Advanced Synthesis of Deuterated 2-Phenylthiazole Compounds for Gout Therapy
Introduction to Next-Generation Gout Therapeutics
The pharmaceutical landscape for treating hyperuricemia and gout is undergoing a significant transformation driven by the need for drugs with superior metabolic profiles. Patent CN103848796A introduces a novel class of deuterated 2-phenylthiazole compounds, which serve as potent xanthine oxidase inhibitors. Unlike traditional small molecules, these deuterated analogs leverage the kinetic isotope effect to enhance metabolic stability without compromising biological efficacy. By strategically replacing specific hydrogen atoms with deuterium on the phenyl ring or the alkyl side chains, the resulting compounds exhibit resistance to rapid oxidative metabolism by cytochrome P450 enzymes. This technological advancement addresses critical limitations of existing therapies, such as the need for frequent dosing and potential toxicity associated with metabolite accumulation. For R&D directors and procurement specialists, understanding the synthesis and supply chain implications of these high-value intermediates is essential for securing a competitive edge in the development of next-generation urate-lowering therapies.
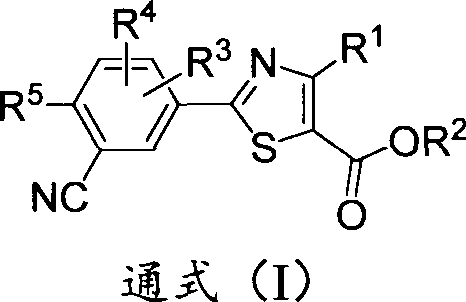
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of 2-phenylthiazole derivatives, such as the widely used drug Febuxostat, typically relies on standard organic transformations that do not account for isotopic modification. Conventional routes often involve the direct coupling of non-deuterated aryl halides with thiazole precursors, resulting in products that are susceptible to rapid hepatic clearance. The carbon-hydrogen bonds in these structures are prone to oxidative cleavage, leading to shorter plasma half-lives and the generation of potentially reactive metabolites. Furthermore, standard manufacturing processes may lack the specificity required to introduce isotopic labels at later stages without extensive re-optimization of reaction conditions. This limitation forces pharmaceutical developers to accept suboptimal pharmacokinetic parameters or invest heavily in complex late-stage functionalization strategies that are difficult to scale. Consequently, the supply chain for non-deuterated intermediates is mature but offers little room for innovation in terms of drug performance enhancement.
The Novel Approach
The innovative methodology described in the patent circumvents these issues by integrating deuteration directly into the core synthetic strategy. Instead of treating deuteration as an afterthought, the process utilizes deuterated starting materials, such as deuterated phenylboronic acids, or employs catalytic hydrogen-isotope exchange on activated intermediates. This approach ensures high isotopic purity and precise positioning of the deuterium atoms, which is critical for maximizing the metabolic benefit. The use of palladium-catalyzed cross-coupling reactions allows for the efficient construction of the biaryl bond under mild conditions, preserving the integrity of the sensitive nitrile and ester functionalities present in the molecule. By shifting the synthesis towards these specialized intermediates, manufacturers can produce drug substances that offer improved patient compliance and safety profiles. This strategic pivot not only enhances the therapeutic value of the final API but also creates a differentiated supply chain niche for suppliers capable of executing these sophisticated chemical transformations reliably.
Mechanistic Insights into Pd-Catalyzed Deuteration and Coupling
The core of this synthetic technology lies in the versatile application of palladium-catalyzed reactions to install the deuterated aryl moiety. One primary pathway involves the activation of a hydroxyl-substituted thiazole intermediate via conversion to a triflate using trifluoromethanesulfonic anhydride. This triflate serves as an excellent electrophile in Suzuki-Miyaura coupling reactions. When reacted with deuterated phenylboronic acids in the presence of a palladium catalyst like tetrakis(triphenylphosphine)palladium(0) and a base such as potassium carbonate, the carbon-carbon bond is formed with high fidelity. The mechanism proceeds through the classic oxidative addition of the palladium species into the carbon-oxygen bond of the triflate, followed by transmetallation with the boronic acid and reductive elimination to release the coupled product. This route is particularly advantageous for introducing perdeuterated phenyl rings, as the isotopic label is pre-installed on the boronic acid reagent, minimizing the risk of isotopic scrambling during the reaction.
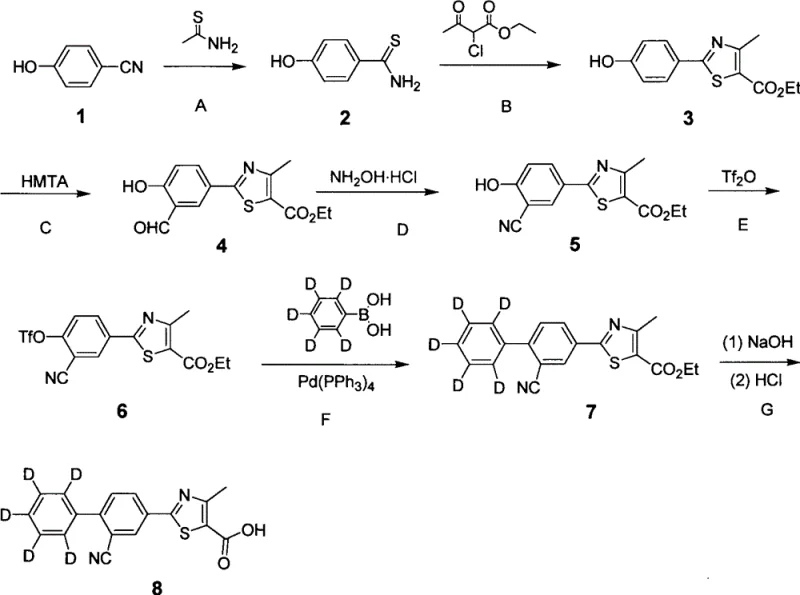
An alternative mechanistic pathway demonstrated in the patent involves direct hydrogen-isotope exchange on the aromatic ring. In this strategy, a brominated precursor is subjected to deuterium gas in the presence of a heterogeneous palladium catalyst, such as palladium on carbon. The mechanism involves the adsorption of the substrate onto the catalyst surface, where oxidative addition of the carbon-bromine bond occurs. Subsequent exposure to deuterium gas leads to the formation of a carbon-deuterium bond upon reductive elimination, effectively replacing the bromine atom with deuterium. Additionally, under these conditions, ortho-metalation can facilitate the exchange of adjacent hydrogen atoms for deuterium, allowing for multi-site labeling. This method is highly efficient for introducing deuterium at specific positions without the need for expensive deuterated coupling partners. Understanding these mechanistic nuances is vital for process chemists aiming to optimize yield and isotopic enrichment levels during scale-up, ensuring that the final intermediate meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Deuterated 2-Phenylthiazole Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction control and purification. The process begins with the construction of the thiazole scaffold, typically achieved by condensing a thioamide with an alpha-halo ketone, followed by formylation to introduce the aldehyde group necessary for subsequent cyanation. Once the core structure is established, the critical deuteration step is executed using one of the previously discussed palladium-catalyzed methods. Careful attention must be paid to the exclusion of moisture and oxygen during the coupling or exchange steps to prevent catalyst deactivation and side reactions. Following the reaction, the crude product is purified using standard chromatographic techniques, such as silica gel column chromatography, to remove residual catalysts and unreacted starting materials. The final hydrolysis of the ester group yields the free carboxylic acid, which is the active pharmaceutical intermediate. Detailed standardized synthesis steps see the guide below.
- Preparation of the thiazole core involves cyclization of thioamides with alpha-halo ketones followed by formylation and cyanation to establish the key nitrile functionality.
- Activation of the phenolic hydroxyl group is achieved using trifluoromethanesulfonic anhydride to create a highly reactive triflate leaving group for subsequent coupling.
- Final deuteration is accomplished either through Suzuki-Miyaura coupling with deuterated boronic acids or catalytic hydrogen-isotope exchange using deuterium gas and palladium on carbon.
Commercial Advantages for Procurement and Supply Chain Teams
Integrating deuterated intermediates into the supply chain offers substantial strategic benefits for pharmaceutical manufacturers seeking to extend product lifecycles and improve market positioning. The ability to source high-purity deuterated building blocks allows companies to develop "me-better" drugs that offer distinct clinical advantages over generic competitors. From a procurement perspective, establishing a reliable supply of these specialized chemicals mitigates the risk of raw material shortages that often plague commodity intermediates. The synthetic routes described are designed to be robust and scalable, utilizing reagents that are commercially available or can be produced in bulk. This ensures continuity of supply even as demand for the final drug substance increases. Furthermore, the enhanced metabolic stability of the resulting API can lead to reduced total daily doses, which indirectly lowers the volume of active ingredient required per patient treatment course, optimizing inventory management and reducing overall material costs over the product's lifecycle.
- Cost Reduction in Manufacturing: While deuterated reagents may carry a premium, the overall cost of goods can be optimized through efficient synthetic design. The use of convergent synthesis strategies, such as the Suzuki coupling described, minimizes the number of linear steps required to reach the final intermediate. This reduction in step count translates to lower labor costs, reduced solvent consumption, and decreased waste generation. Additionally, the improved stability of the deuterated molecule can simplify formulation and storage requirements, further driving down downstream processing expenses. By eliminating the need for complex late-stage modifications, manufacturers can achieve significant cost savings in the long run, making the investment in deuterated technology financially viable for large-scale production.
- Enhanced Supply Chain Reliability: Diversifying the supplier base to include manufacturers capable of handling isotopic chemistry strengthens the overall resilience of the supply chain. The protocols outlined in the patent rely on standard industrial equipment, such as stainless steel reactors capable of handling pressure for hydrogenation steps, which are common in fine chemical facilities. This compatibility means that production can be easily transferred between qualified contract development and manufacturing organizations (CDMOs) without the need for specialized, hard-to-source infrastructure. Ensuring a steady flow of these critical intermediates prevents bottlenecks in the API manufacturing schedule, allowing for consistent delivery of the final drug product to the market and maintaining patient access to essential gout therapies.
- Scalability and Environmental Compliance: The synthetic pathways presented are amenable to green chemistry principles, which is increasingly important for regulatory compliance and corporate sustainability goals. Reactions are typically conducted in common organic solvents that can be recovered and recycled, minimizing environmental impact. The catalytic nature of the key coupling steps ensures high atom economy, reducing the generation of hazardous byproducts. As production scales from kilograms to tons, these efficiency gains become more pronounced, facilitating easier regulatory approval for the manufacturing process. A scalable and environmentally compliant process reduces the risk of regulatory delays and ensures that the supply chain can meet global demand without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterated 2-phenylthiazole compounds. These insights are derived directly from the patented methodologies and are intended to clarify the feasibility and benefits of adopting this technology for your pharmaceutical development pipeline. Understanding these details helps stakeholders make informed decisions about sourcing and process development.
Q: What is the primary advantage of deuterated 2-phenylthiazole compounds over standard Febuxostat?
A: The substitution of hydrogen with deuterium strengthens the carbon-deuterium bond, which significantly reduces the rate of metabolic oxidative degradation. This kinetic isotope effect leads to improved pharmacokinetic profiles, including extended half-life and potentially reduced dosing frequency, while maintaining the potent xanthine oxidase inhibitory activity required for treating gout and hyperuricemia.
Q: How is the deuterium label introduced during the manufacturing process?
A: The patent describes two primary strategies for isotopic incorporation. The first method utilizes Suzuki-Miyaura cross-coupling reactions where a triflated intermediate reacts with commercially available deuterated phenylboronic acids. The second approach involves catalytic hydrogen-isotope exchange, where a brominated precursor is subjected to deuterium gas in the presence of a palladium catalyst, allowing for selective replacement of hydrogen atoms at specific aromatic positions.
Q: Are these intermediates scalable for commercial API production?
A: Yes, the synthetic routes outlined utilize robust and well-established chemical transformations such as thiazole cyclization, Vilsmeier-Haack formylation, and palladium-catalyzed couplings. These reactions are amenable to scale-up from kilogram to multi-ton quantities, provided that appropriate safety measures are taken for handling reagents like triflic anhydride and deuterium gas, ensuring a reliable supply chain for downstream drug substance manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated 2-Phenylthiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of deuterated intermediate meets the exacting standards required for clinical and commercial use. Our expertise in palladium-catalyzed reactions and isotopic labeling positions us as a strategic partner capable of delivering complex molecules with precision and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce time-to-market for your gout therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term business goals. Let us help you secure a stable and cost-effective source for these advanced pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
