Advanced Synthesis of N-Heteroaryl-N-Aryl Amines for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Diarylamine Synthesis
The pharmaceutical industry continuously seeks more efficient pathways to construct complex molecular architectures, particularly for kinase inhibitors where structural precision is paramount. Patent CN101723891A introduces a transformative methodology for the preparation of N-heteroaryl-N-aryl-amines, a critical scaffold found in numerous potent p38 MAPK inhibitors. This technology addresses the longstanding challenge of synthesizing unsymmetrical diarylamines with high fidelity, bypassing the notorious issue of over-arylation that plagues conventional direct coupling strategies. By employing a strategic protection-deprotection sequence using N-aryl carbamic acid esters, the process ensures that the amine functionality reacts selectively, yielding products of exceptional purity. 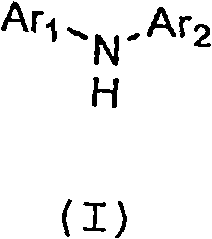 The significance of this invention extends beyond mere academic interest; it offers a practical solution for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines. The ability to generate these scaffolds without excessive byproducts translates directly into reduced downstream processing costs and improved overall process mass intensity. As we delve deeper into the technical specifics, it becomes evident that this approach represents a substantial leap forward in the cost reduction in API manufacturing, particularly for anti-inflammatory agents targeting the p38 pathway.
The significance of this invention extends beyond mere academic interest; it offers a practical solution for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines. The ability to generate these scaffolds without excessive byproducts translates directly into reduced downstream processing costs and improved overall process mass intensity. As we delve deeper into the technical specifics, it becomes evident that this approach represents a substantial leap forward in the cost reduction in API manufacturing, particularly for anti-inflammatory agents targeting the p38 pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of diarylamines has relied heavily on palladium-catalyzed coupling reactions between aryl halides and primary amines. While effective in many contexts, this classical approach suffers from a critical flaw when applied to the synthesis of specific unsymmetrical targets: the propensity for over-arylation. Once the primary amine couples with the first aryl halide, the resulting secondary amine often retains sufficient nucleophilicity to react with a second equivalent of the electrophile. This leads to the formation of tertiary amine impurities that are structurally similar to the desired product, making them notoriously difficult to separate via crystallization or chromatography. Furthermore, traditional methods sometimes necessitate the use of toxic tin reagents or require harsh conditions that compromise sensitive functional groups elsewhere on the molecule. These limitations create significant bottlenecks in the commercial scale-up of complex heterocyclic amines, forcing manufacturers to accept lower yields or invest heavily in complex purification trains.
The Novel Approach
The methodology disclosed in CN101723891A elegantly circumvents these pitfalls by temporarily masking the nucleophilicity of the amine nitrogen. By converting the aniline starting material into an N-aryl carbamic acid ester, the nitrogen atom is rendered less nucleophilic after the initial coupling event, effectively acting as a 'dual-character' species that reacts once and then stops. This strategic modification allows for the use of mono-amine derivatives without the fear of generating di-arylated byproducts. The process is remarkably flexible, accommodating both transition metal-catalyzed conditions and base-mediated nucleophilic aromatic substitution. 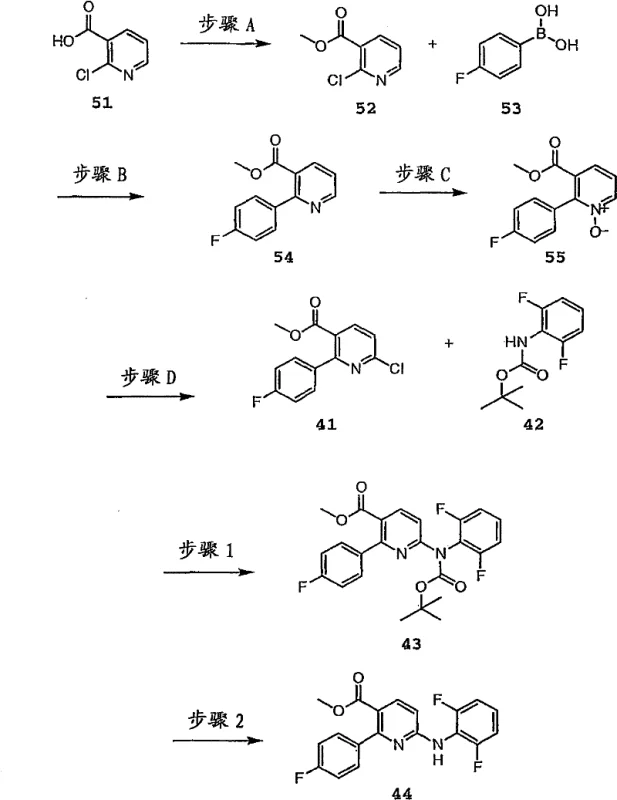 This versatility means that the route can be adapted based on the electronic nature of the substrates, ensuring high-purity diarylamine intermediates are accessible regardless of the specific substitution pattern. The result is a streamlined workflow that eliminates the need for excessive equivalents of expensive aryl halides and simplifies the isolation of the final active pharmaceutical ingredient.
This versatility means that the route can be adapted based on the electronic nature of the substrates, ensuring high-purity diarylamine intermediates are accessible regardless of the specific substitution pattern. The result is a streamlined workflow that eliminates the need for excessive equivalents of expensive aryl halides and simplifies the isolation of the final active pharmaceutical ingredient.
Mechanistic Insights into Protected Amine Coupling
The core of this innovation lies in the controlled reactivity of the protected amine species. In the first stage of the synthesis, a halo-heteroaryl compound, such as a chloro-pyridine derivative, is reacted with the N-aryl carbamate. This coupling can proceed via two distinct mechanistic pathways depending on the substrate activation. For electron-deficient heteroaryls, a direct nucleophilic aromatic substitution mediated by strong bases like cesium carbonate in polar aprotic solvents like NMP is highly effective. Alternatively, for less activated systems, a palladium-catalyzed cross-coupling utilizing ligands such as BINAP or dppf facilitates the bond formation under milder thermal conditions. The choice of base, whether it be potassium phosphate or cesium carbonate, plays a pivotal role in driving the equilibrium towards the product while neutralizing the acid byproduct generated during the substitution. 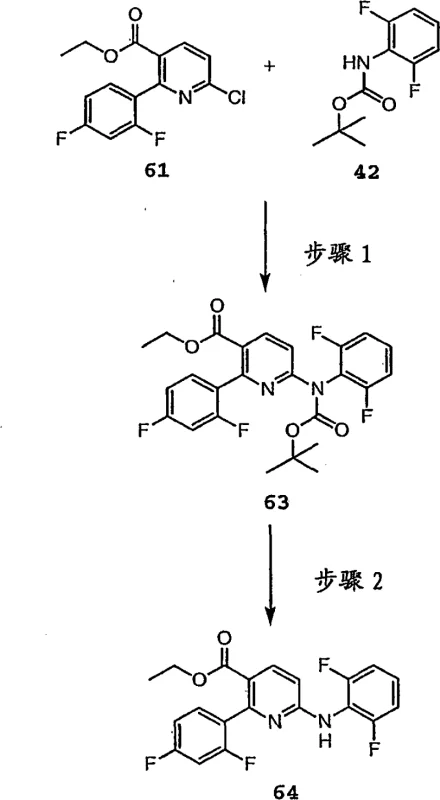 Following the coupling, the second critical step involves the removal of the carbamate protecting group. This is typically achieved using strong acids like trifluoroacetic acid (TFA) in chlorinated solvents. The acidolysis cleaves the carbamate bond cleanly, regenerating the free secondary amine without affecting other sensitive moieties such as esters or halogens on the aromatic rings. This two-step sequence ensures that impurity profiles are tightly controlled, as the intermediate protected amine can often be purified before the final deprotection, guaranteeing the quality of the final output.
Following the coupling, the second critical step involves the removal of the carbamate protecting group. This is typically achieved using strong acids like trifluoroacetic acid (TFA) in chlorinated solvents. The acidolysis cleaves the carbamate bond cleanly, regenerating the free secondary amine without affecting other sensitive moieties such as esters or halogens on the aromatic rings. This two-step sequence ensures that impurity profiles are tightly controlled, as the intermediate protected amine can often be purified before the final deprotection, guaranteeing the quality of the final output.
How to Synthesize N-Heteroaryl-N-Aryl Amines Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and minimize side reactions. The process begins with the preparation of the halo-heteroaryl electrophile, often involving the activation of a pyridine N-oxide followed by halogenation to install the leaving group at the desired position. Once the electrophile and the protected amine are prepared, they are combined under the chosen catalytic or basic conditions. Detailed operational guidelines regarding stoichiometry, temperature ramps, and workup procedures are essential for reproducibility. For a comprehensive breakdown of the specific experimental conditions and reagent grades required for optimal performance, please refer to the standardized protocol below.
- Couple a halo-heteroaryl compound with an N-aryl carbamic acid ester using either a transition metal catalyst like palladium with phosphine ligands or an alkali metal salt such as cesium carbonate in a polar solvent.
- Isolate the protected N-heteroaryl-N-aryl-amine intermediate, ensuring the removal of any unreacted starting materials through standard aqueous workup procedures.
- Remove the carbamate protecting group by treating the intermediate with a strong acid like trifluoroacetic acid in a chlorinated solvent to generate the final free amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary advantage lies in the drastic simplification of the purification process. By virtually eliminating the formation of over-arylated impurities, the need for resource-intensive chromatographic separations is removed, allowing for simpler crystallization-based purifications. This shift significantly reduces the consumption of solvents and silica gel, leading to substantial cost savings in manufacturing. Furthermore, the reagents employed in this process, such as cesium carbonate, potassium phosphate, and common palladium catalysts, are readily available from global chemical suppliers, ensuring reducing lead time for high-purity kinase inhibitors. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of over-arylation byproducts means that the yield of the desired product is maximized relative to the starting materials. In traditional processes, a significant portion of the expensive aryl halide is wasted in forming useless tertiary amine impurities. By preventing this waste, the effective cost per kilogram of the final intermediate is lowered. Additionally, the avoidance of toxic tin reagents removes the costly and environmentally burdensome step of heavy metal scavenging, further optimizing the production budget. The ability to use simpler solvents like toluene or NMP, which are easier to recover and recycle than specialized fluorinated solvents, also contributes to a leaner cost structure.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than bespoke, hard-to-source reagents strengthens the supply chain. Cesium carbonate and standard palladium sources are produced at scale by multiple vendors globally, mitigating the risk of single-source bottlenecks. The process tolerance for various substituents on the aromatic rings allows for flexibility in sourcing different grades of starting anilines or pyridines without compromising the reaction outcome. This flexibility is crucial for maintaining continuous production schedules in the face of market fluctuations or raw material shortages, ensuring a steady flow of intermediates to downstream formulation teams.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, having been demonstrated to work effectively from gram to multi-kilogram scales without loss of efficiency. The use of aqueous workups and standard organic solvents aligns well with existing industrial infrastructure, requiring no specialized high-pressure or cryogenic equipment. From an environmental standpoint, the atom economy is superior to traditional methods due to the higher selectivity, resulting in less chemical waste generation. The absence of tin byproducts simplifies wastewater treatment and disposal, helping facilities meet stringent environmental regulations and sustainability goals with greater ease.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. Understanding these nuances is vital for R&D teams evaluating the feasibility of integrating this route into their existing portfolios. The answers provided are derived directly from the experimental data and claims within the patent specification, ensuring accuracy and relevance for process development scientists.
Q: How does this process prevent over-arylation impurities?
A: By utilizing an N-aryl carbamic acid ester, the nitrogen atom is temporarily protected, reducing its nucleophilicity after the first coupling event and effectively preventing the formation of di-arylated byproducts common in traditional methods.
Q: What catalysts are suitable for this coupling reaction?
A: The process is versatile, supporting both transition metal catalysis using palladium complexes with ligands like BINAP and base-mediated coupling using alkali metal salts such as cesium carbonate or potassium phosphate.
Q: Is this method scalable for commercial production?
A: Yes, the method employs robust reagents and standard solvents like toluene and NMP, avoiding hazardous tin compounds and facilitating straightforward purification, which makes it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Heteroaryl-N-Aryl Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether your project requires custom synthesis of complex heterocycles or the optimization of an existing pathway, our technical expertise ensures that your supply chain remains uninterrupted and compliant with global standards.
We invite you to collaborate with us to leverage these advanced synthetic methodologies for your upcoming projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how our capabilities can accelerate your drug development timeline. Let us help you navigate the complexities of fine chemical manufacturing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
