Scalable Synthesis of High-Purity Pemedolac for Global Oncology Drug Manufacturing
Introduction to Advanced Pemedolac Manufacturing Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology intermediates, and the preparation of Pemedolac stands as a pivotal challenge in the supply chain for Pemetrexed Disodium. Patent CN102887902A introduces a groundbreaking preparation process that fundamentally addresses the historical bottlenecks of low yield and poor purity associated with traditional methods. This technology leverages a stable sulfonate intermediate to bypass the instability of aldehyde precursors, ensuring consistent quality from laboratory bench to commercial reactor. For R&D directors and procurement specialists, understanding this shift is crucial, as it represents a transition from fragile, low-yield chemistry to a resilient, high-efficiency manufacturing protocol. The innovation not only stabilizes the supply of this key building block but also aligns with stringent ICH guidelines regarding impurity control, specifically targeting single unknown impurities below 0.1%. By adopting this methodology, manufacturers can secure a reliable pemedolac supplier status, ensuring continuity for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pemedolac has been plagued by significant scalability issues, primarily stemming from the instability of key aldehyde intermediates generated during the Heck coupling and subsequent oxidation steps. In conventional routes, such as those disclosed in earlier patents, the isolation of 3-(4-methoxycarbonyl)phenyl-1-propionic aldehyde requires distillation, a unit operation that proves disastrous upon scale-up. During the concentration and distillation phases in large reactors, the aldehyde functionality is prone to self-condensation reactions, leading to the formation of high molecular weight byproducts that are difficult to remove. Data from pilot studies indicates that while laboratory yields might hover around 35%, amplification to production scale often sees total recovery plummet to approximately 26% due to these thermal instabilities. Furthermore, the reliance on strong bases for final purification in older methods introduces additional degradation pathways, resulting in product purity that struggles to exceed 90%, far below the requirements for modern oncology APIs. These technical deficiencies create substantial waste, increase raw material consumption, and jeopardize the consistency of the final drug substance.
The Novel Approach
The innovative process detailed in the patent circumvents these pitfalls by introducing a stable sodium sulfonate intermediate, effectively masking the reactive aldehyde group until it is needed for subsequent transformations. Instead of isolating the volatile and reactive aldehyde, the synthesis proceeds through a Heck reaction between methyl 4-bromobenzoate and allyl alcohol, followed immediately by the addition of sodium bisulfite. This generates 3-(4-methoxycarbonyl)phenyl-1-hydroxypropyl sodium sulfonate, a crystalline solid that is thermally stable and easy to handle. This strategic modification eliminates the need for hazardous distillation steps, thereby preventing the self-condensation that plagues traditional routes. The result is a dramatic improvement in process robustness, allowing for seamless translation from gram-scale experiments to multi-kilogram production batches without the characteristic yield collapse. This approach establishes a fundamental base for preparing high-purity Pemedolac, ensuring that the intermediate quality supports the rigorous demands of the final Pemetrexed Disodium API.

Mechanistic Insights into Bisulfite Protection and Weak Base Purification
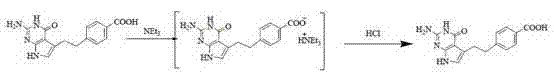
The core chemical innovation lies in the manipulation of the carbonyl equivalent through bisulfite adduct formation, which serves as a protecting group strategy that doubles as a purification handle. Mechanistically, the nucleophilic addition of the bisulfite ion to the transient aldehyde generates a water-soluble sulfonate salt that resists thermal degradation. This allows the intermediate to be purified via crystallization from aqueous systems, effectively washing away palladium catalysts, phase transfer catalysts, and organic side products that typically persist in the organic phase. When the synthesis proceeds to the Henry reaction stage, the sulfonate is cleaved under mild conditions using trimethylchlorosilane, releasing the reactive species in situ for condensation with nitromethane. This controlled release ensures that the concentration of free aldehyde remains low, further suppressing dimerization kinetics. Additionally, the final purification stage employs a weak organic base, specifically triethylamine, to form a salt with the crude Pemedolac. Unlike strong inorganic bases like sodium hydroxide, which can induce hydrolysis or structural rearrangement leading to impurity levels around 5%, the triethylamine salt is chemically benign. This selective salification allows for the precipitation of the product while leaving acidic and neutral impurities in the mother liquor, achieving a purity leap to over 99.5%.
Impurity control is further enhanced by the avoidance of harsh reaction conditions throughout the synthetic sequence. The use of mild solvents like ethyl acetate and acetonitrile, which are easily recycled, minimizes the introduction of solvent-related contaminants. The elimination of distillation not only saves energy but also removes a major vector for thermal stress-induced impurities. In the cyclization step involving 2,4-diamino-6-hydroxypyrimidine, the pre-purified intermediates ensure that the heterocyclic ring closure proceeds cleanly, minimizing the formation of regioisomers or open-chain byproducts. The final Nef reaction and ring closure are conducted under controlled pH conditions, where the initial formation of the triethylamine salt acts as a buffer against localized high pH zones that could degrade the sensitive pyrrolo-pyrimidine core. This comprehensive approach to impurity management ensures that single unknown impurities are consistently maintained below 0.1%, meeting the strictest international regulatory standards for pharmaceutical intermediates.
How to Synthesize Pemedolac Efficiently
The synthesis of high-purity Pemedolac via this novel route involves a sequence of well-defined chemical transformations that prioritize stability and yield. The process begins with the formation of the stable sulfonate intermediate, followed by deprotection, nitro-aldol condensation, elimination, and heterocyclic cyclization. Each step is optimized to minimize work-up complexity and maximize material throughput. Detailed standard operating procedures for each reaction stage, including specific molar ratios, temperature profiles, and quenching protocols, are essential for replicating the high success rates reported in the patent literature. Manufacturers aiming to implement this technology should focus on the precise control of the bisulfite addition step and the subsequent crystallization conditions, as these are critical for removing heavy metal residues early in the sequence. The standardized synthesis steps outlined below provide a roadmap for achieving the documented efficiency gains.
- Perform Heck coupling of methyl 4-bromobenzoate with allyl alcohol, followed immediately by sodium bisulfite addition to generate stable intermediate A1.
- Deprotect A1 using trimethylchlorosilane and conduct Henry reaction with nitromethane to form nitro-alcohol A2.
- Execute elimination reaction on A2 using methanesulfonyl chloride and triethylamine to yield nitro-alkene A3.
- React A3 with 2,4-diamino-6-hydroxypyrimidine to cyclize and form intermediate A4.
- Conduct Nef reaction and ring closure using potassium hydroxide, followed by acidification to obtain crude Pemedolac.
- Purify crude product by forming a triethylamine salt, filtering, and regenerating the free acid with hydrochloric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits regarding cost structure and supply reliability. The primary advantage stems from the substantial increase in total process yield, which directly reduces the cost of goods sold by minimizing raw material waste. By eliminating the yield losses associated with aldehyde distillation and condensation, the process ensures that a significantly higher proportion of starting materials are converted into saleable product. This efficiency gain translates into a more competitive pricing model for the intermediate, allowing downstream API manufacturers to optimize their own production costs. Furthermore, the use of recyclable solvents and the avoidance of energy-intensive distillation steps contribute to a lower environmental footprint, aligning with modern sustainability goals and reducing waste disposal expenses. The robustness of the process also means fewer failed batches, ensuring a steady flow of material to meet production schedules.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the drastic reduction in material loss lead to significant economic benefits. By avoiding the need to discard large fractions of product due to thermal degradation, the overall consumption of expensive starting materials like halobenzene derivatives and specialized catalysts is optimized. The ability to recycle solvents such as ethyl acetate further lowers operational expenditures, making the process economically superior to legacy methods that rely on volatile aldehydes. This cost efficiency is critical for maintaining margins in the competitive generic oncology market.
- Enhanced Supply Chain Reliability: The stability of the sulfonate intermediate removes the risk of batch-to-batch variability caused by thermal sensitivity. Traditional methods often suffer from unpredictable yields during scale-up, leading to supply shortages and delayed timelines. In contrast, this novel route offers predictable kinetics and consistent output, enabling manufacturers to commit to firm delivery schedules. The simplified workflow also reduces the dependency on specialized equipment for high-vacuum distillation, allowing production to be flexible across different manufacturing sites. This reliability is paramount for securing long-term contracts with major pharmaceutical companies.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing mild conditions that are easier to manage in large reactors. The replacement of toxic solvents like methylene chloride with greener alternatives like ethyl acetate simplifies regulatory compliance and worker safety protocols. Additionally, the reduction in waste generation through higher yields and solvent recycling supports corporate sustainability initiatives. The straightforward crystallization-based purifications reduce the load on wastewater treatment facilities, ensuring that the manufacturing process remains environmentally compliant and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Pemedolac synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing clarity on the mechanistic advantages and practical benefits of the new route. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The answers highlight the specific solutions to historical pain points such as yield loss and impurity control.
Q: Why is the sodium bisulfite intermediate critical for scaling Pemedolac production?
A: Traditional routes rely on isolating a reactive aldehyde intermediate which undergoes self-condensation during distillation, causing severe yield loss at scale. The novel sulfonate intermediate is stable, eliminating the need for distillation and preventing impurity formation.
Q: How does the new purification method achieve >99.5% purity?
A: Conventional purification uses strong bases like NaOH which degrade the product and generate impurities. This process utilizes a weak organic base (triethylamine) to form a stable salt, effectively separating impurities without degrading the Pemedolac structure.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process was specifically designed to solve scale-up issues. By avoiding unstable intermediates and harsh distillation steps, the total recovery increases significantly from roughly 27% to over 65%, making it highly viable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemedolac Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against cancer. Our technical team has extensively analyzed the pathway described in CN102887902A and possesses the expertise to execute this complex synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in practice. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical single impurity thresholds required for oncology drugs. We are committed to delivering Pemedolac that not only meets but exceeds the quality benchmarks necessary for the synthesis of Pemetrexed Disodium.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this technology can optimize your supply chain. By leveraging our manufacturing capabilities, you can secure a stable source of high-purity intermediates that drive efficiency in your final API production. Contact us today to discuss how we can support your long-term strategic goals in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
