 Beta Alkyl Steroid Efficiently
The practical implementation of this synthesis involves a sequential addition of reagents in a controlled inert atmosphere, typically nitrogen, to prevent oxidation of sensitive intermediates. The process begins with the formation of the enolate or activated species using a strong base, followed by the careful addition of the carbonate source. Once the substitution is confirmed, the alkylating agent is introduced directly into the reaction mixture, capitalizing on the activated state of the molecule.
- Perform 16-site substitution reaction between the first steroid compound and carbonic ester under alkaline conditions to introduce an active alpha-formate group.
- Conduct 16-site alkylation reaction on the intermediate using halogenated hydrocarbon under mild temperatures to install the alkyl group.
- Execute 16-position hydrolysis decarboxylation under acidic or alkaline conditions to remove the formate group and obtain the final 16 beta alkyl steroid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers tangible economic and operational benefits that extend beyond mere chemical yield. The ability to utilize widely available and cost-effective halogenated hydrocarbons, such as chloroalkanes or bromoalkanes, instead of expensive iodo-derivatives, directly impacts the raw material cost structure. Additionally, the reduction in synthetic steps eliminates the need for purchasing and managing inventories of complex protecting group reagents and the solvents required for their removal. This consolidation of the supply chain reduces the risk of delays caused by the shortage of niche reagents and simplifies the logistics of raw material sourcing for large-scale campaigns.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently drives down manufacturing costs by reducing solvent consumption, energy usage, and labor hours associated with multiple isolation steps. By avoiding the high-temperature conditions and prolonged reaction times typical of conventional routes, the facility can achieve higher throughput with existing reactor capacity. The elimination of expensive iodine-based reagents in favor of cheaper chlorine or bromine sources further contributes to a leaner cost of goods sold (COGS), making the final API intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures from -20°C to 60°C, ensures consistent batch-to-batch performance even when scaling up. This reliability is critical for maintaining continuous supply lines to downstream API manufacturers who cannot afford interruptions. Moreover, the high selectivity of the reaction means that less starting material is wasted on unwanted isomers, effectively increasing the yield per kilogram of input and stabilizing the supply volume against fluctuations in raw material availability.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the milder reaction parameters reduce the thermal load on plant infrastructure and minimize the generation of hazardous waste streams associated with aggressive reagents. The simplified workup procedure, which often involves straightforward precipitation and filtration, reduces the volume of organic waste solvents requiring treatment. This alignment with green chemistry principles facilitates easier regulatory compliance and supports the sustainability goals of modern pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced steroid synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making.
Q: How does this new method improve selectivity compared to traditional routes?
A: Traditional routes often result in a mixture of 16-alpha and 16-beta isomers with poor selectivity. This novel method utilizes a transient alpha-formate group that directs the subsequent alkylation specifically to the beta position, achieving selectivity up to 95% and significantly reducing impurity profiles.
Q: Can this process utilize cost-effective halogenated hydrocarbons?
A: Yes, unlike conventional methods that may require expensive iodo-hydrocarbons to drive the reaction, this process activates the 16-position sufficiently to allow the use of cheaper chloro- or bromo-alkanes, drastically reducing raw material costs without compromising yield.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Absolutely. The reaction conditions are mild (ranging from -20°C to 60°C) and avoid complex protection-deprotection sequences found in older methods. The simplified workup involving precipitation and filtration makes it highly amenable to commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16 Beta Alkyl Steroid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving corticosteroid therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 16 beta alkyl steroid conforms to the highest industry standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a successful and profitable partnership.
